Biosafety Cabinets and Laminar Flow Hoods: Certification and Qualification
1. Purpose and Scope
This article defines the certification, qualification, and lifecycle control expectations for biosafety cabinets and laminar flow hoods used as ISO 5 primary engineering controls in regulated environments.
The scope includes the technical content of certification testing, the structured qualification approach including Installation Qualification and Operational Qualification, and the conditions under which Performance Qualification may be required based on documented risk assessment. It also addresses ongoing control, requalification triggers, and integration with change management and maintenance programs.
The objective is to clarify how these systems are demonstrated to be fit for intended use and how that validated state is maintained over time. Particular emphasis is placed on distinguishing standardized certification from process-specific qualification and on aligning lifecycle controls with regulatory expectations and risk-based contamination control strategy.
2. Certification: Scope, Standards, and Technical Content
Certification and qualification represent different layers of control within the lifecycle of primary engineering controls.
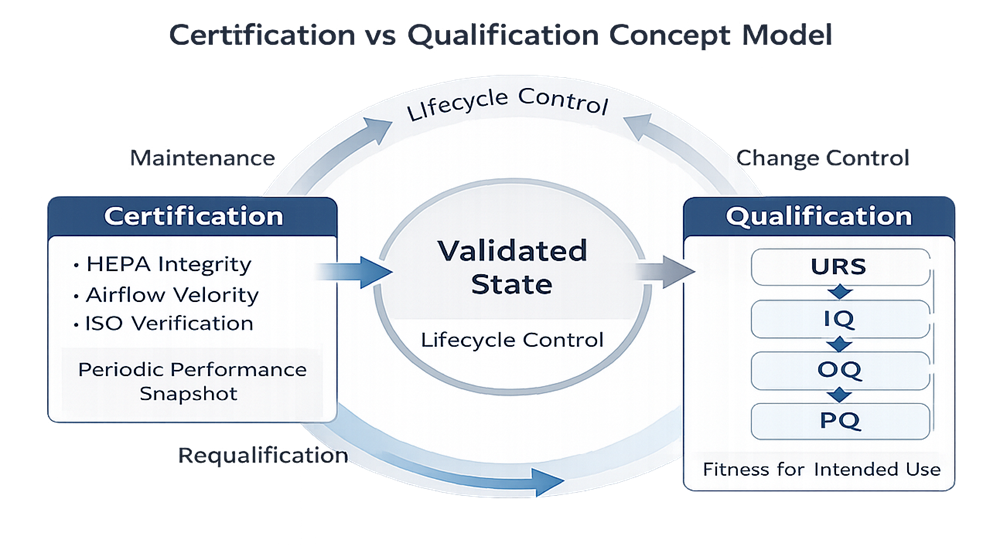
Certification is formal field performance testing conducted by trained and qualified personnel using calibrated instrumentation. It verifies that a biosafety cabinet or laminar flow hood meets recognized technical performance criteria at the time of testing. Certification evaluates airflow, filtration integrity, and containment parameters against defined standards. It does not evaluate process workflow or demonstrate fitness for a specific application.
For Class II biosafety cabinets, certification is typically performed in accordance with recognized field test standards such as NSF/ANSI 49 or equivalent manufacturer and regulatory procedures. Laminar flow hoods are certified against defined airflow velocity, HEPA integrity, and ISO classification expectations consistent with applicable cleanroom standards.
Certification is required at installation and at defined periodic intervals. It is also required following relocation, HEPA filter replacement, major repair, or any modification that may affect airflow balance or containment.
Certification Test Matrix – Laminar Flow Hoods vs Class II Biosafety Cabinets
| Test Parameter | Laminar Flow Hood (Certification Scope) | Class II Biosafety Cabinet (Certification Scope) |
|---|---|---|
| HEPA Filter Integrity Testing | Aerosol challenge and scan of supply HEPA filter | Aerosol challenge and scan of supply and exhaust HEPA filters |
| Air Velocity Measurement | Average work zone velocity and uniformity across filter face or work plane | Downflow velocity profile and inflow velocity at sash opening |
| Airflow Visualization | Directional airflow confirmation within work zone | Containment smoke pattern testing at sash opening and internal airflow visualization |
| Particle Count Verification | ISO 5 classification verification under defined static conditions | ISO 5 classification verification under defined static conditions |
| Alarm and Indicator Verification | Not typically applicable unless equipped | Verification of airflow alarms, interlocks, and indicator functionality |
| Sash Height Verification | Not applicable | Verification of certified operating sash height and corresponding inflow velocity |
| Exhaust Performance Verification | Not applicable | Verification of exhaust airflow and duct static pressure where applicable |
Certification confirms technical performance. It does not replace qualification.
2.1 Laminar Flow Hood Certification
Certification of a laminar flow hood focuses on verifying stable unidirectional airflow and HEPA filter integrity.
