Qualification and Verification of Facility Automation Systems
Facility automation systems that support GMP operations are subject to qualification and ongoing verification to ensure they perform as intended and continue to operate in a state of control. Qualification establishes documented evidence that system design, installation, and operation meet defined requirements. Ongoing verification confirms that validated performance is maintained throughout the system lifecycle.
Automation systems are qualified based on their intended use and impact, not on system complexity or vendor classification.
Systems in Scope
Facility automation systems subject to qualification and verification may include:
- Building Management Systems
- Environmental Monitoring Systems
- Utility monitoring and control systems
- Alarm and notification systems
- Data acquisition and reporting platforms
Interfaces between systems are considered part of the qualified scope where they affect control, monitoring, alarms, or data integrity.
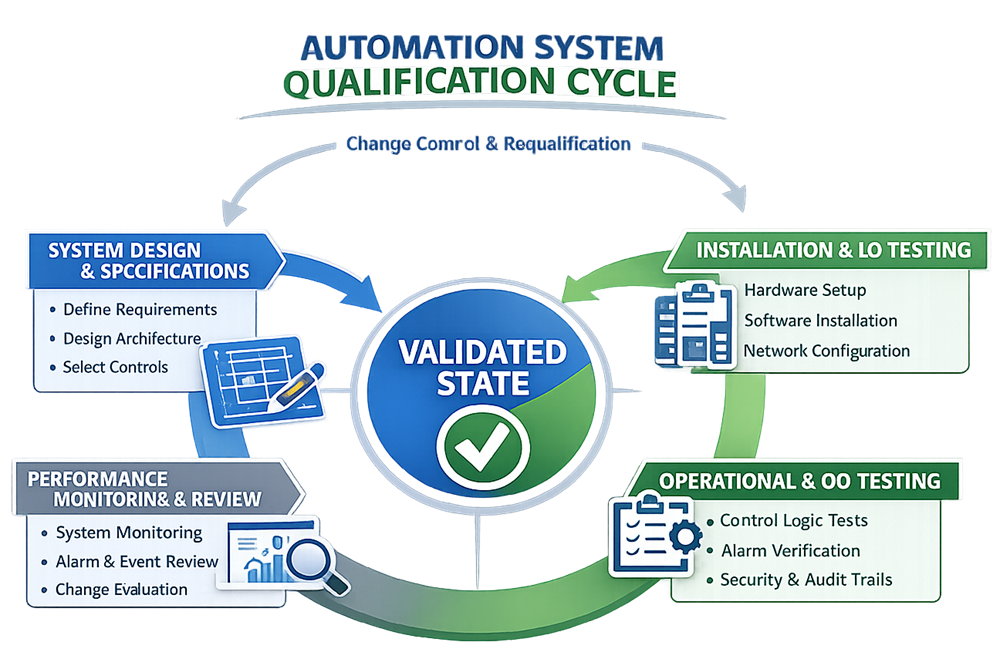
Qualification Strategy
Automation system qualification follows a lifecycle-based approach aligned with facility qualification activities. The level of qualification is determined by risk and system function.
Key principles include:
- Qualification is focused on intended use
- Not all system functions require the same level of testing
- Vendor documentation may be leveraged where appropriate
- Configuration, not hardware alone, is the primary qualification driver
Qualification activities are planned to avoid redundancy while ensuring coverage of critical functionality.
Design Qualification Considerations
Design Qualification confirms that the automation system architecture, functions, and controls are suitable for their intended GMP use.
Typical DQ considerations include:
- System boundaries and interfaces
- Control and monitoring responsibilities
- Data integrity features
- Alarm handling and notification concepts
- User access and role definitions
Design assumptions are documented early to prevent misalignment during later stages.
Installation Qualification Overview
Installation Qualification verifies that automation system components are installed in accordance with approved design and specifications.
IQ activities typically include:
- Verification of hardware installation
- Confirmation of network and communication setup
- Sensor and instrument installation checks
- Verification of software installation and version control
- Documentation of system configuration
IQ establishes the baseline from which operational testing is performed.
Operational Qualification Overview
Operational Qualification verifies that the automation system operates as intended under defined conditions.
OQ testing commonly addresses:
- Control logic and functional operation
- Monitoring and data acquisition
- Alarm generation and notification
- Security and access controls
- Audit trail functionality
- System response to defined challenge conditions
Testing is focused on functionality that supports GMP control and compliance.
Verification and Continued Performance
Following qualification, automation systems are subject to continued verification through routine operation, monitoring, and review.
Verification activities may include:
- Periodic review of system performance
- Alarm and event trend analysis
- Review of deviations and system-related investigations
- Assessment of change impact
- Periodic access and configuration review
Continued verification supports requalification decisions and lifecycle management.
Change Control and Requalification
Changes to automation systems are evaluated through formal change control. The scope of requalification is determined based on the nature of the change, affected functions, and historical performance.
Requalification may range from targeted functional testing to partial or full requalification depending on risk.
Practical Perspective
Automation system qualification should be structured, proportionate, and sustainable. Over-qualification creates unnecessary maintenance burden, while under-qualification exposes compliance risk.
Well-qualified systems require minimal intervention, generate reliable data, and support facility operations without drawing attention. That outcome is the objective.supports compliance quietly and consistently, which is its intended role.
