Airflow Patterns and Pressure Cascades
Purpose and Regulatory Context
Airflow patterns and pressure cascades are foundational controls in GMP facilities. Their role is straightforward: move air in a predictable manner that protects product, prevents cross-contamination, and sustains a demonstrable state of control. Regulators expect these controls to be intentional, documented, qualified, and monitored. There is no appetite for improvisation here. This is settled practice.
From a compliance standpoint, expectations align with long-standing requirements under U.S. Food and Drug Administration cGMP regulations, EU GMP, and guidance such as ISO cleanroom standards. Airflow direction and pressure differentials are not aesthetic design features. They are contamination control measures.
Airflow Patterns
Airflow patterns define how supply air is introduced, distributed, and removed from a controlled space.
Common patterns include:
- Unidirectional airflow: Typically vertical, from ceiling-mounted HEPA filters downward to low-level returns. This approach is standard for higher-risk operations where particulate control is critical.
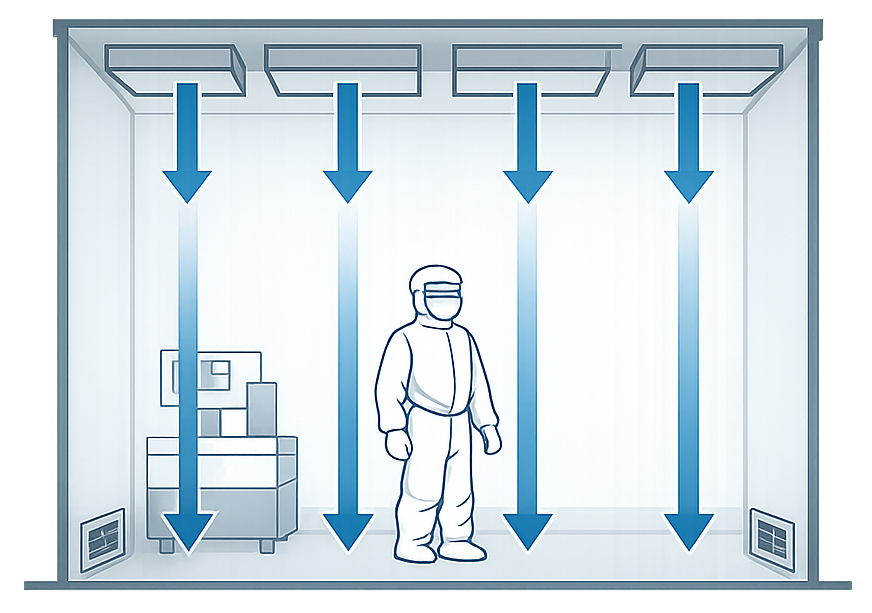
- Non-unidirectional (turbulent or mixed) airflow: Used in lower-classification rooms where dilution and removal, rather than sweeping, is sufficient.
In GMP environments, airflow is designed to move from clean to less clean areas. That principle has not changed in decades and for good reason. Any design that violates this logic invites regulatory scrutiny.
Pressure Cascades
Pressure cascades are the mechanism that enforces airflow direction across rooms and corridors.