HEPA filter integrity testing is performed using an aerosol challenge and scan method to identify leaks in the filter media or seal. Any detected leakage requires corrective action and retesting.
Air velocity measurements are conducted across the filter face or work plane to confirm average velocity and uniformity within specified limits. Insufficient velocity compromises displacement of room air. Excessive velocity may introduce turbulence.
Airflow visualization may be performed to confirm directional stability and absence of backflow within the work zone.
Where the hood is used as an ISO 5 work area in a regulated environment, particle counting may be performed to confirm compliance with ISO 14644 classification requirements under defined conditions.
General inspection includes verification of blower performance, prefilter condition, lighting, structural integrity, and potential influence from adjacent room airflow sources.
The objective is to confirm that the hood provides a stable ISO 5 airflow field under static certification conditions.
2.2 Biosafety Cabinet Certification
Certification of a Class II biosafety cabinet evaluates both product protection and containment performance.
HEPA filter integrity testing is performed on both supply and exhaust filters using an aerosol challenge and scan method.
Downflow velocity is measured to verify adequate product protection. Inflow velocity at the sash opening is measured to verify containment performance. These parameters must remain within defined tolerances and maintain proper airflow balance.
Smoke pattern testing is performed to visualize airflow at the front opening, along the sash plane, and within the work zone. This confirms proper formation of the air curtain and absence of reflux or escape patterns.
Alarm systems, airflow indicators, and interlocks are verified to ensure proper response to out-of-specification conditions. Sash height is measured to confirm operation at the certified opening dimension, since inflow velocity is dependent on opening geometry.
Certification demonstrates that the cabinet meets accepted field performance criteria for airflow and containment at the time of testing. It does not evaluate operator technique, staging configuration, or process-specific aseptic controls. Those elements are addressed through qualification.
3. Qualification Strategy
Certification alone verifies standardized field performance. Qualification is broader. It establishes documented evidence that the specific hood or cabinet, as installed, integrated, and used in your facility, supports its intended aseptic function under defined operating conditions.
The depth of qualification should be risk-based. In low-risk nonhazardous LFH applications, qualification may be limited to documented installation review and verification of certified performance. In higher-risk applications involving hazardous materials, aseptic compounding, or critical sterile manufacturing steps, structured IQ and OQ are expected, and a process-integrated performance assessment may be warranted.
Qualification demonstrates fitness for intended use. It is not a repetition of certification. It evaluates installation context, facility integration, and operational use. Qualification follows a structured lifecycle extending beyond periodic certification.
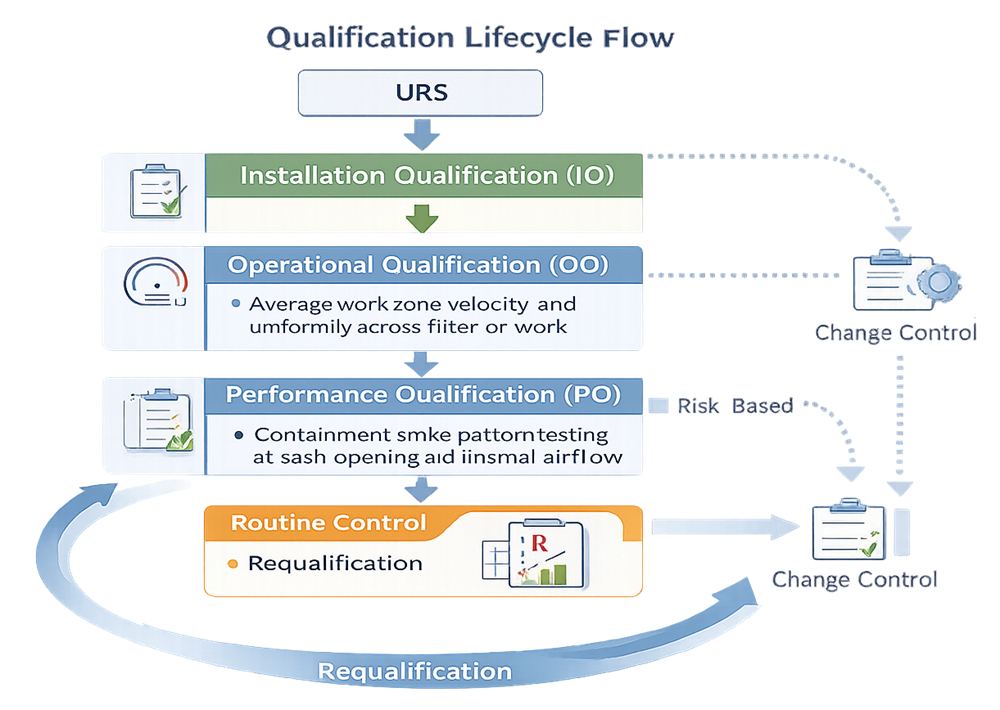
3.1 User Requirements Specification
The URS defines why the unit is being installed and what it must achieve. It should document:
• Intended process application
• Types of materials handled and associated hazard profile
• Required protection functions: product only or product and personnel
• Required ISO classification at the work zone
• Exhaust configuration expectations and facility interface
The URS establishes the benchmark against which installation and operational performance are assessed.
