Environmental Monitoring for Aseptic Filling
Environmental monitoring during aseptic filling is a contamination control verification program. It provides documented evidence that ISO 5 exposure zones and surrounding classified areas remain in a state of microbiological control during dynamic production conditions.
This article addresses monitoring during active aseptic filling operations. Cleanroom classification theory and barrier system design are addressed separately.
1. Regulatory Framework
Environmental monitoring during aseptic processing is required under:
• 21 CFR 211.42 – Design and environmental control
• 21 CFR 211.113 – Control of microbiological contamination
• EU GMP Annex 1 – Manufacture of Sterile Medicinal Products
• ISO 14644 – Cleanroom classification (non-viable particles)
• PDA Technical Reports addressing sterile manufacturing
21 CFR does not prescribe exact sampling frequency or locations. It requires control and prevention of contamination. Regulatory authorities interpret this to require viable monitoring in ISO 5 during active aseptic operations.
EU GMP Annex 1 explicitly requires viable monitoring in Grade A / ISO 5 zones during operation.
Recent FDA observations have cited firms for:
• Failure to perform viable air sampling during filling
• Inadequate glove fingertip monitoring
• Insufficient monitoring during interventions
• Overreliance on non-viable particle data
Non-viable monitoring does not demonstrate microbiological control. Viable monitoring is required.
2. Purpose of Environmental Monitoring During Filling
Aseptic filling has no terminal sterilization step. Sterility assurance depends entirely on:
• Environmental control
• Equipment design
• Operator technique
• Procedural discipline
Environmental monitoring verifies that contamination control is functioning during actual product exposure. It serves four functions:
• Detect loss of control
• Support batch release decisions
• Support media fill interpretation
• Provide trend data for lifecycle control
It is a verification tool, not a substitute for process validation.
3. Risk-Based Monitoring Design
Monitoring locations and frequency must be justified by documented risk assessment. Selection should consider:
• Open container exposure points
• Filling needle location
• Stopper placement zone
• Operator glove proximity
• Intervention frequency
• Airflow patterns
Sampling must focus on critical exposure zones, not convenient locations.
Historical trend data must be used to refine sampling strategy over time.
The diagram below illustrates a representative ISO 5 monitoring layout for an aseptic filling line operating under dynamic conditions. Sampling locations are positioned at critical exposure points, including the filling needle zone, open container pathway, stopper handling area, and operator interface locations. Placement must be justified through documented risk assessment and supported by airflow studies and historical trend data.
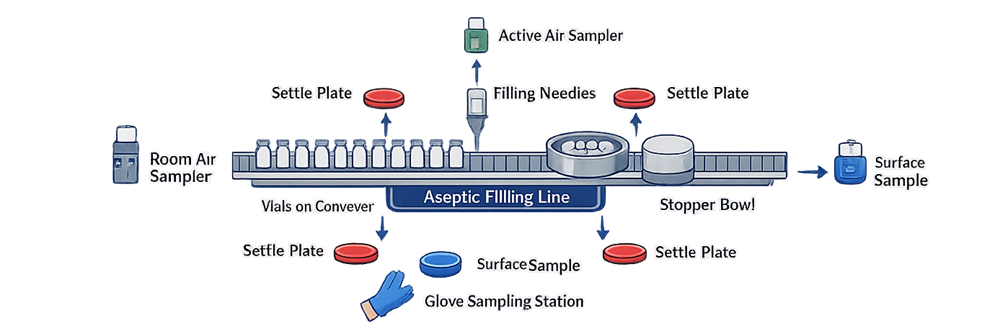
4. Viable Monitoring Requirements
Viable monitoring during aseptic filling must include dynamic sampling in ISO 5 areas. Typical methods include:
Active Air Sampling
• Quantitative collection of defined air volume
• Performed in critical exposure zone
• Conducted during active filling
Settle Plates
• Passive monitoring of microbial fallout
• Placed adjacent to exposure zones
• Limited sensitivity compared to active air
Surface Monitoring
• Contact plates for equipment surfaces
• Focus on high-touch areas
Glove Fingertip Sampling
• Performed at end of operations
• May also be performed after major interventions
• Direct indicator of operator aseptic technique
Monitoring must occur during operation, not only before or after filling.
5. Non-Viable Particle Monitoring
Non-viable particle monitoring verifies airflow integrity and particulate control. It does not detect microorganisms. Continuous particle monitoring is typically required in ISO 5 zones during filling. Particle excursions may indicate:
• Airflow disruption
• Barrier breach
• Mechanical particle generation
Particle compliance does not confirm microbiological control.
6. Alert and Action Levels
Alert and action limits must be predefined in the environmental monitoring program and approved prior to use. Limits must be aligned with regulatory expectations for ISO 5 environments and supported by historical performance data.
Alert levels represent early warning thresholds. They indicate potential process drift but do not automatically represent loss of control. Action levels represent defined loss-of-control thresholds requiring formal investigation and documented response.
Escalation must be structured and consistent. Repeated alert-level events may indicate instability and require formal investigation even if action limits are not exceeded.
