Analytical Instrument Performance Qualification
Performance Qualification establishes documented evidence that an analytical instrument performs consistently and reliably under routine operating conditions using approved analytical methods. It confirms that the system, as installed and functionally verified, is suitable for its intended use in a controlled GMP environment.
Performance Qualification demonstrates sustained performance using actual workflows, materials, and personnel representative of routine operation. The diagram below illustrates the sequence of activities performed during Performance Qualification. It shows execution of approved analytical methods under routine conditions, evaluation against predefined acceptance criteria, and controlled decision-making to approve the system for routine use or require re-execution following deviation resolution.
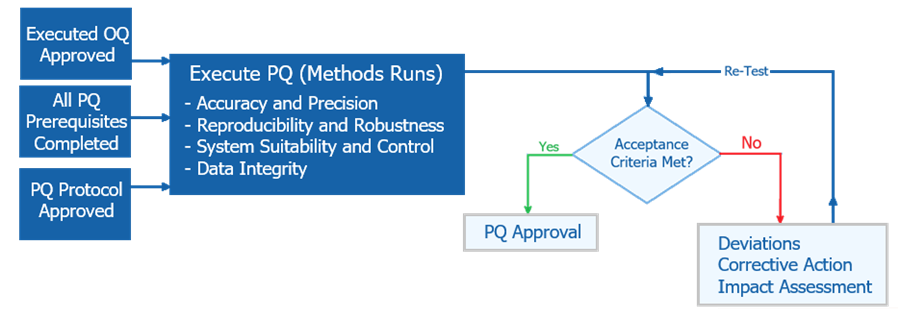
1. Objective
The objective of Performance Qualification is to demonstrate that:
- the instrument performs consistently under routine operating conditions
- approved analytical methods generate reliable and reproducible results
- system performance meets predefined, method-based acceptance criteria
- variability introduced by operators, materials, and conditions is controlled
Completion of Performance Qualification establishes readiness for routine GMP use.
2. Scope
Performance Qualification applies to:
- standard compendial methods or validated analytical methods executed on the instrument
- routine operating conditions and workflows
- trained personnel performing typical operations
- sample types representative of intended use
The scope must align with intended use defined in the User Requirements Specification.
3. Prerequisites
Performance Qualification must not begin until:
- Operational Qualification is executed and approved
- all critical OQ deviations are resolved
- analytical methods are approved for use
- system calibration is current and within validity period
- applicable SOPs are approved and effective
- personnel performing testing are trained and qualified
Failure to meet prerequisites invalidates results.
4. Method-Based Performance Verification
Performance Qualification is executed using approved compendial methods or validated analytical methods representative of routine use. The diagram below illustrates how Performance Qualification is executed using approved compendial or validated analytical methods. It shows the relationship between method steps, system operation, and generation of analytical results under routine conditions.
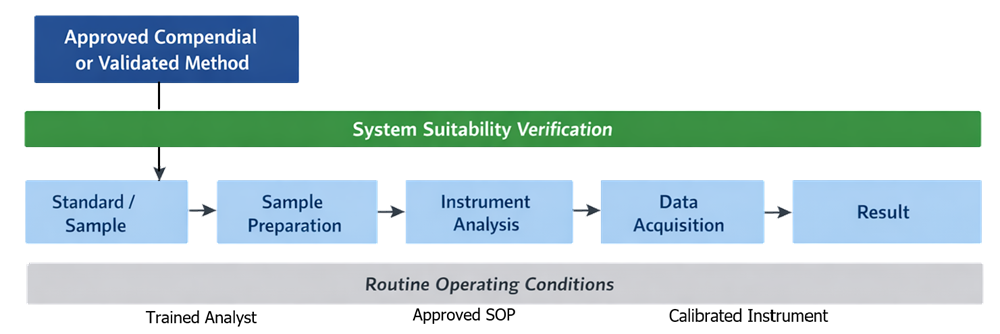
Testing must demonstrate that:
- methods execute correctly under routine operating conditions
- system performance supports method requirements
- results are consistent across repeated analyses
Typical activities include:
- execution of full analytical methods as written
- analysis of standards, controls, and representative samples
- verification of system suitability criteria prior to and during analysis
Testing must reflect actual use conditions, including sample preparation, sequence execution, and data processing.
5. Accuracy and Precision
Performance Qualification must verify that the system produces results that are both accurate and precise. Accuracy is the closeness of measured values to the true or accepted reference value. It is typically evaluated using certified reference standards or known concentrations and expressed as percent recovery or bias. Precision is the degree of agreement among repeated measurements under specified conditions. It includes:
- Repeatability: same analyst, same equipment, short time interval
- Intermediate precision: different analysts, different days, same laboratory
Typical PQ expectations:
- replicate measurements of standards or samples
- evaluation of %RSD or equivalent statistical measure
- predefined limits based on method requirements or compendial standards
6. Reproducibility and Robustness
Performance Qualification must demonstrate that the system maintains acceptable performance under normal sources of variability. Reproducibility refers to consistency of results when analyses are performed across multiple runs and conditions. PQ must define and execute:
- number of runs, typically at least 3 independent runs
- number of replicates per run, typically 3–6 injections or measurements
- inclusion of different analysts where applicable
- testing across different days where feasible
Robustness refers to the ability of the system and method to remain unaffected by small, controlled variations in operating conditions. Where applicable, PQ may include:
- minor variation in environmental conditions within allowable limits
- normal operational variability such as sample preparation differences
Results across all runs and conditions must remain within predefined acceptance criteria. The diagram below illustrates the structure of Performance Qualification testing used to demonstrate reproducibility. It shows multiple runs, replicate measurements within each run, and variation across analysts or days to confirm consistent system performance.