3.2 Installation Qualification
Installation Qualification confirms that the unit is correctly installed within the facility and matches the approved configuration.
IQ should verify:
• Correct model, type, and configuration per URS
• Proper placement relative to doors, supply diffusers, return grilles, and traffic patterns
• Electrical supply, grounding, and power stability
• Exhaust configuration, canopy or duct connection where applicable
• Presence and function of instrumentation, alarms, and indicators
• Labels, safety notices, and operating limits
The objective is to confirm that the cabinet or hood is physically and functionally integrated into the facility environment in a manner consistent with design intent.
3.3 Operational Qualification
Operational Qualification verifies that the unit performs within defined technical parameters under controlled test conditions. OQ confirms that airflow, filtration, alarms, and containment-related functions operate within established limits before the unit is released for routine use. OQ typically includes:
• Air velocity measurement, including downflow and inflow for BSCs
• HEPA filter integrity testing
• Airflow visualization to confirm directional stability
• Alarm and control function verification
• Particle counts in the work zone under defined static or controlled dynamic conditions
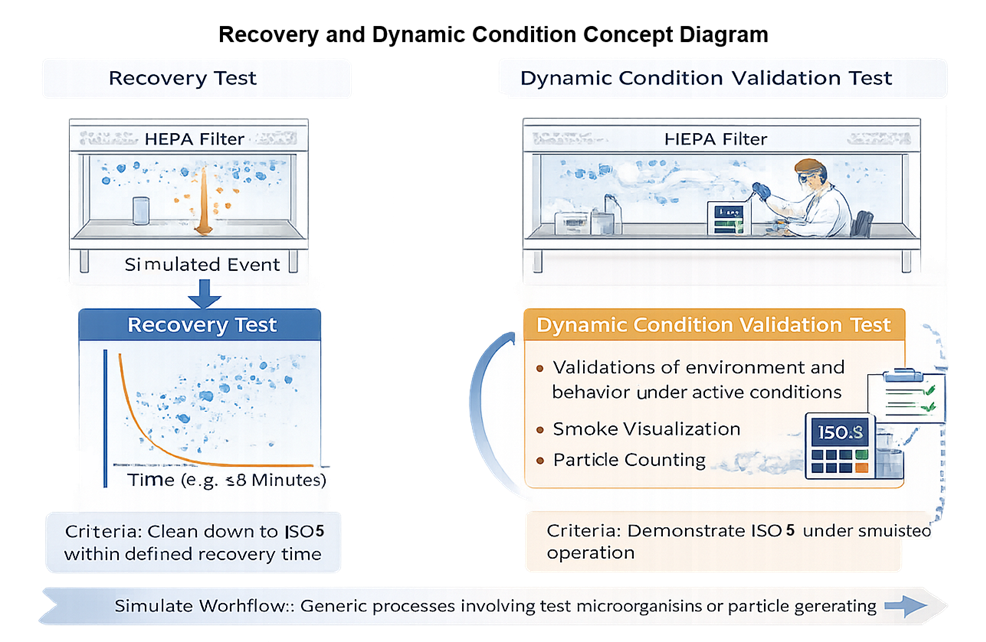
• Airflow recovery testing following simulated disturbance
Recovery testing and dynamic condition validation are performed using non-viable particle monitoring to evaluate airflow control within the ISO 5 work zone. Recovery testing determines the time required for the work area to return to specified ISO 5 particle limits after a defined disturbance, such as arm intrusion, staged material placement, or other controlled disruption. A calibrated particle counter measures airborne particle concentrations at predetermined locations, and the elapsed time to re-establish compliant levels defines the recovery performance. Dynamic condition validation is conducted during simulated routine manipulations while non-viable particle counts are monitored and airflow visualization, typically with smoke, is used to assess directional stability and containment behavior. These tests demonstrate that ISO 5 conditions are maintained during active workflow; viable microbiological sampling is not part of airflow recovery testing and instead belongs to the environmental monitoring program.
Acceptance criteria must be established in advance and traceable to risk assessment, manufacturer specifications, and applicable standards. OQ confirms that airflow and filtration functions operate within validated limits and that the system can re-establish control following normal operational disturbances.
3.4 Performance Qualification
Whether a formal PQ is required depends on intended use and documented risk assessment. For cabinets supporting critical aseptic processing steps, sterile compounding, or hazardous drug handling programs, a documented performance evaluation under realistic operating conditions is appropriate. Where the risk assessment concludes that the application presents low exposure and contamination risk, qualification may reasonably conclude with documented OQ and procedural controls, provided that justification is recorded.
