Analytical Instrument Installation Qualification Protocol
Installation Qualification establishes documented evidence that an analytical instrument and its supporting components are installed correctly, in accordance with approved design specifications, manufacturer recommendations, and applicable regulatory requirements. It confirms that the system delivered matches what was approved during Design Qualification and that the installation environment is suitable for operation.
Installation Qualification is a verification activity. It does not evaluate performance. It confirms that the foundation for testing and operation is correct and controlled.
1. Objective
The objective of Installation Qualification is to demonstrate that:
- the instrument and all components are received as specified
- installation is complete and consistent with design documentation
- utilities and environmental conditions meet defined requirements
- supporting systems are properly connected and configured
- documentation required for operation and maintenance is available
Completion of Installation Qualification establishes readiness for Operational Qualification.
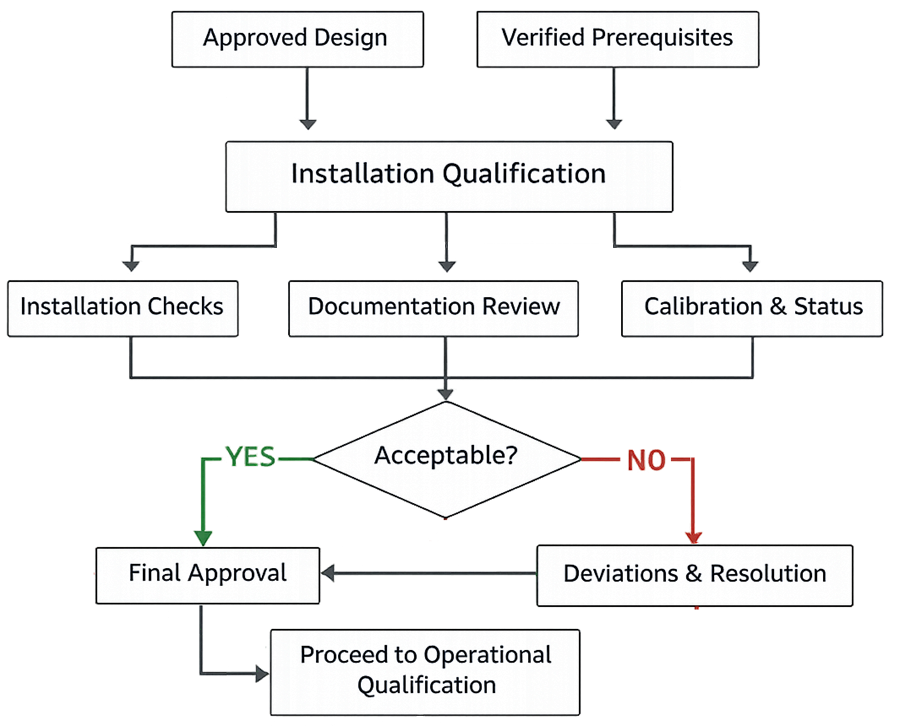
The diagram below illustrates the structured sequence of activities performed during Installation Qualification. It shows how prerequisite verification, installation checks, and documentation review lead to a controlled decision to proceed to Operational Qualification or to resolve identified deficiencies.
2. Scope
Installation Qualification applies to:
- analytical instrument hardware and modules
- associated computer systems and software
- data storage and network connections
- utilities required for operation
- supporting accessories and peripherals
The scope must align with the system boundary defined during Design Qualification.
3. Prerequisites
Installation Qualification must not begin until prerequisites are met:
- Design Qualification approved
- User Requirements Specification approved
- instrument delivered and inspected for damage
- installation area prepared and verified
- required utilities available and qualified where applicable
- calibration status of critical components defined
- vendor installation documentation available
Incomplete prerequisites lead to invalid qualification results.
4. Installation Verification
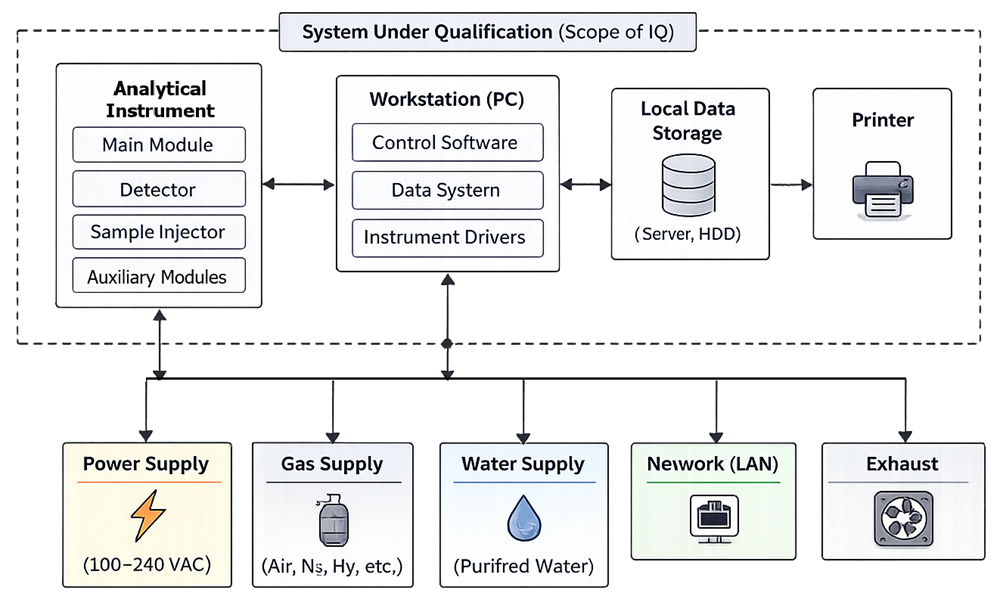
Installation Qualification verifies that the system is installed as intended. The diagram below defines the installation architecture of an analytical instrument system, including hardware modules, computer system, and supporting utilities. It establishes system boundaries and identifies critical interfaces that must be verified during Installation Qualification.