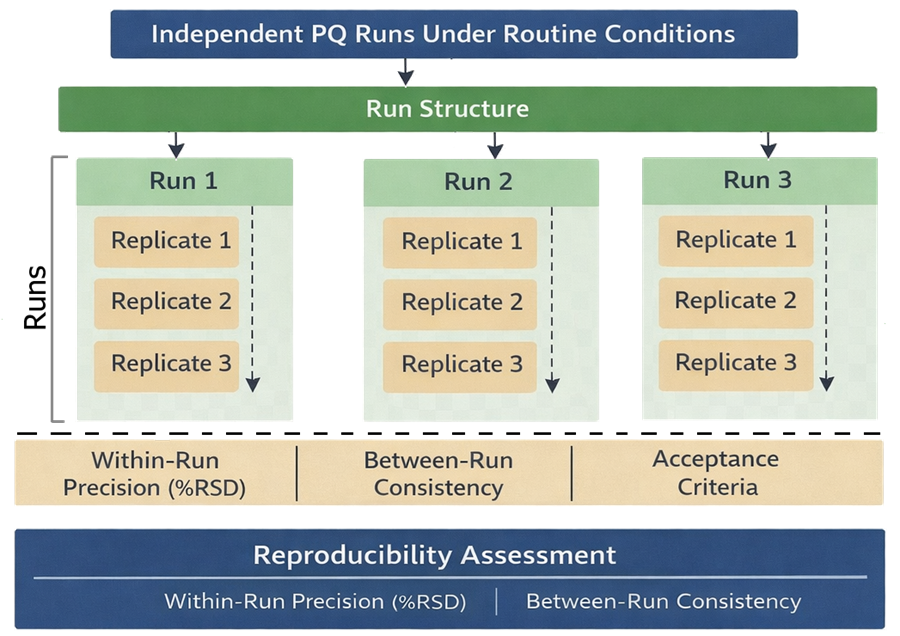
7. System Suitability and Control
System suitability ensures that the instrument is capable of producing valid analytical results prior to and during routine use. System suitability parameters are method-defined and may include:
- detector response and sensitivity
- resolution between peaks
- retention time consistency
- baseline stability
- signal-to-noise ratio
During PQ:
- system suitability must be verified prior to sample analysis
- criteria must be met before data is considered valid
- failure of system suitability invalidates the associated run
System suitability serves as an ongoing control mechanism linking PQ to routine operation.
8. Data Integrity in Routine Use
Performance Qualification must confirm that data integrity controls function during actual operation. This includes:
- correct recording and storage of analytical data
- consistent enforcement of user roles and permissions
- audit trail generation for routine activities
- protection of data from unauthorized modification
Data generated during PQ must be complete, consistent, and attributable.
9. Acceptance Criteria
Acceptance criteria for Performance Qualification must be:
- predefined prior to execution
- based on method requirements, compendial standards, or validated limits
- statistically and scientifically justified
Acceptance criteria must address:
- Accuracy: recovery within defined limits, typically 98–102% unless otherwise justified
- Precision: %RSD within method-defined limits
- Reproducibility:
consistent results across runs, analysts, and days
no significant drift or trend across sequences - System Suitability: all parameters within defined limits prior to and during analysis
- Overall Performance:
all runs meet acceptance criteria
no unexplained variability
no unresolved critical deviations
Failure to meet acceptance criteria requires investigation, corrective action, and re-execution as appropriate.
10. Deviations and Resolution
All deviations identified during Performance Qualification must be documented and evaluated in relation to analytical performance and intended use.
Each deviation must include:
- description of the issue
- impact on analytical results and data reliability
- assessment of potential effect on product quality
- corrective action and retesting where required
- documented resolution
Performance Qualification cannot be approved if deviations compromise data integrity, method validity, or intended use.
11. Deliverables
Performance Qualification must generate controlled documentation including:
- Performance Qualification protocol and executed records
- analytical data and calculations
- system suitability records
- statistical evaluation where applicable
- deviation log and resolution
- summary report
All records must be complete, accurate, and attributable.
12. Approval and Release
Performance Qualification documentation must be reviewed and approved by authorized personnel.
Approval confirms that:
- the instrument performs as intended under routine conditions
- analytical results are accurate, precise, and reproducible
- the system is suitable for its intended GMP use
Performance Qualification establishes the verified performance baseline for routine operation and ongoing monitoring.