When required, PQ is less about repeating technical measurements and more about demonstrating airflow performance under actual use conditions. In practice, PQ typically centers on:
• Airflow visualization studies, commonly using smoke, performed under dynamic conditions
• Environmental qualification of the work zone under representative operator activity
• Simulated routine manipulations using typical load configurations
• Worst-case staging scenarios that challenge airflow paths
• Verification that critical sites remain in first air during active manipulation
Dynamic particle monitoring may be performed to confirm that ISO 5 conditions are sustained during operation, not only under static test conditions.
PQ must be executed in accordance with previously approved SOPs that define:
• Test methods and acceptance criteria
• Smoke study methodology and documentation requirements
• Definition of dynamic conditions
• Operator qualification requirements
• Response to observed airflow disturbance
The objective is to confirm that airflow integrity and, where applicable, containment performance are maintained during realistic workflow.
The scope and necessity of PQ must be justified through formal risk assessment aligned with the cabinet’s intended use and process criticality.
4. Ongoing Control and Requalification
Initial qualification establishes documented fitness for intended use. Sustained compliance, however, depends on maintaining that state through structured lifecycle control. Biosafety cabinets and laminar flow hoods are dynamic airflow systems. Filter loading, mechanical wear, facility HVAC changes, and operational practices can degrade performance over time. Ongoing control ensures that certified performance and qualified use conditions remain aligned.
Ongoing lifecycle control includes:
• Routine certification at defined intervals to verify airflow and filtration performance
• Preventive maintenance of blowers, motors, seals, and instrumentation
• Defined start-up stabilization time before use to re-establish airflow stability
• Cleaning and disinfection practices validated not to compromise airflow or materials of construction
• Change control impact assessment for facility, equipment, or process modifications
These controls bridge the gap between periodic certification events and daily operational use.
Requalification is required when changes or events could reasonably affect airflow balance, containment, or ISO 5 performance. Triggers typically include:
• Relocation of the unit within or between rooms
• HEPA filter replacement or repair
• Exhaust system modification or ductwork changes
• Control system or alarm setpoint changes
• Significant process or workflow changes that alter staging or manipulation patterns
Requalification scope should be risk-based and may range from targeted OQ testing to partial or full requalification, depending on the nature and impact of the change.
5. Regulatory and Standards Context
Certification and qualification of biosafety cabinets and laminar flow hoods must be aligned with applicable regulatory requirements and recognized consensus standards. While regulations do not prescribe specific airflow velocities or test methods, they require documented control of aseptic environments and demonstrable evidence that engineering controls consistently support sterility assurance and, where applicable, personnel protection.
Under 21 CFR 211, sterile drug manufacturing must be performed under conditions that prevent microbiological contamination. This includes maintaining suitable air quality at critical processing locations and documenting the control of equipment used in aseptic operations. Engineering controls that establish ISO 5 conditions at the point of use fall within this requirement.
USP <797> and USP <800>, where applicable, define expectations for primary engineering controls used in sterile compounding and hazardous drug handling. These chapters address certification frequency, containment requirements, and operational controls necessary to maintain environmental and personnel protection.
ISO 14644 establishes cleanroom classification criteria, including particle concentration limits for ISO 5 environments. While ISO standards define environmental classification methodology, they do not replace process-specific qualification or validation requirements.
For Class II biosafety cabinets, certification testing is typically performed in accordance with NSF/ANSI 49 or equivalent field performance standards. These standards define containment and airflow test methods but do not address facility-specific qualification strategy.
Regulatory compliance therefore requires more than periodic certification. Facilities must demonstrate through documented qualification, risk assessment, and lifecycle control that these systems are appropriate for their intended use and remain in a validated state over time.
6. Summary
Biosafety cabinets and laminar flow hoods used as ISO 5 primary engineering controls require more than periodic certification to ensure ongoing compliance and sterility assurance. Certification verifies that airflow and filtration meet defined technical performance criteria at a specific point in time. It is equipment-focused and does not assess process use.
Qualification establishes documented evidence that the installed unit, within its specific facility context, is fit for its intended use. Installation Qualification confirms proper integration into the facility. Operational Qualification verifies airflow, filtration, alarms, and performance limits. Performance Qualification, when justified by risk assessment, demonstrates that airflow integrity and containment are maintained during actual workflow under dynamic conditions.
Sustained compliance depends on structured lifecycle control. Routine certification, preventive maintenance, documented startup practices, change control, and risk-based requalification ensure that validated performance is preserved as equipment, facility conditions, and processes evolve.
In regulated environments, airflow performance must be demonstrated, documented, and maintained. Certification confirms technical function. Qualification confirms functional fitness. Lifecycle control preserves both.