Typical checks include:
4.1 Equipment Verification
- model number, serial number, and configuration match approved specifications
- all modules and accessories are present
- materials of construction consistent with intended use
4.2 Physical Installation
- instrument positioned correctly within designated area
- proper clearances and accessibility maintained
- installation performed according to manufacturer instructions
4.3 Utilities Verification
- electrical supply meets voltage, frequency, and grounding requirements
- gas supplies meet purity and pressure specifications where applicable
- environmental conditions such as temperature and humidity are within defined limits
4.4 Connections and Interfaces
- proper connection of system modules
- network connectivity established if required
- data interfaces configured correctly
4.5 Configuration Verification
Configuration Verification confirms that the installed system is configured in accordance with approved design specifications and intended use. This ensures that the baseline system setup is correct before functional testing begins. Typical checks include:
- installed hardware configuration matches approved configuration or bill of materials
- firmware versions verified and documented where applicable
- instrument configuration parameters set according to manufacturer recommendations and intended use
- module configuration and recognition verified within control software
- system options, licenses, and enabled features match procurement and design specifications
- network configuration parameters defined and documented where required
- default settings reviewed and adjusted as required for controlled operation
5. Software Installation and Configuration
Where computerized systems are used, Installation Qualification must confirm:
- correct software version installed
- system configuration consistent with approved design
- communication between instrument and software verified
- required drivers and interfaces installed
Initial configuration must be documented and controlled.
6. Data Integrity Setup Verification
Installation Qualification must verify that basic data integrity controls are in place:
- user accounts created according to defined roles
- password and access controls configured
- audit trail functionality enabled where applicable
- system time and date correctly configured
Full functional testing of these controls is performed during Operational Qualification.
7. Documentation Verification
Installation Qualification must confirm availability and adequacy of required documentation:
- instrument manuals and technical documentation
- calibration certificates and traceability
- software documentation
- maintenance and service procedures
- vendor-supplied qualification documents if applicable
Documentation must be reviewed for suitability within a GMP environment.
8. Calibration Status
Critical components must be verified as calibrated:
- calibration certificates available and current
- traceability to recognized standards documented
- calibration status labels applied where applicable
If calibration is not acceptable, the system cannot proceed to Operational Qualification.
9. Labeling and Identification
The system must be properly identified:
- equipment ID assigned according to site procedures
- labels applied and legible
- software system identification documented
Clear identification ensures traceability throughout the lifecycle.
10. Deviations and Resolution
All discrepancies identified during Installation Qualification must be formally documented and evaluated within the executed protocol or associated deviation record. Deviation handling during IQ is focused on determining whether the installation state of the system remains acceptable to support subsequent qualification activities. Each deviation must include:
- clear description of the observed condition, including reference to the specific test step or requirement
- classification of the deviation where applicable, based on predefined criteria
- documented impact assessment on installation status, system configuration, and intended use
- determination of whether the deviation affects the ability to proceed with qualification
- defined corrective action or remediation plan
- re-verification of affected installation elements following correction
- final resolution with documented evidence of closure
Deviations must be assessed in the context of Installation Qualification objectives. The key determination is whether the system, as installed, continues to meet design specifications and is suitable to proceed to Operational Qualification.
Minor deviations that do not impact installation integrity or system readiness may be documented and justified without delaying progression. However, any deviation that affects system configuration, required utilities, documentation completeness, or compliance with design specifications must be resolved prior to approval.
Operational Qualification must not begin until all critical and major deviations are closed or formally justified and approved. Open deviations that compromise installation integrity invalidate the qualification baseline and are not acceptable.
11. Acceptance Criteria
Installation Qualification is acceptable when:
- all components are installed as specified
- utilities and environment meet requirements
- documentation is complete and approved
- calibration status is acceptable
- no unresolved critical deviations remain
Acceptance criteria must be predefined in the protocol.
12. Deliverables
Installation Qualification must produce controlled documentation including:
- Installation Qualification protocol and executed test records
- equipment verification records
- utility verification records
- software installation records
- deviation log and resolution
- final summary report
All records must be complete, accurate, and attributable.
13. Approval and Release
Installation Qualification documentation must be reviewed and approved by authorized personnel. Approval confirms that:
- installation is complete and compliant
- system is ready for Operational Qualification
Installation Qualification establishes the verified baseline configuration of the system. Subsequent testing assumes this configuration remains controlled and unchanged.