The following matrix defines the required response framework.
| Level | Definition | Typical Trigger | Required Response | Product Impact Assessment | Documentation Requirement |
|---|---|---|---|---|---|
| Normal | Within established environmental limits | No excursions | Routine review and trending | Not required | Batch-level EM review |
| Alert | Early warning threshold exceeded | Single low-level viable count or particle trend | Documented review and trend evaluation | Not typically required | Record in EM review |
| Repeated Alert | Multiple alerts in same location or trend toward instability | Recurring low-level viable counts | Formal investigation | Risk-based evaluation | Investigation record |
| Action | Action limit exceeded | Viable count above ISO 5 limit | Immediate investigation | Required | Deviation report |
| Critical Action | Significant or repeated action-level events | Multiple action excursions or ISO 5 failure | Escalated investigation and management review | Mandatory | Deviation + CAPA |
Alert and action limits must not be treated as statistical artifacts. They are control boundaries. Exceedance of action levels requires documented root cause analysis and determination of product impact. Failure to consistently escalate repeated alerts undermines the integrity of the monitoring program.
7. Monitoring During Interventions
Interventions represent the highest contamination risk. Monitoring strategy must ensure:
• Viable sampling during intervention-heavy periods
• Consideration of intervention timing relative to excursions
• Correlation of glove data to intervention type
Failure to monitor during interventions undermines contamination control verification.
8. Environmental Monitoring for Manual Aseptic Filling
Manual filling operations conducted in an ISO 5 laminar airflow hood or biosafety cabinet require the same level of viable monitoring discipline as automated filling lines. In manual operations, contamination risk is often higher because:
• Operator hands are continuously within the critical zone
• Interventions are constant rather than discrete
• Product exposure time may be longer
• Airflow disruption risk is greater
Monitoring in manual filling should include:
• Active air sampling positioned adjacent to the critical work zone
• Settle plates placed near open containers
• Surface sampling of work surfaces and equipment
• Mandatory glove fingertip sampling at end of operation
• Monitoring during extended operations
The diagram below illustrates a representative ISO 5 monitoring layout for manual aseptic filling performed in a unidirectional airflow hood. Sampling locations are positioned within the critical work zone at points of direct product exposure and operator interaction.
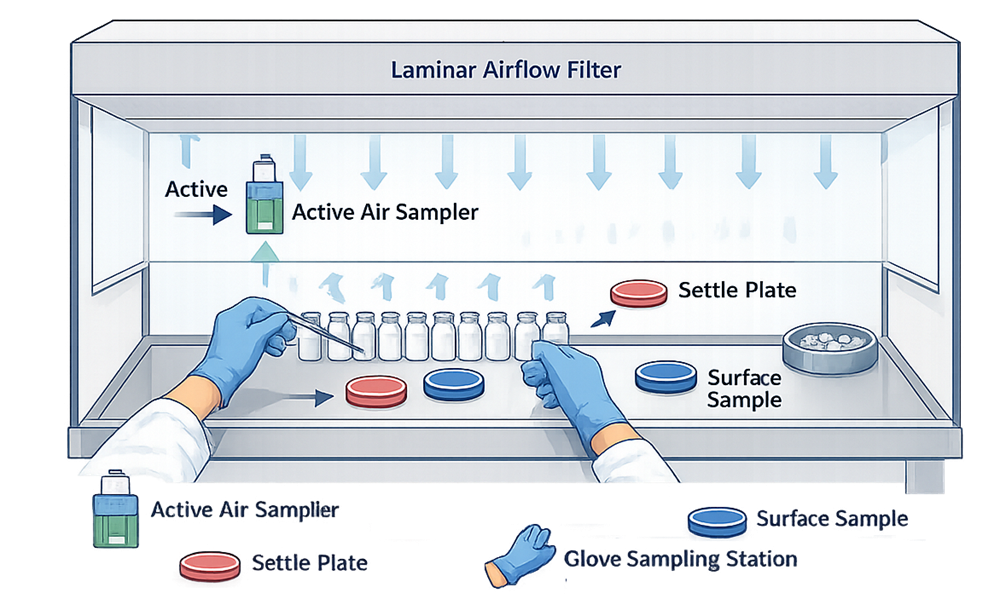
Sampling locations must be placed as close as technically feasible to the point of product exposure without disrupting airflow.
9. Data Review and Trending
Environmental monitoring data must be:
• Reviewed per batch
• Reviewed periodically for trends
• Evaluated by trained personnel
Trend analysis should identify:
• Operator-specific contamination patterns
• Location-specific recurring excursions
• Seasonal or facility-related variability
Trend review is part of continued process verification.
10. Investigation of Excursions
Any action-level excursion requires documented investigation. Investigation must evaluate:
• Timing relative to filling operations
• Intervention activity
• Environmental trends
• Equipment performance
• Barrier integrity
• Organism identification
Microbial identification supports root cause analysis and comparison to environmental flora.
Failure to identify root cause does not justify dismissal.
11. Integration with Media Fill and Lifecycle Control
Environmental monitoring supports interpretation of media fill results. If media fill contamination occurs, EM data provides context for:
• Intervention-related contamination
• Environmental instability
• Operator-related patterns
Monitoring strategy must be periodically reassessed based on:
• Trend data
• Facility modification
• Equipment change
• Process expansion
Environmental monitoring is not static. It evolves with process knowledge.
Conclusion
Environmental monitoring during aseptic filling is a regulatory expectation and a critical contamination control verification tool. Viable monitoring in ISO 5 during dynamic operation is required and cannot be replaced by particle monitoring alone.
A risk-based, well-documented, and consistently executed monitoring program provides defensible evidence that aseptic filling operations remain in a state of microbiological control.
