Washer Qualification
Automated parts washers used in pharmaceutical manufacturing and laboratory environments must be qualified to demonstrate that the equipment operates reliably and consistently supports validated cleaning processes. Qualification activities verify proper installation, confirm operational functionality, and demonstrate that the washer can effectively clean components under defined conditions.
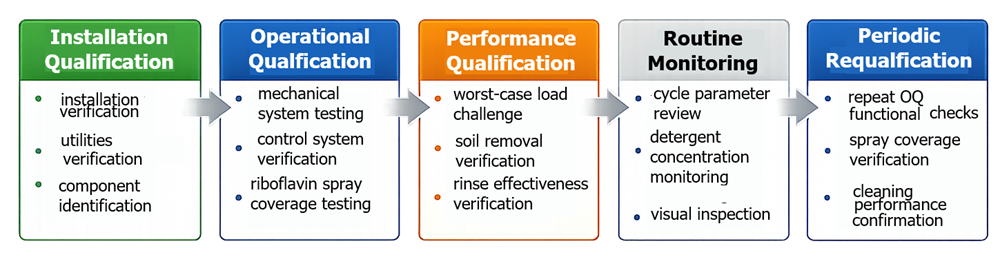
Washer qualification typically follows the standard equipment qualification lifecycle consisting of Installation Qualification, Operational Qualification, and Performance Qualification.
The diagram below illustrates the relationship between Installation Qualification, Operational Qualification, and Performance Qualification within the overall validation lifecycle.
1. Installation Qualification
Installation Qualification verifies that the washer is installed according to manufacturer specifications, engineering drawings, and facility standards. This stage confirms that the equipment configuration and utility connections support its intended operation. Typical Installation Qualification checks include:
- equipment identification and model verification
- chamber construction materials and surface finish
- rack systems and loading configuration
- spray arm assemblies
- detergent dosing systems
- filtration components
- heating systems or heat exchangers
- control system hardware
Utility connections are also verified, including:
- electrical power supply
- water supply connections including USP purified and/or WFI
- drain connections
- compressed air where applicable
- steam supply for heating systems
Documentation such as manuals, wiring diagrams, and piping drawings should be reviewed and included in the qualification package.
2. Operational Qualification
Operational Qualification demonstrates that the washer operates correctly and consistently across its defined operating ranges. Testing focuses on mechanical functionality, control system performance, and verification of critical process parameters.
2.1 Mechanical System Verification
Mechanical testing confirms that key washer components function correctly during operation. Typical tests include:
- spray arm rotation verification
- circulation pump operation
- flow distribution through spray arms
- rack positioning and stability
- detergent injection functionality
- filter installation and accessibility
These tests confirm that the mechanical cleaning system operates as designed.
2.2 Control System and Cycle Verification
Automated washers typically use PLC-based control systems with predefined cleaning cycles. OQ testing must verify that these cycles execute correctly and maintain defined process parameters. Testing typically includes:
- verification of programmed cycle steps
- confirmation of wash and rinse sequencing
- verification of detergent injection timing
- verification of drying cycle operation
- alarm testing for abnormal conditions
- safety interlock testing
Critical parameters monitored during testing often include:
- wash temperature
- rinse temperature
- cycle duration
- detergent dosing events
These tests confirm that the washer control system consistently executes programmed cleaning cycles.
2.3 Riboflavin Spray Coverage Test
One of the most important operational tests for parts washers is verification of spray distribution within the chamber. This is typically performed using a riboflavin coverage test. Conceptual diagram of riboflavin spray coverage testing in a parts washer showing application of riboflavin solution and ultraviolet inspection used to identify areas with insufficient spray coverage.

Riboflavin is a fluorescent compound that becomes highly visible when exposed to ultraviolet light. During testing, a riboflavin solution is applied to the surfaces of representative equipment components and rack locations. The washer cycle is then executed under defined conditions. After completion of the cycle, components are inspected under UV light.

