Analytical Instrument Requirements
1. Introduction
Analytical instruments must be defined in terms of their intended use before qualification activities begin. User requirements establish what the instrument is expected to do, how it will be used, and what performance characteristics are necessary to generate reliable analytical data.
For analytical instruments, user requirements must focus on measurement capability, data integrity, and integration with laboratory processes. These requirements form the foundation for qualification and ensure that the selected instrument is suitable for its intended analytical application.
2. Purpose of Analytical Instrument User Requirements
The purpose of user requirements is to define the functional and performance expectations for the analytical instrument in a clear and testable manner.
The diagram below presents the structure of analytical instrument requirements, showing how different requirement categories are defined to ensure that the instrument is suitable for its intended use. These requirement groups collectively define what the instrument must do, how it must perform, and how it must be controlled within a regulated laboratory environment.
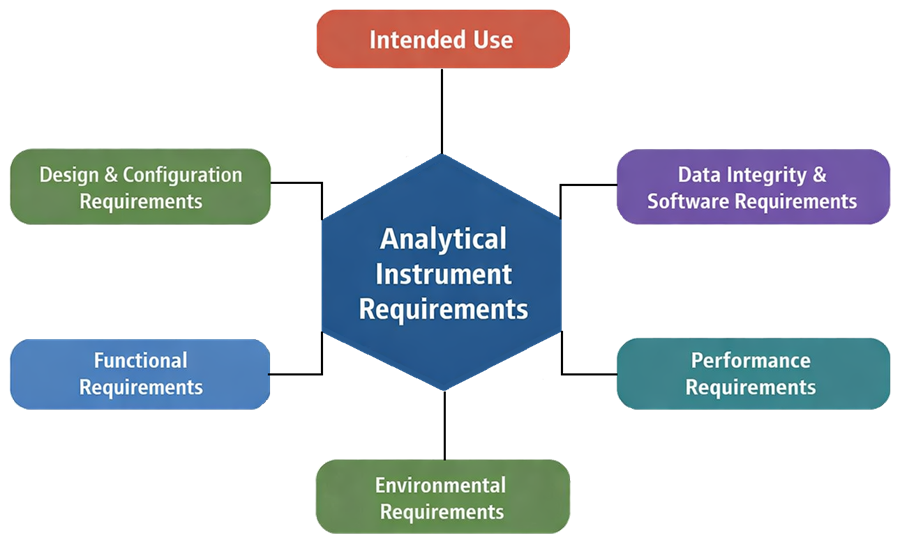
User requirements ensure that:
- the instrument is suitable for the intended analytical method
- critical measurement capabilities are defined
- data integrity expectations are established
- qualification activities are based on documented requirements
- traceability between requirements and testing is maintained
User requirements provide the basis for qualification testing and acceptance decisions.
3. Definition of Intended Use
Intended use is the primary driver for analytical instrument requirements. The intended use must clearly define:
- type of analysis performed
- analytical technique used
- role of the instrument in the laboratory process
- whether data supports GMP decisions such as product release or stability
Examples:
- HPLC system used for assay and impurity testing for product release
- spectrophotometer used for in-process testing
- balance used for preparation of analytical standards
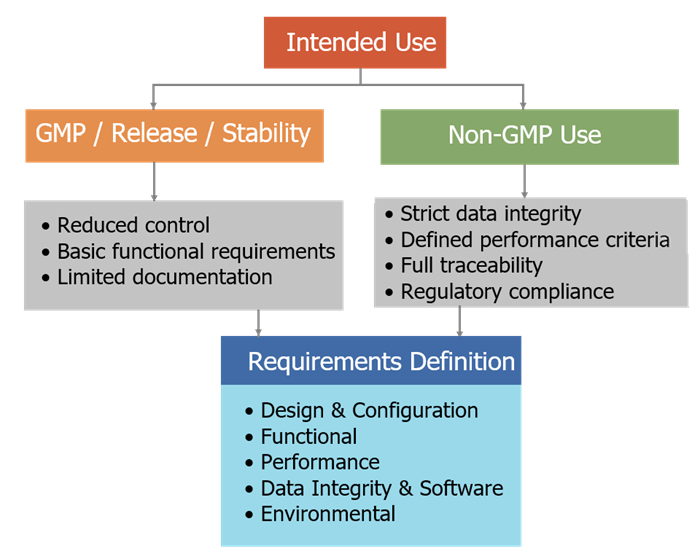
Intended use determines the level of performance, control, and qualification required. The diagram below illustrates how the intended use of an analytical instrument drives the definition of requirements. Instruments used for non-GMP or supporting activities require a reduced level of control and documentation, while instruments used for GMP release, stability, or other critical decisions require stricter performance, data integrity, and compliance requirements. Both pathways lead to a structured set of analytical instrument requirements that define how the system must be designed, configured, and controlled.
