Biological Indicators
Biological indicators are standardized microbial preparations used to challenge and verify the lethality of sterilization processes. They contain a defined population of highly resistant microorganisms and provide a direct measure of sterilization effectiveness under worst-case conditions.
Biological indicators are not used to establish sterility of finished product. They are used to validate and monitor the performance of sterilization processes.
Definition
A biological indicator is a test system containing viable microorganisms of known resistance to a specific sterilization modality. The microorganisms are selected based on their relative resistance to the sterilization method being validated. Typical characteristics include:
- Defined population (commonly 10⁶ spores)
- Known D-value under specified conditions
- Certified resistance profile
- Standardized carrier format
Biological indicators are supplied in multiple physical formats depending on sterilization modality and validation strategy.

Spore Strip Biological Indicator
Paper carrier inoculated with approximately 10⁶ resistant spores, used for placement in challenging load locations.

Self-Contained Biological Indicator
Integrated system containing spore carrier and growth medium for post-cycle incubation and interpretation.
Microorganisms Used
Different sterilization methods require different biological indicator organisms:
- Moist Heat: Geobacillus stearothermophilus
- Ethylene Oxide: Bacillus atrophaeus
- Dry Heat: Often Bacillus atrophaeus or other validated resistant strains
- Hydrogen Peroxide: Geobacillus stearothermophilus commonly used
- Radiation: Dose-based validation typically uses bioburden methods rather than classical BI strips
The selected organism must represent a conservative challenge relative to expected product bioburden.
Population and Resistance
Most biological indicators contain approximately 10⁶ spores. This defined population allows validation using the overkill concept.
Resistance is characterized by the D-value: D-value = time required at a specific condition to achieve a 1-log reduction in the microbial population. Microbial resistance to sterilization conditions is commonly represented using a survivor curve, illustrating log-linear reduction over time.
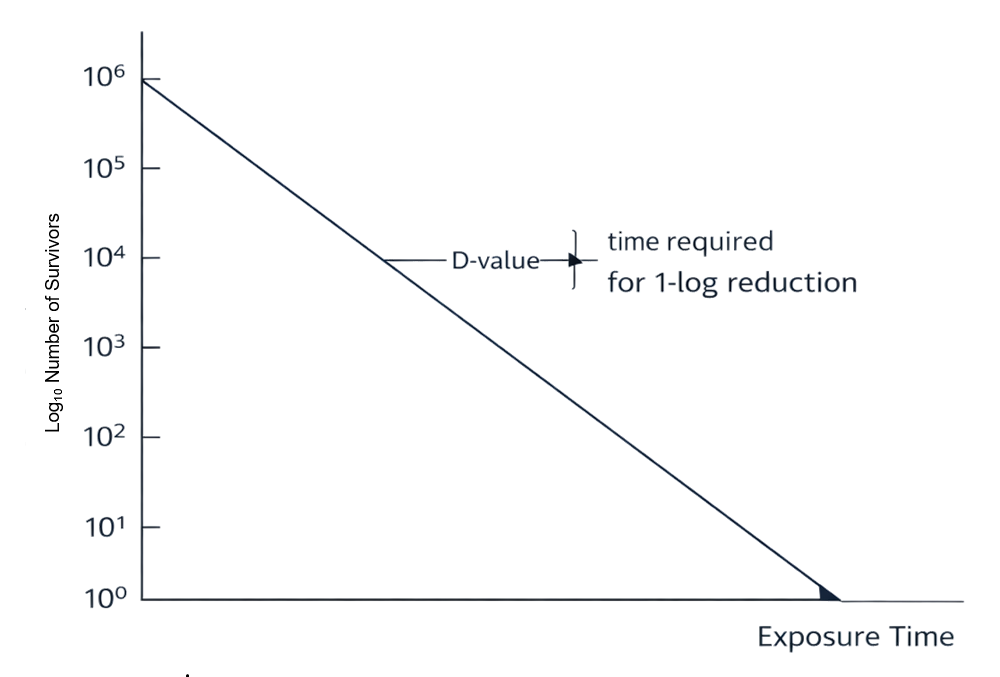
Higher D-values indicate greater resistance.
Validation calculations rely on demonstrated reduction of the biological indicator population to support achievement of the required Sterility Assurance Level.
Overkill Validation Concept
In the overkill approach, the sterilization process is validated to demonstrate at least a 12-log reduction capability against a 10⁶ spore population. This provides:
- 6 logs to inactivate the biological indicator
- 6 additional logs as safety margin
- Theoretical survivor probability of 10⁻⁶
This conservative approach is widely used for moist heat sterilization.
The overkill approach demonstrates sterilization lethality beyond the biological indicator population to provide a defined safety margin relative to expected product bioburden.

Placement and Worst-Case Challenge
Biological indicators must be placed in locations representing worst-case sterilization conditions, including:
- Cold spots
- Densely packed areas
- Most difficult-to-penetrate regions
- Maximum load configuration
Incorrect placement invalidates the challenge.
Incubation and Interpretation
Following sterilization exposure:
- Biological indicators are incubated under defined conditions
- Growth indicates process failure
- No growth supports lethality demonstration
Interpretation must follow manufacturer instructions and validation protocol requirements.
Limitations
Biological indicators:
- Do not confirm sterility of finished product
- Do not replace routine process monitoring
- Must be supported by physical parameter control
- Are not a substitute for validated cycle development
Sterilization validation requires integration of physical data, lethality calculations, and biological challenge studies.
Qualification and Control of Biological Indicators
Biological indicators themselves are controlled materials and must be qualified prior to use in sterilization validation.
Qualification of biological indicators includes verification of:
- Certified population (typically ~10⁶ spores)
- Certified D-value under defined reference conditions
- Expiration date and storage requirements
- Lot traceability and supplier certification
Manufacturers provide a certificate of analysis documenting population and resistance characteristics. These values must be reviewed to confirm suitability for the intended sterilization modality.
Population Verification
The stated spore population must fall within the manufacturer’s specified range. Excessive variability in population can affect lethality interpretation.
Routine re-enumeration of each lot is not typically required in pharmaceutical practice when qualified commercial suppliers are used. However, procedures must define:
- Acceptance of new BI lots
- Documentation of certificate review
- Controls for storage and handling
D-Value Considerations
The D-value defines the resistance of the biological indicator organism at a specific exposure condition. Validation calculations depend on this resistance characteristic.
When using the overkill method, the sterilization cycle must demonstrate sufficient lethality relative to the certified D-value to achieve the required log reduction.
The D-value provided by the manufacturer is determined under defined laboratory conditions and may not exactly represent resistance within product loads. Therefore, placement studies and fractional cycle development remain critical.
Ongoing Control
Biological indicator control is part of lifecycle validation and includes:
- Lot tracking
- Controlled storage
- Expiry management
- Review of unexpected positive results
- Investigation of resistance variability
Biological indicators are a validated challenge system. Their control must be documented within the sterilization validation program.
Lifecycle Control
Ongoing use of biological indicators may include:
- Routine cycle monitoring
- Requalification studies
- Change control evaluation
- Trending of BI performance
Periodic review of biological indicator lots, resistance certificates, and storage conditions is required to maintain confidence in the validation system.