Interpretation of results is based on fluorescence:
- areas where riboflavin has been removed indicate adequate spray coverage
- areas where fluorescence remains indicate insufficient spray contact
Riboflavin testing helps identify:
- spray shadowing caused by load configuration
- insufficient spray pressure
- limitations in spray arm distribution
- improper rack positioning
Testing is typically performed using worst-case load configurations to challenge the spray distribution system.
3. Performance Qualification
Performance Qualification demonstrates that the washer can consistently clean equipment components under routine operating conditions. Testing focuses on cleaning effectiveness and rinse efficiency using representative loads. PQ studies typically challenge the washer using conditions that represent the most difficult cleaning scenarios.
3.1 Soil Removal Challenge Testing
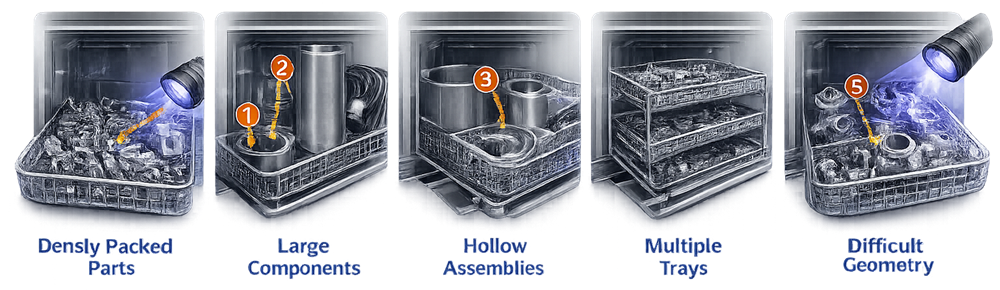
Cleaning effectiveness is evaluated by applying defined soils or residues to equipment components before washing. Testing should incorporate worst-case challenge conditions representing the most difficult cleaning scenarios that may occur during routine operation. Worst-case conditions may include the most difficult-to-clean residues, maximum soil load, complex component geometry, or load configurations that limit spray access. These conditions provide a conservative challenge to demonstrate that the washer cleaning cycle is capable of removing residues under the most demanding circumstances.
The illustration below shows examples of worst-case load configurations used to challenge washer cleaning performance during Performance Qualification studies. Such configurations intentionally create conditions that may restrict spray distribution, including densely packed components, large parts that block spray paths, hollow assemblies, stacked trays, and complex geometries. Evaluating cleaning performance under these challenging load arrangements helps demonstrate that the washer cycle can effectively remove residues even under the most demanding operating conditions.
Challenge materials may include:
- protein-based soils
- carbohydrate residues
- fatty or oily materials
- sticky or dried residues representing aged contamination
- standardized test soils used in washer validation studies
The washer cycle is executed and components are evaluated after cleaning. Evaluation methods may include:
- visual inspection for residue
- gravimetric measurements
- analytical residue testing
The objective is to demonstrate that the washer consistently removes representative soils.
3.2 Rinse Effectiveness Verification
Rinse effectiveness must be demonstrated to confirm that detergent residues and loosened soils are removed from equipment components during the rinse phase of the washer cycle. Verification of rinse performance is an important element of Performance Qualification because inadequate rinsing may leave cleaning agents or dissolved contaminants on component surfaces.
Rinse effectiveness testing evaluates whether the rinse phase of the washer cycle adequately removes detergent and residual contaminants following the wash step. Samples collected from the final rinse water are typically analyzed to confirm that detergent residues have been effectively removed.

The illustration below shows the general concept of rinse effectiveness verification during washer Performance Qualification. After the wash cycle is completed and the final rinse is executed, samples of the rinse water are collected and analyzed to confirm that detergents and dissolved residues have been removed from the system.
Common evaluation methods include the following analytical techniques.
