Bioreactor Sensor Integration
1. Introduction
Sensor integration within a bioreactor system is the structured incorporation of measurement devices, transmitters, and control interfaces that enable continuous monitoring and regulation of critical process parameters. In regulated pharmaceutical environments, sensors are not auxiliary components. They are direct-impact elements that define process control capability, batch reproducibility, and ultimately product quality.
The objective of sensor integration is to ensure that measured values are accurate, reliable, stable, and traceable throughout the operating lifecycle of the system. Measurement failure is process failure. Inadequate sensor selection, poor installation, or uncontrolled drift compromises the validated state even when mechanical systems remain intact.
This article focuses on engineering integration, measurement reliability, calibration control, and high-level data integrity expectations. Detailed computerized system validation and Part 11 compliance strategy are addressed separately.
2. Role of Sensors in Process Control
Bioreactor performance depends on maintaining stable environmental conditions. Sensors provide the real-time data required to regulate:
- pH
- Dissolved oxygen
- Temperature
- Pressure
- Foam formation
- Liquid level
- Gas flow
- Feed rate
The diagram below illustrates the complete measurement architecture from process probe to control interface.
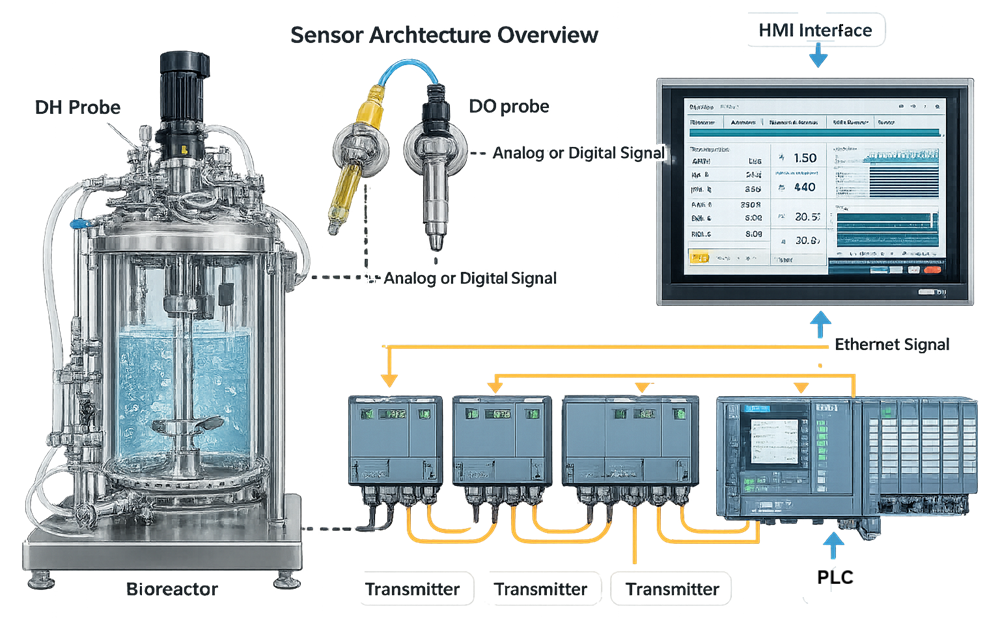
Control loops operate only as effectively as the measurement input allows. Sensor accuracy, response time, and signal stability determine whether control logic functions as intended. Sensors supporting critical process parameters are considered direct-impact instrumentation. Their performance must be defined, controlled, and periodically verified.
