Sterility Assurance Level Concepts
Sterility Assurance Level, or SAL, is the quantitative expression of the probability that a single viable microorganism remains on a product after sterilization. It does not mean absolute sterility. It defines the statistical confidence associated with a validated sterilization process.
In pharmaceutical manufacturing, terminal sterilization processes are typically required to achieve an SAL of 10⁻⁶, representing a maximum probability of one non-sterile unit in one million processed units.
SAL is therefore a probabilistic concept based on validated microbial reduction, not an observational test of sterility.
Definition of SAL
Sterility Assurance Level is defined as:
The probability of a single viable microorganism surviving a sterilization process on an individual item.
An SAL of:
- 10⁻¹ represents a one in ten probability of survival
- 10⁻³ represents a one in one thousand probability
- 10⁻⁶ represents a one in one million probability
Regulated sterile pharmaceutical products typically require SAL 10⁻⁶ for terminal sterilization processes.
Log Reduction and Sterility Assurance
Sterility assurance is achieved through logarithmic reduction of microbial populations during sterilization. Microbial inactivation is generally log-linear, meaning each incremental exposure reduces the population by a constant factor.
A 1-log reduction represents a tenfold reduction in viable organisms, corresponding to approximately 90 percent inactivation.
A 6-log reduction represents a one million-fold reduction, corresponding to approximately 99.9999 percent inactivation.
Log reduction alone, however, does not define the Sterility Assurance Level. The total lethality required depends on the initial microbial population present on the product.
Mathematical Relationship Between Bioburden, Log Reduction, and SAL
If:
- N₀ = initial microbial population per unit
- L = total log reduction achieved
- N = expected number of survivors per unit
Then:
N = N₀ × 10⁻ᴸ
Because SAL represents the probability of survival per unit, the relationship may be expressed as:
SAL = N₀ × 10⁻ᴸ
Rearranging:
L = log₁₀(N₀) − log₁₀(SAL)
This equation defines the mathematical connection between starting bioburden, achieved lethality, and sterility assurance. The relationship between microbial population, log reduction, and sterility assurance can be visualized on a logarithmic scale.

Example Calculation
Assume:
Initial bioburden = 10³ organisms per unit
Target SAL = 10⁻⁶
L = 3 − (−6)
L = 9 logs required
This means the sterilization process must achieve a 9-log reduction to ensure a theoretical probability of survival of 10⁻⁶.
Expressed differently:
Initial load (10³)
Required probability margin (10⁶)
Total lethality required = 10⁹ reduction
This quantitative relationship forms the scientific basis for sterilization validation and overkill methodology.
Overkill and Bioburden-Based Approaches
Two primary validation approaches are used in terminal sterilization:
Overkill Method
The process is validated to inactivate a highly resistant biological indicator population, typically demonstrating at least a 12-log reduction of spores. This approach provides a margin of safety beyond expected product bioburden.
The overkill approach is commonly used for moist heat sterilization. In an overkill validation approach, lethality is demonstrated against a defined biological indicator population to provide a safety margin beyond expected product bioburden.
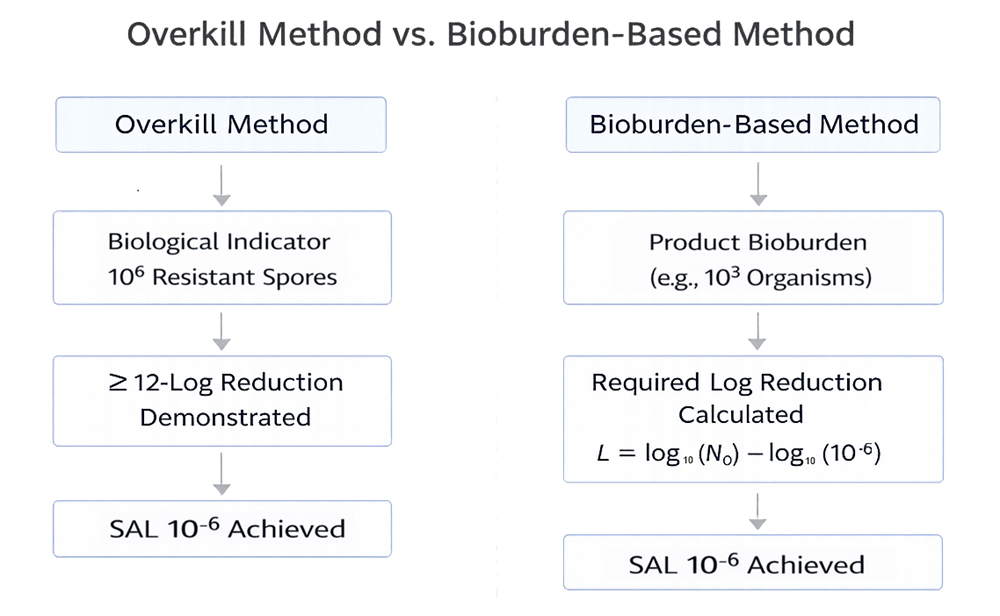
Bioburden-Based Method
The process lethality is calculated based on measured product bioburden and microbial resistance characteristics. This approach may be used when product characteristics limit exposure conditions.
Both approaches must demonstrate achievement of the required SAL.
SAL and Sterility Testing
Sterility testing does not establish SAL.
Sterility tests sample a limited number of units and are incapable of statistically confirming a probability of 10⁻⁶. SAL is demonstrated through validated process control and documented lethality, not finished product testing.
This distinction is critical from a regulatory perspective.
Regulatory Expectations
Regulatory authorities require sterilization processes to:
- Be scientifically validated
- Demonstrate achievement of defined SAL
- Include biological indicator qualification where applicable
- Be supported by documented lethality calculations
- Be subject to periodic requalification
Applicable references include:
- 21 CFR Part 211
- EU GMP Annex 1
- ISO 17665 for moist heat sterilization
- ISO 11135 for EO sterilization
- ISO 11137 for radiation sterilization
SAL must be justified in the validation report and supported by data.
SAL in Aseptic Processing
Aseptic processing does not rely on terminal lethality to achieve SAL 10⁻⁶. Instead, sterility assurance is achieved through:
- Sterile filtration
- Environmental control
- Operator qualification
- Media fills
- Continuous monitoring
While the target assurance level remains equivalent, the mechanism of achieving confidence differs fundamentally from terminal sterilization.
Lifecycle Control
Achievement of SAL is not a one-time demonstration. Ongoing control requires:
- Routine cycle monitoring
- Biological indicator trending
- Review of critical process parameters
- Change control assessment
- Periodic requalification
Maintaining sterility assurance is a lifecycle obligation, not an initial validation milestone.
