Requalification and Continued Verification
Steam sterilization systems must remain in a validated state throughout their operational lifecycle. Initial qualification establishes process capability; continued verification ensures that capability is preserved over time.
Regulatory authorities expect sterilization processes to be maintained under ongoing control, supported by documented review and risk-based reassessment.
1. Regulatory Expectation
Requalification and periodic review are supported by:
- 21 CFR 211.113 – Control of microbiological contamination
- 21 CFR 211.68 – Control of automated systems
- EU GMP Annex 1 – Requirement for periodic review and revalidation
- ISO 17665 – Ongoing process monitoring and requalification
These frameworks require that sterilization processes remain validated following changes or over defined time intervals.
2. Continued Process Verification
Continued verification involves routine monitoring and periodic review of sterilization performance.
2.1 Routine Cycle Monitoring
Each production cycle should be evaluated for:
- Achievement of exposure temperature
- Minimum exposure time
- Pressure–temperature correlation
- Alarm events
- Cycle deviations
Routine review confirms that validated parameters are consistently achieved.
2.2 Trend Analysis
Trend evaluation should include:
- F₀ values at cold spot
- Come-up time consistency
- Alarm frequency
- Vacuum leak rate trends
- Steam quality monitoring results
Unfavorable trends may indicate emerging loss of control.
3. Triggers for Requalification
Requalification is not automatically identical to initial PQ. It is driven by change or risk. Typical triggers include:
- Control system modification
- Steam supply changes
- Chamber repair or modification
- Drain relocation
- Load configuration changes
- Significant maintenance intervention
- Repeated cycle deviations
- Adverse trend analysis
Each trigger requires documented impact assessment.
4. Risk-Based Scope Determination
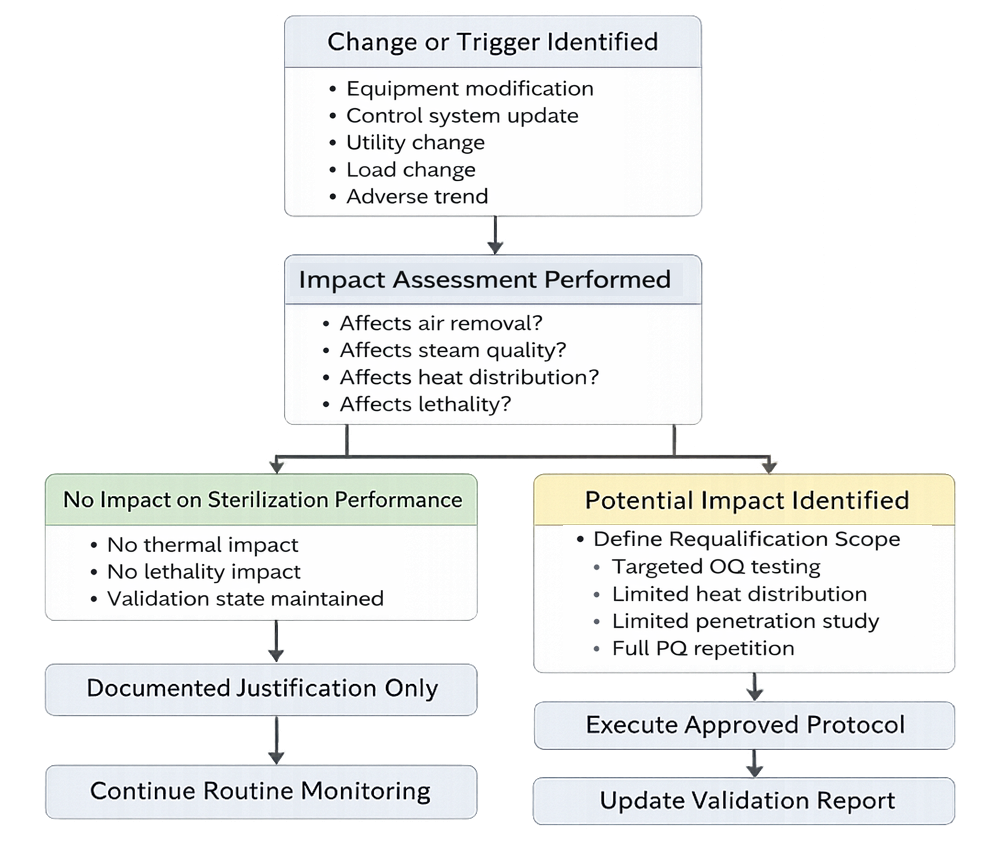
The extent of requalification depends on:
- Nature of the change
- Impact on sterilization lethality
- Affected components
- Historical performance data
Possible outcomes:
- Documentation update only
- Targeted verification testing
- Partial heat distribution study
- Limited heat penetration study
- Full requalification (IQ/OQ/PQ repetition)
Requalification scope must be scientifically justified and documented. Automatic repetition of full PQ without risk evaluation is neither efficient nor necessary.
5. Periodic Requalification
Some facilities establish defined periodic requalification intervals. Periodic requalification may include:
- Repeat heat distribution study
- Limited heat penetration runs
- Steam quality reassessment
- Review of historical performance
Interval justification should be risk-based and supported by documented review.
6. Documentation Requirements
Requalification and continued verification require:
- Change control documentation
- Impact assessment
- Approved protocol (if testing required)
- Updated validation report
- Management approval
All assessments must demonstrate continued assurance of sterility.
7. Lifecycle Perspective
Validation is not static. It is a controlled lifecycle.
Initial qualification establishes capability.
Routine monitoring confirms stability.
Trend analysis identifies drift.
Requalification restores documented assurance when change or risk justifies it.
A sterilization process is considered validated only when it is actively maintained in a state of control.