3. Sensor Selection and Technology Considerations
Sensor selection must align with process requirements, sterilization conditions, and operating range. Examples include:
pH Measurement:
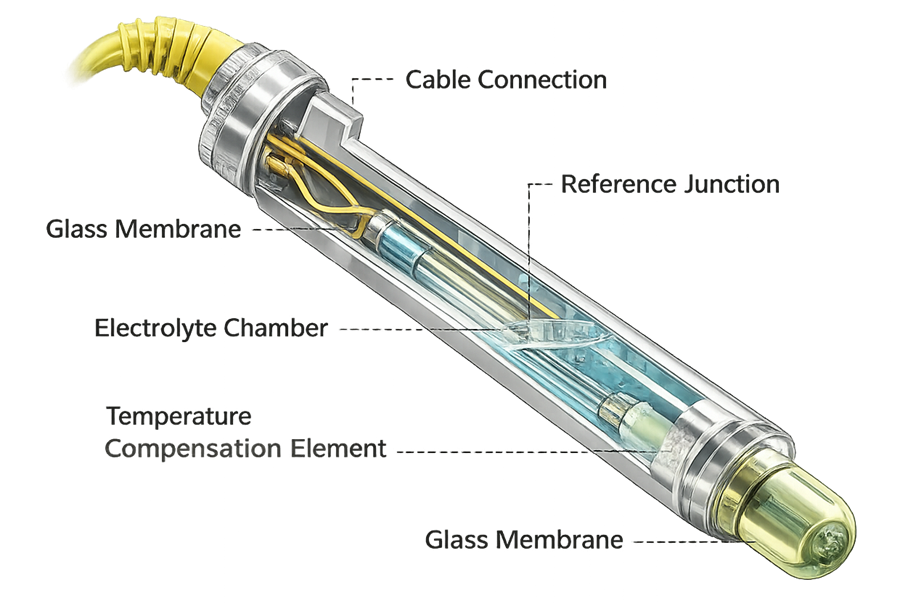
Glass electrode pH probes are common in bioprocessing. Key considerations include sterilization survivability, drift characteristics, reference junction stability, and temperature compensation accuracy.
The diagram below presents the internal architecture of a sterilizable bioreactor pH probe. The glass membrane, reference junction, electrolyte chamber, and temperature compensation element define measurement performance and susceptibility to drift following repeated steam-in-place cycles.
Dissolved Oxygen Measurement:
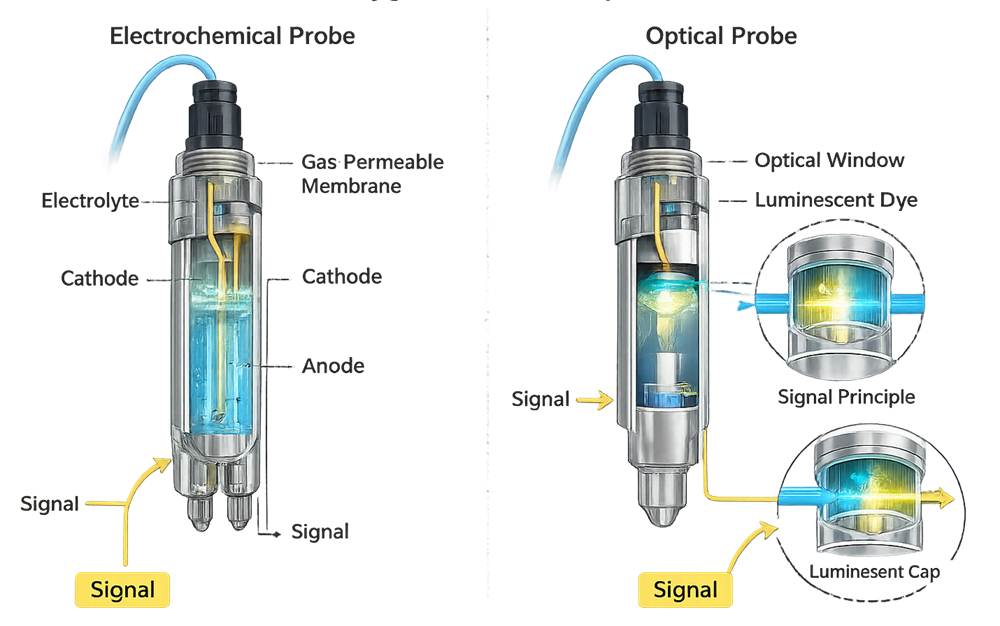
Electrochemical probes and optical probes differ in maintenance frequency, drift profile, and response time. Optical sensors generally exhibit reduced drift but require evaluation of calibration stability across sterilization cycles.
