Sequential Validation Approaches
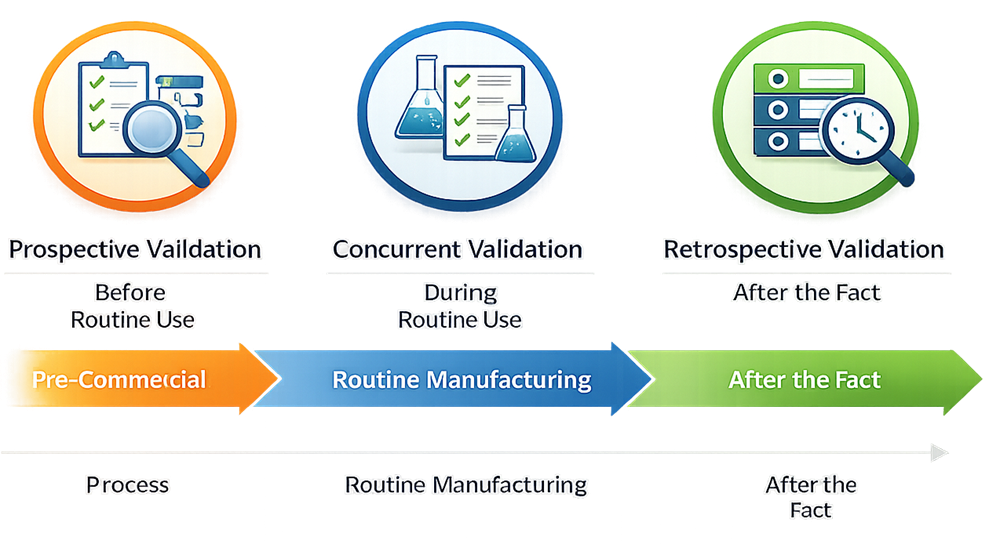
Validation activities may be executed using different approaches depending on the lifecycle stage of the process, the availability of data, and regulatory expectations. In regulated pharmaceutical and medical device environments, validation approaches are typically categorized as prospective, concurrent, or retrospective.
These approaches are not equivalent alternatives. Each applies under specific circumstances and carries different levels of regulatory risk and scrutiny.
Prospective Validation
Prospective validation is performed before a process is released for routine commercial use. It is the preferred and expected validation approach for new products, new processes, new equipment, and significant process changes.
Prospective validation establishes documented evidence that a process is capable of consistently producing product that meets predefined quality attributes and specifications prior to routine manufacturing.
This approach typically includes:
- Process design and risk assessment
- Equipment and system qualification IQ, OQ, and PQ
- Identification of critical process parameters and control strategies
- Defined acceptance criteria and planned data collection
Prospective validation provides the highest level of regulatory confidence and forms the baseline for process approval and routine production.
Concurrent Validation
Concurrent validation is performed during routine production while product is being manufactured for distribution. It is typically applied when prospective validation is not feasible, such as during limited production runs or when immediate product supply is required.
Under concurrent validation, process performance data are collected and evaluated in real time while batches are released under defined controls and oversight.
Key characteristics of concurrent validation include:
- Enhanced monitoring and data collection
- Defined batch release controls and justification
- Statistical evaluation of process performance
- Formal investigation of deviations or atypical results
Concurrent validation carries higher regulatory risk than prospective validation and requires strong scientific justification, risk assessment, and quality oversight.
Retrospective Validation
Retrospective validation is based on the evaluation of historical production data from processes that have been operating without formal validation documentation. It relies on the analysis of batch records, test results, deviations, and trends to assess process consistency and control.
Historically, retrospective validation was used to validate legacy processes. Current regulatory expectations significantly limit its applicability.
Retrospective validation typically involves:
- Review of historical batch and testing data
- Statistical analysis of process performance
- Assessment of variability, trends, and failures
- Determination of whether the process has demonstrated consistent control
If historical data do not adequately demonstrate process capability, additional prospective or concurrent validation activities are required.
Regulatory Considerations
Regulatory agencies strongly favor prospective validation as the standard approach. Concurrent validation is accepted under defined and justified circumstances. Retrospective validation is generally limited to legacy processes and is rarely acceptable for new or modified processes.
Selection of the validation approach must be justified based on:
- Process lifecycle stage
- Risk to product quality and patient safety
- Availability and quality of data
- Regulatory expectations
Summary
Prospective, concurrent, and retrospective validation represent different approaches to demonstrating process control based on timing and context. These approaches are not interchangeable. A structured, risk-based validation strategy selects the appropriate approach to ensure compliance, product quality, and long-term process control.
