Barrier System Decontamination and Control Strategy
1. Purpose and Scope
This article defines the validation and control framework governing decontamination of pharmaceutical barrier systems used in aseptic processing. It describes how enclosure decontamination cycles are developed, qualified, controlled, and maintained within a lifecycle model to ensure sustained sterility assurance under routine manufacturing conditions.
The scope includes vaporized hydrogen peroxide processes, biological indicator strategy, critical parameter control, residual management, integration into contamination control strategy, qualification lifecycle, change control, and continued verification.
2. Decontamination Process Principle
Barrier system decontamination is a defined, repeatable process executed within a sealed enclosure. In pharmaceutical applications, vaporized hydrogen peroxide is the predominant modality. The process typically includes:
• Conditioning phase to achieve target humidity
• Controlled injection and vapor distribution
• Defined exposure period
• Aeration phase to reduce residual peroxide
Process effectiveness depends on:
• Uniform vapor distribution throughout the enclosure
• Controlled temperature and relative humidity
• Defined concentration profile
• Sufficient exposure duration
• Effective residual removal prior to access
Critical parameters must be scientifically established during development studies. Acceptable operating ranges must be defined before performance qualification and incorporated into routine cycle control. Material compatibility must be evaluated during design qualification. Gloves, seals, elastomers, instrumentation components, and cable penetrations must withstand repeated peroxide exposure without compromising enclosure integrity.
3. Integration into Contamination Control Strategy
Decontamination is one component of a broader contamination control strategy. Its effectiveness depends on coordinated control of:
• Glove integrity
• HEPA filter integrity
• Pressure control and cascade stability
• Environmental monitoring
• Transfer system performance
• Alarm functionality
• Preventive maintenance
Intervention management is critical. Each defined intervention must be evaluated to determine whether full decontamination is required or whether predefined recovery procedures are sufficient. Decision logic must be risk-based and formally documented.
4. Qualification Lifecycle
This lifecycle addresses qualification of the decontamination process and its control strategy, not the mechanical qualification of the isolator or RABS enclosure itself. The objective is to demonstrate that the enclosure can be reproducibly returned to a validated microbiological state through a defined, controlled decontamination cycle and that this state is maintained over time.
Design Qualification of the Decontamination Process
Design Qualification focuses on the scientific and technical suitability of the decontamination concept. This includes evaluation of:
• Selection of vaporized hydrogen peroxide as the decontamination modality
• Definition of critical process parameters and their theoretical lethality relationship
• Vapor injection and distribution concept
• Aeration capability and exhaust efficiency
• Residual monitoring strategy
• Control system logic, parameter recording, and alarm architecture
• Data integrity controls
Cycle development studies must define:
• Target concentration profile
• Required exposure time
• Humidity conditioning requirements
• Temperature influence
• Minimum effective lethality conditions
Worst-case assumptions must be identified during this phase. These include enclosure geometry, shadowing, airflow influence, and representative internal configurations. The outcome of DQ is a defined, scientifically justified cycle concept ready for operational challenge.
Installation Qualification of Decontamination Controls
Installation Qualification verifies that all components required to execute and monitor the decontamination cycle are correctly installed and documented.
This includes:
• Vapor generator installation verification
• Injection and distribution hardware verification
• Aeration components and exhaust routing
• Installation and calibration status of peroxide, temperature, humidity, and pressure sensors
• Verification of control system configuration
• Confirmation of data recording and secure storage
IQ for decontamination ensures that the monitoring and control infrastructure necessary to maintain validated parameters is reliable and traceable.
Operational Qualification of the Decontamination Cycle
Operational Qualification establishes and challenges the validated operating envelope of the decontamination cycle. The objective of OQ is to define the minimum effective conditions that consistently achieve required microbial lethality while maintaining safe and controlled residual levels.
