Instrument Categories
1. Introduction
Analytical instruments used in pharmaceutical laboratories vary significantly in complexity, function, and regulatory impact. Some instruments perform simple physical measurements, while others generate complex analytical data used to support product release, stability programs, process validation, and regulatory submissions.
Because of this diversity, laboratories must establish a structured approach for categorizing analytical instruments. Instrument categorization provides a consistent framework for determining qualification requirements, documentation expectations, calibration programs, and ongoing lifecycle controls.
A clear categorization model ensures that qualification activities are applied proportionally to the complexity and risk associated with each instrument. Simple measuring devices may require limited qualification activities, while complex computerized analytical systems require comprehensive validation and lifecycle management.
This article describes the principles used to categorize analytical instruments in regulated laboratory environments and explains how instrument categories support a risk-based qualification strategy.
2. Purpose of Instrument Categorization
Instrument categorization establishes a standardized method for grouping laboratory equipment according to functional complexity and operational characteristics. The purpose of this classification is to support consistent decision-making regarding qualification scope and lifecycle control.
Categorization allows organizations to apply structured governance to laboratory instrumentation while avoiding unnecessary qualification activities for simple devices that present minimal regulatory risk. Key objectives of instrument categorization include:
- establishing consistent qualification expectations across laboratory equipment
- aligning validation effort with instrument complexity and intended use
- supporting risk-based qualification strategies
- improving lifecycle management and documentation control
- ensuring regulatory compliance in GMP laboratory environments
Instrument categories provide the foundation for subsequent risk classification and qualification strategy determination.
3. Principles of Analytical Instrument Categorization
Analytical instrument categorization is typically based on the complexity of the instrument, the nature of the measurements performed, and the level of computerized control or data processing involved. Several factors are commonly considered during categorization:
3.1 Instrument Complexity
The structural and operational complexity of an instrument strongly influences the level of qualification required. Simple instruments that perform direct measurements require less extensive qualification than complex analytical systems with multiple integrated components. Factors affecting complexity may include:
- number of functional modules or subsystems
- automation level
- software-controlled operation
- internal data processing algorithms
- integration with other laboratory systems
Instruments with higher complexity generally require more extensive qualification and lifecycle management.
3.2 Measurement and Data Processing
Some instruments perform direct physical measurements, such as weight, temperature, or pressure. Other instruments perform analytical separation, detection, and complex data interpretation.
Instruments that generate processed analytical data used for quality decisions typically require more rigorous qualification and data integrity controls.
3.3 Computerized System Involvement
Modern analytical laboratories rely heavily on computerized instrumentation. Instruments may contain embedded firmware, integrated analytical software, or external workstation software used for data acquisition and analysis. The presence of computerized control introduces additional regulatory expectations related to:
- software validation
- electronic records
- audit trails
- data security
- backup and archival procedures
Instrumentation with extensive computerized functionality often requires additional validation activities beyond traditional equipment qualification.
4. Typical Analytical Instrument Categories
Although categorization models may vary between organizations, analytical instruments are commonly grouped into three general categories based on functional complexity.
The following diagram presents representative examples of laboratory instruments within each category. The examples illustrate how commonly used analytical equipment may be grouped according to functional complexity and operational characteristics within a laboratory governance framework.

4.1 Simple Measuring Devices
Simple measuring devices perform direct physical measurements and typically operate without complex data processing or integrated analytical software. Examples may include:
- thermometers
- pressure gauges
- timers
- tachometers
- basic pH meters without advanced data systems
These devices typically require calibration and basic installation verification but may not require extensive IQ/OQ/PQ qualification activities.
4.2 Stand-Alone Analytical Instruments
Stand-alone analytical instruments perform defined analytical functions but operate as relatively self-contained systems. They may include limited software or embedded control systems but do not typically involve complex networked data management. Examples include:
- ultraviolet visible spectrophotometers
- analytical balances
- refractometers
- conductivity meters
- simple gas analyzers
These instruments usually require formal qualification activities including installation verification and operational testing to demonstrate that the instrument operates according to manufacturer specifications.
4.3 Complex Analytical Systems
Complex analytical systems consist of multiple integrated modules and typically rely on computerized control, specialized analytical software, and electronic data management. Examples include:
- high-performance liquid chromatography systems
- gas chromatography systems
- liquid chromatography–mass spectrometry systems
- inductively coupled plasma spectrometry systems
- automated dissolution testing systems
These systems require comprehensive qualification and validation activities, including equipment qualification, analytical software validation, and data integrity controls.