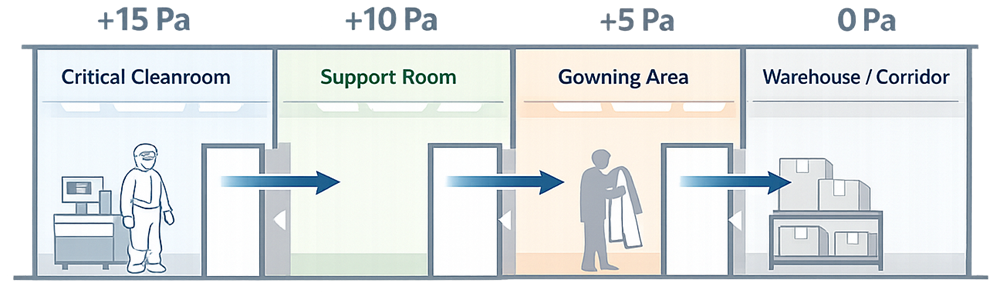
A pressure cascade is established by maintaining progressively higher pressures in cleaner areas relative to adjacent, less controlled spaces. When doors open or minor leaks occur, air moves outward from the clean space, not inward. Typical cascade logic:
- Critical processing rooms at the highest pressure
- Support rooms at intermediate pressure
- Corridors, warehouses, or unclassified areas at the lowest pressure
This hierarchy is deliberate. It is one of the simplest and most effective contamination control strategies ever adopted in pharmaceutical facility design. Pressure differentials are established to drive airflow from cleaner, higher-pressure areas toward less controlled, lower-pressure spaces, maintaining contamination control.
Relationship Between Airflow and Pressure
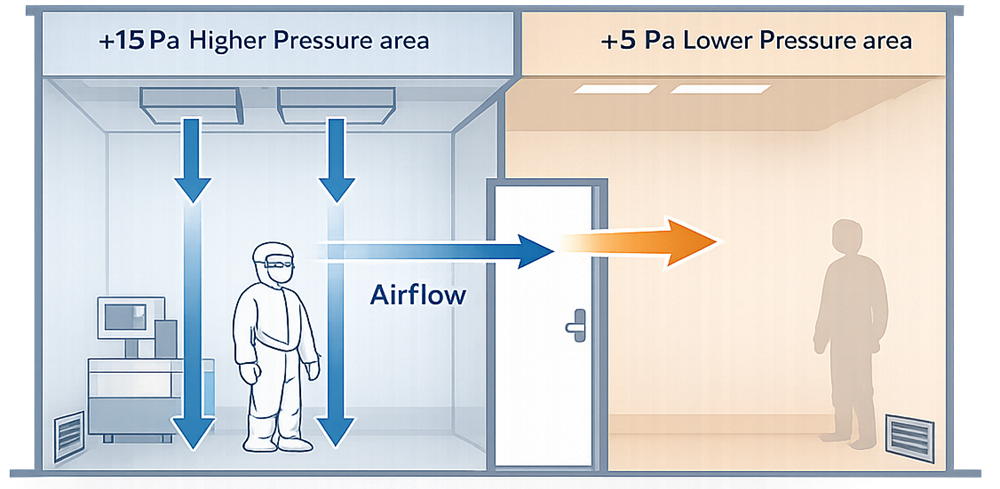
Airflow patterns and pressure cascades are inseparable. Pressure differentials without defined airflow paths are meaningless. Conversely, airflow patterns without maintained pressure relationships are unstable. In practice:
- Pressure differentials establish directionality between rooms
- Airflow distribution ensures effective particle removal within rooms
Airflow patterns and pressure differentials function as an integrated control strategy and must be designed together, verified together, and maintained together to ensure consistent contamination control.
Qualification and Verification
From a lifecycle perspective, airflow and pressure controls are addressed across qualification stages:
- Design Qualification
Confirmation that airflow concepts and pressure hierarchy align with intended use and regulatory expectations. - Installation and Operational Qualification
Verification that air supply, returns, controls, alarms, and pressure setpoints function as designed. - Performance Verification and Monitoring
Demonstration that airflow direction and pressure differentials are sustained during normal operations, supported by routine monitoring and alarm review.
Smoke visualization, differential pressure measurements, and trend review are established tools. They are expected. Reinventing this wheel rarely ends well.
Operational Reality
Pressure cascades drift when facilities are poorly maintained, doors are misused, or change control is weak. Experienced inspectors recognize this immediately. Facilities that treat airflow and pressure as living systems, not static drawings, consistently perform better during inspections.
The practical takeaway is simple:
Design it conservatively, qualify it rigorously, and monitor it continuously. This is not an area where shortcuts pay off.