Key OQ activities include:
• Confirmation of parameter ranges for hydrogen peroxide concentration, exposure time, relative humidity, and temperature
• Vapor distribution and concentration mapping across the enclosure
• Identification and confirmation of worst-case biological indicator locations
• Determination of minimum effective cycle parameters based on biological challenge data
• Use of chemical indicators to support vapor exposure assessment and distribution confirmation
• Alarm setpoint verification and interlock testing
• Validation of aeration performance and residual peroxide reduction capability
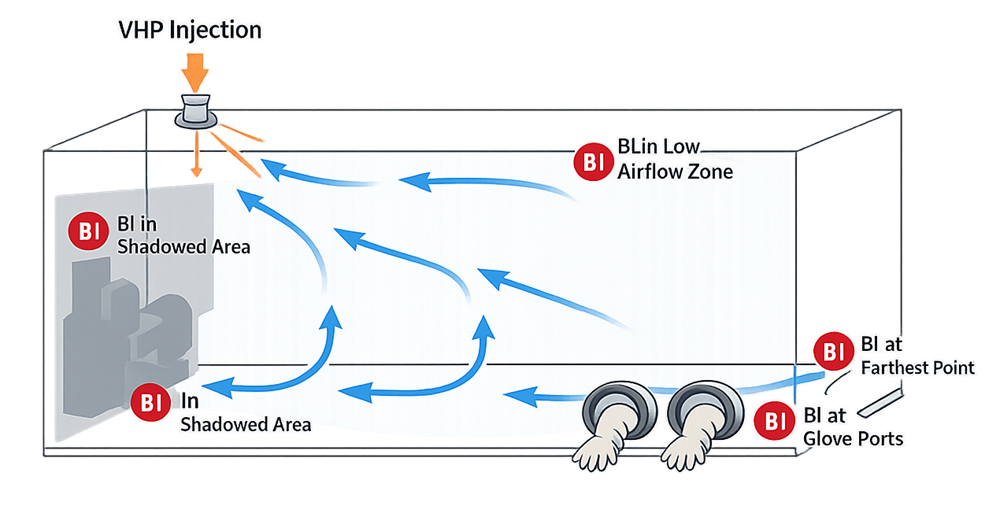
Biological indicators are a central component of OQ. For hydrogen peroxide processes, Geobacillus stearothermophilus spores are typically used due to their established resistance profile. Indicators must be placed at locations representing worst-case conditions, including:
• Regions of lowest airflow velocity
• Shadowed or obstructed surfaces
• Downstream of structural elements
• Farthest points from vapor injection
• Glove ports and transfer interfaces
Placement rationale must be supported by documented airflow evaluation and risk assessment. Biological challenge studies during OQ establish the relationship between process parameters and lethality and define the minimum validated cycle conditions rather than relying solely on nominal programmed setpoints.
The following schematic illustrates representative worst-case biological indicator placement within a barrier enclosure based on vapor distribution and airflow dynamics.
Chemical indicators may be used during OQ to demonstrate vapor exposure and to support distribution studies. They provide qualitative confirmation of peroxide contact but do not replace biological lethality confirmation.
Residual validation is also performed during OQ. Aeration duration and exhaust performance must be demonstrated to consistently reduce hydrogen peroxide concentration below predefined safety thresholds prior to personnel access or product exposure. Residual monitoring instrumentation must be calibrated and verified to ensure accuracy. The curve below illustrates a representative hydrogen peroxide concentration profile across conditioning, injection and exposure, and aeration phases, demonstrating reduction below the validated residual acceptance threshold.