5. Application of Instrument Categories in Laboratory Governance
Once analytical instruments are categorized, the classification supports consistent governance across the laboratory environment. Instrument categories influence several aspects of laboratory quality systems, including:
- qualification documentation requirements
- level of operational testing during OQ and PQ
- software validation requirements
- calibration and maintenance expectations
- documentation and recordkeeping practices
Categorization also supports efficient resource allocation by ensuring that validation effort is focused on instruments with the highest regulatory impact.
6. Relationship Between Instrument Categories and Risk Assessment
Instrument categorization groups analytical instruments according to functional complexity and technical characteristics. This classification establishes the structural framework used to organize laboratory equipment but does not by itself determine the required level of qualification. Determination of qualification scope requires evaluation of two primary factors:
- instrument complexity
- intended use of the instrument within the laboratory environment
Instrument complexity reflects the technical characteristics of the system, including the number of integrated components, level of automation, presence of analytical software, and degree of data processing performed by the instrument.
Intended use determines the regulatory impact of the data generated by the instrument. Instruments that generate data used for product release, stability studies, process validation, or regulatory submissions require a higher level of qualification and lifecycle control than instruments used for general laboratory support or research activities.
For example, a chromatography system used for release testing must operate under strict qualification and lifecycle control because the generated data directly supports product quality decisions. The same type of instrument used in early method development or research may operate under a reduced qualification framework because the data does not directly support regulated manufacturing decisions. Risk assessment integrates these two elements by evaluating:
- complexity of the instrument criticality of the generated data
- potential impact on product quality and patient safety
This combined evaluation allows laboratories to determine qualification strategies that are proportional to both technical complexity and regulatory impact.
The illustration below presents a conceptual model showing the relationship between instrument complexity and qualification effort. As instrument complexity increases, the depth of qualification testing, documentation, and lifecycle control typically increases to ensure that the instrument operates in a controlled and reliable state throughout its operational lifecycle.
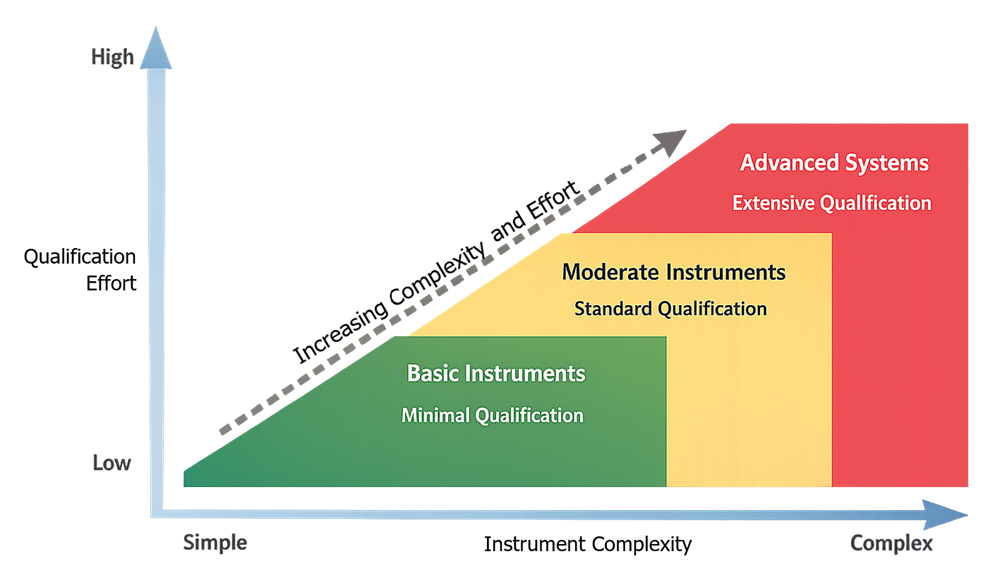
7. Summary
Instrument categorization provides the structural foundation for analytical instrument governance in regulated laboratories. By grouping instruments according to functional complexity and operational characteristics, organizations can establish consistent expectations for qualification, lifecycle control, and regulatory compliance.
Simple measuring devices, stand-alone analytical instruments, and complex analytical systems represent common classification levels used in pharmaceutical laboratories. These categories support risk-based qualification strategies while ensuring that validation activities remain proportional to instrument complexity and regulatory impact.
When combined with risk classification and qualification strategy determination, instrument categorization enables laboratories to implement a structured and efficient approach to analytical instrument lifecycle management.