4. Design and Configuration Requirements
Design and configuration requirements define how the analytical instrument must be structured and configured to support its intended use. These requirements ensure that the selected system, including hardware and software components, is suitable for the analytical methods it is intended to perform.
The diagram below illustrates the concept of analytical instrument design and configuration, showing how hardware components, software systems, and configuration settings are combined to form a complete analytical system capable of supporting the intended analytical application.
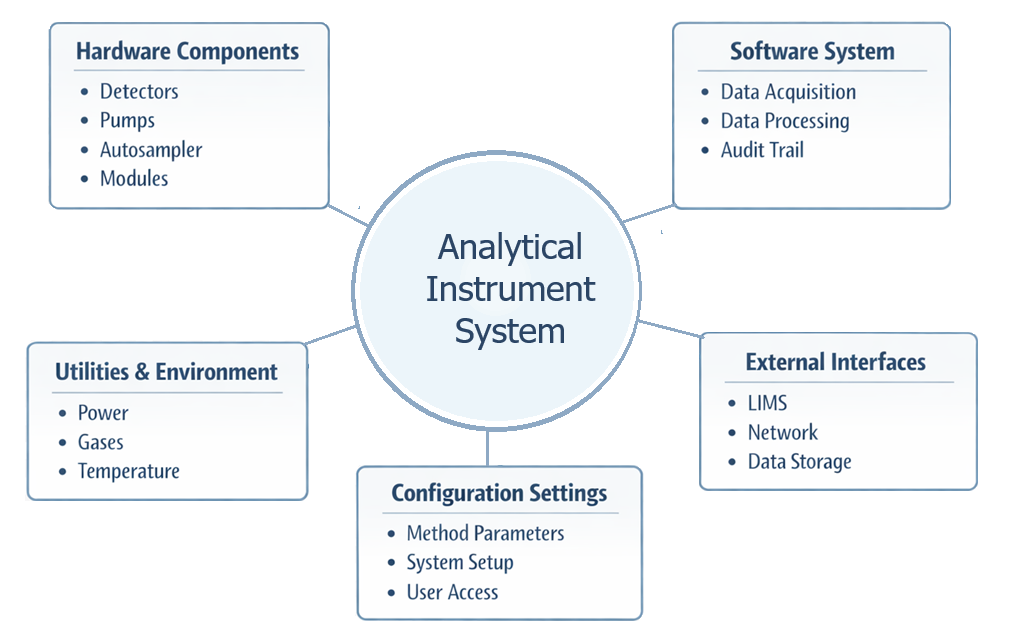
Design requirements typically address:
- selection of appropriate analytical technique and instrument type
- required system components such as detectors, pumps, autosamplers, columns, or modules
- compatibility with intended analytical methods and sample types
- required measurement ranges and operating capabilities
- vendor specifications and performance capabilities
Configuration requirements define how the instrument will be set up within the laboratory environment, including:
- hardware configuration and module integration
- software configuration and system settings
- user access roles and permissions
- data acquisition and processing setup
- interface with external systems such as LIMS or network storage
For computerized analytical instruments, configuration requirements must also address:
- software version and configuration control
- audit trail functionality and data integrity settings
- data storage, backup, and archival configuration
Design and configuration requirements must be clearly defined and documented to ensure that the selected instrument system can support qualification and routine operation without the need for uncontrolled modifications. These requirements provide the basis for subsequent verification during design qualification and ensure alignment between intended use and system capabilities.