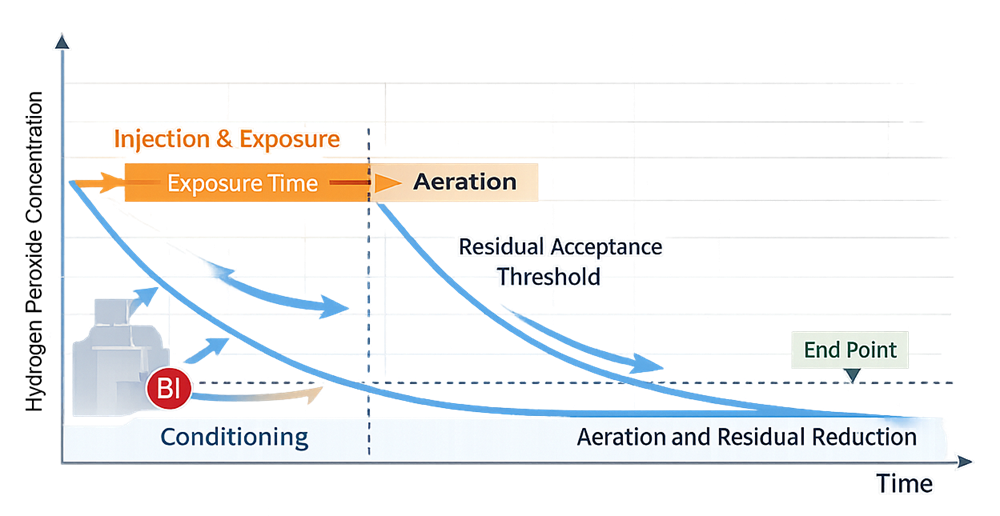
OQ therefore defines the validated operating envelope for lethality, distribution, alarm control, and safe residual reduction.
Performance Qualification of Decontamination
Performance Qualification confirms reproducibility of the finalized decontamination cycle under defined routine operational configuration. The objective of PQ is not to re-establish parameter limits, but to demonstrate that the cycle, as defined during OQ, consistently achieves validated lethality and residual safety outcomes in normal use.
PQ typically includes:
• Execution of consecutive full decontamination cycles under routine configuration
• Placement of biological indicators at previously established worst-case locations
• Verification that all critical parameters remain within validated operating ranges
• Confirmation that residual hydrogen peroxide levels consistently meet predefined acceptance criteria at cycle completion
• Documentation of stable control system performance and absence of unexplained alarms
Biological indicators during PQ serve to confirm reproducibility of lethality at the defined minimum validated cycle conditions. Indicator placement should remain consistent with worst-case locations established during OQ unless configuration changes justify additional evaluation.
Chemical indicators may be used as supplemental exposure confirmation where defined by protocol but are not substitutes for biological confirmation.
Residual verification during PQ confirms that aeration performance consistently achieves safe endpoint criteria under routine conditions. Monitoring data must demonstrate that peroxide concentrations are reduced below validated thresholds before release of the enclosure for operation.
PQ therefore demonstrates that the validated decontamination cycle performs reliably, not only under development challenge conditions but during defined routine operational execution.
Routine Control and Monitoring
Once qualified, the decontamination process becomes a controlled routine operation. Routine control includes:
• Review of critical parameter traces for every cycle
• Verification that concentration, exposure time, humidity, and temperature remain within validated ranges
• Confirmation of aeration endpoint criteria
• Alarm review and documentation
• Periodic biological verification as defined by site risk assessment
Parameter trending must be performed to detect gradual drift, sensor degradation, or control instability.
Periodic Requalification
Periodic requalification confirms that the decontamination process continues to meet validated lethality expectations. Triggers for requalification may include:
• Vapor generator maintenance or replacement
• Sensor replacement or recalibration impact
• Control system modification
• Changes in injection configuration
• Repeated deviation trends
• Significant enclosure modifications affecting vapor dynamics
Requalification scope must be risk-based and may include:
• Targeted parameter verification
• Repeat vapor distribution mapping
• Repeat biological indicator studies
• Partial or full repetition of PQ
Lifecycle control requires documented periodic review of cycle data, alarm history, residual performance, and deviation trends. Continued validated state must be demonstrated through objective evidence, not historical assumption.
5. Change Control and Data Integrity
All decontamination cycles must be recorded and retained in accordance with data integrity principles. Parameter records must be attributable, legible, contemporaneous, original, and accurate.
Changes affecting enclosure geometry, airflow distribution, vapor injection configuration, control software, load configuration, or material surfaces require formal change control and documented impact assessment. The validated state must be preserved through structured evaluation and, where necessary, requalification.
6. Conclusion
Barrier system decontamination is a lifecycle-controlled, validated process embedded within the overall contamination control strategy. Sustained sterility assurance depends on scientifically justified parameters, documented biological confirmation, integrated system control, disciplined change management, and structured requalification.