The illustration below compares the two primary dissolved oxygen measurement technologies used in bioreactor applications: electrochemical and optical probes. Electrochemical sensors rely on oxygen diffusion through a gas-permeable membrane into an electrolyte solution, where a cathode–anode reaction generates a measurable current proportional to oxygen concentration. Optical sensors use a luminescent dye exposed to excitation light; the degree of fluorescence quenching by dissolved oxygen determines the measured value. The structural differences shown in the diagram directly influence maintenance frequency, drift characteristics, response time, sterilization tolerance, and long-term stability during extended production campaigns.
Temperature Measurement:
Resistance temperature detectors are commonly used due to stability and accuracy. Redundancy may be required in large-scale systems to confirm temperature uniformity.
Pressure Measurement:
Pressure transmitters must tolerate SIP conditions and provide stable readings during aeration and backpressure control.
Selection must consider:
- Operating range
- Accuracy requirements
- Chemical compatibility
- Sterilization resistance
- Maintenance burden
- Replacement availability
Improper technology selection introduces recurring deviations.
4. Installation and Sanitary Integration
Sensor performance depends on installation configuration. Critical installation factors include:
- Proper insertion depth
- Orientation relative to mixing flow
- Avoidance of dead zones
- Sanitary tri-clamp or welded connections
- Seal integrity
Improper placement may expose probes to stagnant regions, gas pockets, or excessive shear. These conditions produce inaccurate readings independent of probe calibration. Sanitary integration must ensure:
- Cleanability
- Steam penetration during SIP
- Absence of crevices
- Mechanical stability under agitation
Probe housings and compression fittings must maintain integrity throughout repeated sterilization cycles.
The illustration below demonstrates the impact of sensor placement on measurement accuracy within a bioreactor. In the proper configuration, probes are positioned within the fully mixed liquid zone, downstream of the impeller and away from surface turbulence or gas entrainment. This location ensures representative sampling of bulk process conditions. In the improper configuration, sensors are installed in stagnant regions or near gas pockets, where localized gradients distort readings. Poor placement can produce stable but misleading values, resulting in inappropriate control responses, unnecessary adjustments, or undetected deviations. Sensor location must therefore be justified based on mixing dynamics, sparger position, and fluid flow patterns rather than mechanical convenience.

5. Signal Transmission and Control Interface
Sensors generate electrical signals that must be conditioned, transmitted, and interpreted by the control system. Integration includes:
- Signal conversion
- Shielding against electrical noise
- Grounding strategy
- Cable routing
- Transmitter configuration
- Analog or digital communication protocol
Signal degradation or electrical interference may produce erratic readings without physical probe failure. Configuration control is essential. Changes to scaling factors, input type, or signal filtering directly affect recorded values.
6. Calibration and Traceability
Calibration establishes the relationship between sensor output and true value. Effective calibration programs must ensure:
- Defined calibration frequency
- Traceability to recognized standards
- Documented adjustment history
- Acceptance criteria based on process risk
- Verification following sterilization
Calibration records must demonstrate traceability and reproducibility. Calibration factors stored within transmitters or control systems must be protected from unauthorized modification. Failure to maintain calibration integrity invalidates process data.