5. Functional Requirements
Functional requirements define what the instrument must be capable of doing to support the intended analytical method. Typical functional requirements include:
- measurement capability appropriate to the analytical technique
- required operating ranges for critical parameters
- system control functions and user interface
- sample handling and processing capability
- compatibility with analytical methods
Functional requirements must be specific enough to support verification during qualification.
6. Performance Requirements
Performance requirements define how well the instrument must perform to produce reliable analytical data. These may include:
- accuracy and precision requirements
- repeatability and reproducibility
- detection limits and sensitivity
- linearity across the analytical range
- system suitability criteria
Performance requirements are often aligned with method validation requirements and must support the intended analytical application.
7. Data Integrity and Software Requirements
Analytical instruments frequently include computerized systems for data acquisition and processing. User requirements must address data integrity and regulatory expectations. Typical requirements include:
- user access control and security
- audit trail functionality
- electronic record integrity
- data storage and backup
- compliance with 21 CFR Part 11 where applicable
These requirements must be implemented as an integrated control framework that governs how data is generated, processed, stored, and protected throughout the instrument lifecycle. User access must be restricted through unique accounts with defined roles and permissions to ensure that only authorized personnel can create, modify, or review data. Audit trails must automatically record all relevant actions, including data creation, modification, and deletion, with timestamps and user identification, and must be secure and reviewable.
Electronic records must be protected from unauthorized alteration through system controls such as access restrictions, configuration management, and controlled workflows. Data storage must ensure that original data is preserved, with backup mechanisms in place to prevent loss and to enable recovery in case of system failure. Where applicable, systems must support secure, computer-generated audit trails and electronic signatures in compliance with 21 CFR Part 11.
Data integrity controls must also include procedures for periodic audit trail review, management of user accounts, and control of system configuration changes. These elements collectively ensure that analytical data remains accurate, complete, consistent, and attributable throughout its lifecycle and can be relied upon for regulatory and quality decisions.
8. Integration and Interface Requirements
Analytical instruments often interface with laboratory systems and processes. Requirements may include:
- integration with laboratory information management systems
- compatibility with data acquisition software
- communication with network infrastructure
- data export and reporting capabilities
Integration requirements ensure that the instrument operates effectively within the laboratory environment.
9. Environmental and Installation Requirements
User requirements must define the environmental and installation conditions necessary for proper instrument operation. These may include:
- temperature and humidity ranges
- vibration control
- utility requirements such as power and gases
- space and layout considerations
Proper definition of installation conditions supports successful installation qualification.
10. Maintenance and Support Requirements
User requirements should address ongoing support needed to maintain instrument performance. These may include:
- calibration requirements
- preventive maintenance expectations
- availability of service support
- spare parts and consumables
These requirements ensure that the instrument can be maintained in a qualified state.
11. Documentation and Compliance Requirements
User requirements must define documentation expectations to support qualification and regulatory compliance. These may include:
- requirement for qualification documentation
- traceability between requirements and testing
- availability of vendor documentation
- compliance with applicable regulatory standards
Clear documentation requirements must ensure full traceability between defined requirements, qualification testing, and documented results. Each requirement must be linked to one or more test cases within qualification protocols, and each executed test must generate recorded results that demonstrate whether acceptance criteria have been met. These results are then documented in qualification reports, providing objective evidence that the instrument meets its intended use.
Traceability is typically maintained through structured documentation or a traceability matrix that links requirements to test scripts, test results, and final reports. This linkage ensures that all requirements are verified, no critical functions are omitted from testing, and all qualification activities are supported by complete and auditable records.
This approach ensures consistency, supports deviation assessment, and provides a clear audit trail demonstrating that the analytical instrument has been qualified in accordance with defined requirements and regulatory expectations.
12. Summary
Analytical instrument user requirements define the design and configuration, functional, performance, and compliance expectations necessary to ensure that an instrument is fit for its intended use. By clearly defining intended use, measurement capability, data integrity, and integration requirements, user requirements provide the foundation for qualification and lifecycle control of analytical instruments in regulated laboratory environments.