- Conductivity Testing: Rinse water samples are collected during the final rinse stage and compared with the conductivity of the incoming rinse water supply. When detergent residues have been effectively removed, conductivity values of the final rinse water are expected to approach those of the supply water.
- Total Organic Carbon Testing: Total Organic Carbon analysis may be used to detect trace levels of organic contaminants remaining after the rinse cycle. TOC testing is particularly useful when cleaning processes involve organic residues or detergent formulations containing organic components.
- pH Monitoring: Measurement of pH in final rinse water can provide an additional indication of detergent removal. If detergent residues remain present, the pH of the rinse water may deviate from the pH of the supply water.
Together, these tests provide objective evidence that the rinse cycle effectively removes cleaning agents and residual contaminants from equipment components. Verification of rinse effectiveness supports demonstration that the washer cleaning process produces components suitable for reuse in pharmaceutical manufacturing operations.
3.3 Repeatability Studies
Cleaning performance must be demonstrated across multiple washer cycles to confirm consistent operation. Repeatability studies typically include:
- multiple consecutive cleaning cycles
- use of defined load configurations
- evaluation of cleaning results for each run
Consistent results across runs demonstrate reliable washer performance under routine operating conditions.
4. Acceptance Criteria
Acceptance criteria must be defined before execution of washer qualification protocols. These criteria establish the conditions that must be met to demonstrate that the washer is properly installed, operates as intended, and is capable of supporting validated cleaning processes. Acceptance criteria should be defined separately for each stage of qualification.
4.1 Installation Qualification Acceptance Criteria
Installation Qualification acceptance criteria confirm that the washer and associated systems are installed in accordance with manufacturer specifications, approved engineering documentation, and facility requirements. Typical acceptance criteria include:
- equipment model and identification match approved specifications
- chamber construction materials and surface finishes meet design requirements
- racks, spray arms, and internal components are installed correctly
- all required utilities are connected and available
- electrical, water, and drain connections comply with installation drawings
- equipment documentation, manuals, and drawings are available and reviewed
Successful completion of Installation Qualification demonstrates that the washer installation is suitable for operation and further qualification testing.
4.2 Operational Qualification Acceptance Criteria
Operational Qualification acceptance criteria confirm that the washer operates within defined operating ranges and that all functional components perform as intended. Typical acceptance criteria include:
- spray arms rotate freely and without obstruction during operation
- circulation pumps operate and deliver adequate flow to spray systems
- detergent dosing occurs at programmed stages of the cycle
- wash and rinse temperatures reach programmed setpoints
- programmed cycle steps execute in the correct sequence
- alarms and safety interlocks activate correctly under test conditions
- control system accurately displays and records process parameters
Successful completion of Operational Qualification demonstrates that the washer operates consistently and that critical cleaning parameters can be controlled during operation.
4.3 Performance Qualification Acceptance Criteria
Performance Qualification acceptance criteria demonstrate that the washer cleaning cycle consistently removes residues and effectively rinses equipment components under defined operating conditions. Typical acceptance criteria include:
- riboflavin coverage tests demonstrate complete removal of fluorescent residue from exposed surfaces
- challenge soils are removed from test components after the cleaning cycle
- rinse water parameters such as conductivity or TOC return to acceptable levels comparable to supply water
- no visible residues remain on cleaned components after washing and rinsing
- repeatability studies demonstrate consistent results across multiple runs
Successful completion of Performance Qualification provides documented evidence that the washer cleaning process performs effectively and consistently under routine operating conditions.
5. Qualification Documentation
Qualification documentation provides the records necessary to demonstrate that the washer has been properly evaluated and meets defined performance requirements. Typical qualification documentation includes:
- executed qualification protocols
- calibration records for measurement instruments
- raw test data and observations
- deviation documentation where applicable
- final qualification report
Completion of washer qualification provides documented evidence that the equipment operates within defined parameters and can reliably support validated cleaning processes.