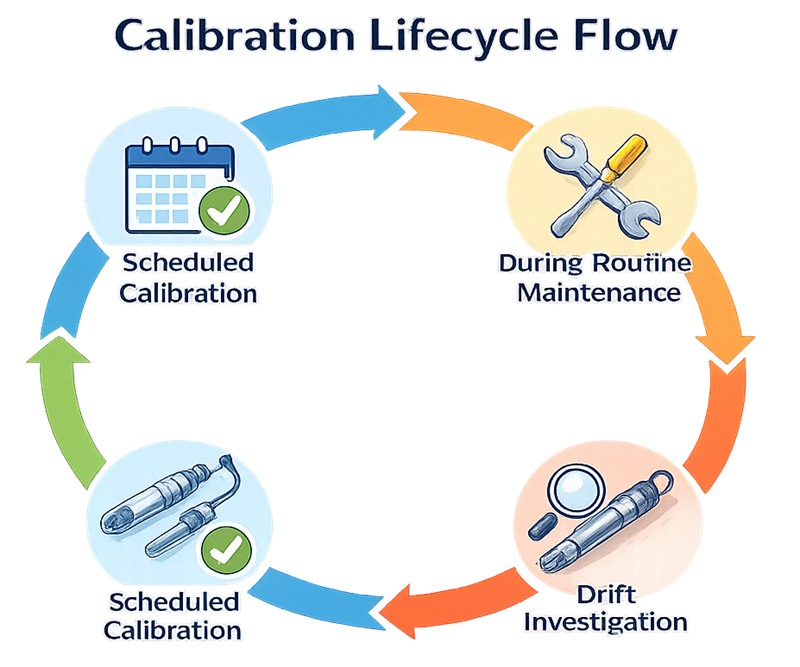
The diagram below presents the structured lifecycle of bioreactor sensor calibration from scheduled verification through adjustment, documentation, and return to service. Calibration is not a single event but a controlled process that ensures measurement accuracy is maintained throughout equipment operation. The lifecycle includes predefined calibration intervals, evaluation of as-found condition, adjustment when required, assessment of drift trends, and formal documentation before the instrument is released back into production use. When deviations or abnormal drift are identified, investigation and impact assessment must determine whether process data remain reliable. A defined calibration lifecycle ensures traceability, measurement confidence, and preservation of the validated state.
7. Sterilization Impact and Sensor Drift
Steam-in-place exposure subjects probes to thermal stress and pressure cycling. Common risks include:
- Reference junction degradation
- Electrode aging
- Seal deterioration
- Membrane damage in electrochemical DO probes
Repeated sterilization accelerates drift. Drift may be gradual and not immediately evident without trending.
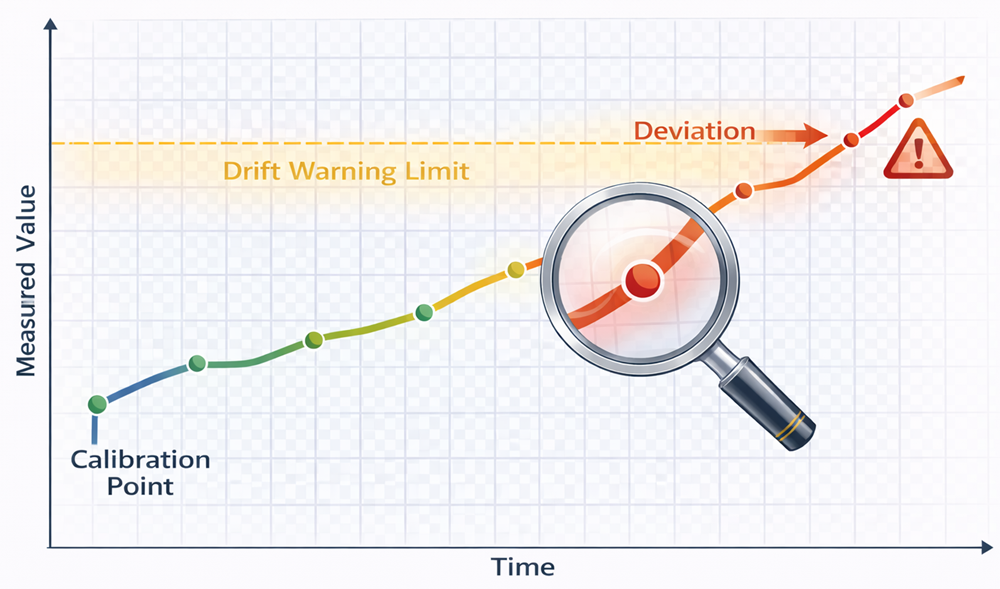
The chart below illustrates a typical sensor drift trend over time relative to predefined acceptance limits. Immediately following calibration, the sensor output aligns with the reference standard. As operating time and sterilization cycles accumulate, gradual deviation may develop due to electrode aging, membrane degradation, reference junction instability, or thermal stress. Trending measured deviation against time enables early detection of drift before the warning or action limit is exceeded.
The horizontal axis represents operating time or number of SIP cycles, while the vertical axis represents deviation from true value or calibration offset. The defined drift warning limit establishes a threshold for investigation prior to full out-of-tolerance condition. If trending indicates progressive deviation toward or beyond the acceptance band, corrective action such as recalibration, probe replacement, or impact assessment must be initiated. Routine drift analysis supports predictive maintenance, reduces unplanned failures, and preserves measurement reliability within the validated operating envelope.
Sensor integration strategy must address:
- Post-SIP verification
- Defined replacement intervals
- Trend review of calibration offsets
- Drift acceptance limits
Predictive replacement reduces unplanned failure during campaigns.
8. Sensor Failure Modes and Risk Control
Typical failure modes include:
- Signal loss
- Output freezing at fixed value
- Gradual drift
- Response time delay
- Noise spikes
- Mechanical breakage
Control systems should detect abnormal behavior through:
- Range checks
- Rate-of-change alarms
- Sensor disagreement detection
- Fail-safe interlocks
Reliance on a single unverified sensor increases operational risk. Redundancy or cross-verification may be justified for high-risk parameters.
9. Data Integrity Considerations
Sensor data supporting GMP decisions must meet data integrity principles. Key ALCOA+ attributes include:
- Attributable – Data must be linked to identifiable source and user when configuration changes occur.
- Legible – Recorded values must remain readable and interpretable throughout retention period.
- Contemporaneous – Data must be recorded at the time of measurement without reconstruction.
- Original – Electronic records must preserve the first capture of data without uncontrolled alteration.
- Accurate – Measurement must reflect true process conditions within defined uncertainty.
Additional ALCOA+ expectations include:
- Complete – No selective data deletion or omission.
- Consistent – Time sequencing and synchronization maintained.
- Enduring – Data preserved against loss or degradation.
- Available – Records retrievable throughout retention period.
Sensor integration intersects with data integrity when:
- Calibration factors are adjusted
- Setpoints are modified
- Signal scaling is changed
- Manual overrides are performed
- Firmware updates occur
While detailed Part 11 requirements are addressed in computerized system validation sections, sensor integration must ensure that measurement data remain trustworthy, traceable, and protected from unauthorized manipulation.
10. Configuration Control and Change Management
Any change to sensor type, model, firmware, scaling, or location must be evaluated for impact on validated state. Change control must assess:
- Measurement accuracy impact
- Control loop stability
- Historical data comparability
- Calibration interval modification
- Sterilization survivability
Uncontrolled configuration changes create hidden variability.
11. Redundancy and Criticality Assessment
Not all sensors carry equal process risk. Criticality assessment should consider:
- Impact of failure on product quality
- Detectability of failure
- Availability of secondary indicators
- Process tolerance to deviation
High-risk parameters may justify:
- Dual sensors
- Cross-check logic
- Periodic verification during batch
Redundancy should be risk-based, not arbitrary.
12. Lifecycle Management of Instrumentation
Sensor integration is not static. Instrumentation performance degrades over time. Lifecycle control includes:
- Preventive maintenance
- Scheduled replacement
- Trend review
- Deviation analysis
- Periodic requalification impact assessment
Sustained validated performance requires continuous oversight.
13. Conclusion
Sensor integration defines the reliability of bioreactor control. Accurate and stable measurement underpins process stability, batch consistency, and regulatory compliance.
Mechanical functionality alone does not ensure validated state. Measurement integrity, calibration traceability, controlled configuration, and adherence to ALCOA+ data integrity principles are essential components of a compliant and reliable bioprocessing system.
The next level of detail concerning computerized system validation, electronic records, and full 21 CFR Part 11 compliance should be addressed within dedicated control system and CSV sections.
