Calibration Program and Metrology Control
A compliant calibration program ensures that instruments used to control or monitor regulated processes consistently perform within approved limits and support defensible qualification data.
Purpose and Scope
In GMP-regulated environments, measurement accuracy directly affects product quality decisions and the credibility of qualification and monitoring data. Parameters such as temperature, pressure, flow, humidity, differential pressure, conductivity, and time must be measured using instruments that are demonstrably suitable for their intended use.
Calibration functions as a foundational lifecycle control. It provides documented evidence that measurement systems remain accurate, traceable, and capable throughout installation, operational qualification, performance verification, and ongoing GMP operation.
This article defines the structure, regulatory basis, and operational expectations of a compliant calibration and metrology framework. The scope applies to:
- Production equipment instrumentation
- Utility system sensors and transmitters
- Environmental monitoring devices
- Laboratory measuring instruments
- Building management and automation system sensors
- Critical monitoring devices impacting product quality
Regulatory and Standard Alignment
Calibration and metrology controls are required under established GMP and quality system regulations.
Applicable U.S. regulatory references include:
- 21 CFR 211.68 – Automatic, mechanical, and electronic equipment controls to ensure proper performance
- 21 CFR 211.160 – Laboratory controls requiring scientifically sound specifications and standards
- 21 CFR 211.194 – Complete and reliable laboratory data documentation
- 21 CFR 211.100 – Written procedures and adherence
- 21 CFR 820.72 – Control of inspection, measuring, and test equipment for medical device manufacturers
International standards frequently incorporated into measurement control programs include:
- ISO 10012 – Measurement management systems
- ISO/IEC 17025 – Competence of testing and calibration laboratories
Collectively, these requirements establish expectations for:
- Controlled and documented measurement systems
- Traceable reference standards
- Defined tolerances and acceptance criteria
- Justified calibration intervals
- Out-of-tolerance investigation and impact assessment
- Data integrity and record retention
Calibration is therefore a regulated control function, not a maintenance activity.
Governance Structure and Instrument Classification
A structured program begins with defined governance and ownership. Engineering or Metrology typically manages execution, with Quality providing oversight to ensure compliance and impact evaluation.
All instruments must be uniquely identified and entered into a controlled calibration inventory. The inventory should include:
- Equipment or instrument ID
- Location
- Parameter measured
- Range and accuracy
- Tolerance
- Calibration interval
- Criticality classification
- Last and next calibration date
Instruments should be classified according to their impact on product quality and GMP decisions. Typical categories include:
- Critical instruments influencing product acceptance
- Utility monitoring instruments affecting controlled systems
- Environmental monitoring sensors
- Informational or non-product-impact devices
Classification determines interval rigor, tolerance justification, and investigation depth.
Traceability and Metrology Control
Measurement credibility depends on traceability.
Calibration standards must be traceable to recognized national or international standards. Traceability documentation must demonstrate an unbroken chain of comparisons linking the production instrument to a higher-level reference standard.
Metrology control requires:
- Defined reference and working standards
- Documented uncertainty values
- Defined accuracy-to-tolerance ratios
- Controlled environmental conditions during calibration
- Evaluation of measurement uncertainty where applicable
Calibration certificates must clearly state:
- As-found condition
- As-left condition
- Reference standards used
- Calibration results
- Acceptance criteria
- Statement of traceability
Without traceability, measurement results cannot be defended during audit or inspection. The following hierarchy illustrates the structured traceability chain that links production instruments to recognized reference standards and defines the relationship between accuracy, uncertainty, and recalibration control.
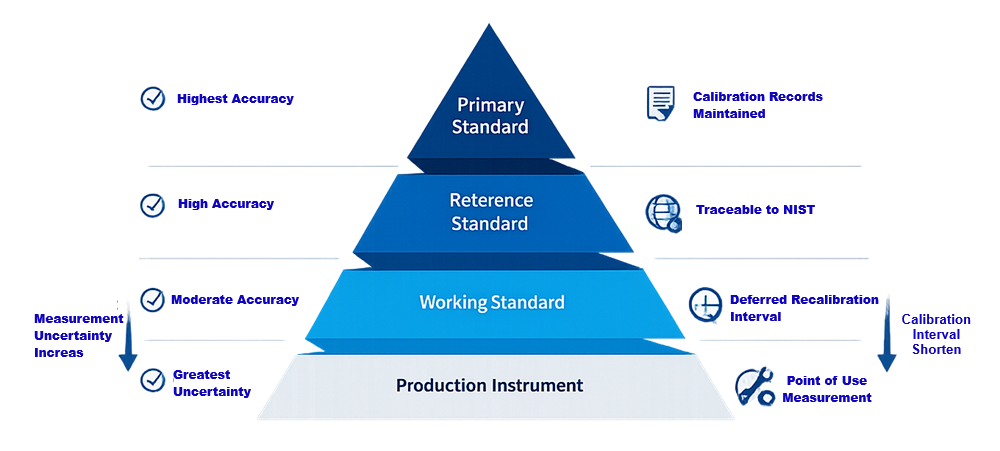
Risk-Based Calibration Intervals
Intervals must be justified, not assumed. While annual calibration remains common, interval determination should consider:
- Instrument criticality
- Historical drift performance
- Process tolerance sensitivity
- Environmental exposure
- Frequency of use
- Manufacturer recommendations
- Consequence of measurement failure
Programs that extend intervals must rely on documented trend analysis and historical performance data. Extension without statistical or documented justification is a common regulatory deficiency. Conversely, repeated drift or out-of-tolerance conditions may require interval reduction. A defensible approach includes:
- Periodic review of calibration history
- Drift analysis
- Documented rationale for interval adjustments
- Quality approval of changes
Intervals are a risk decision supported by data. Calibration intervals must be established using a documented and risk-based decision process rather than fixed default timeframes. The following logic flow illustrates the structured evaluation used to justify interval assignment, adjustment, extension, or reduction.
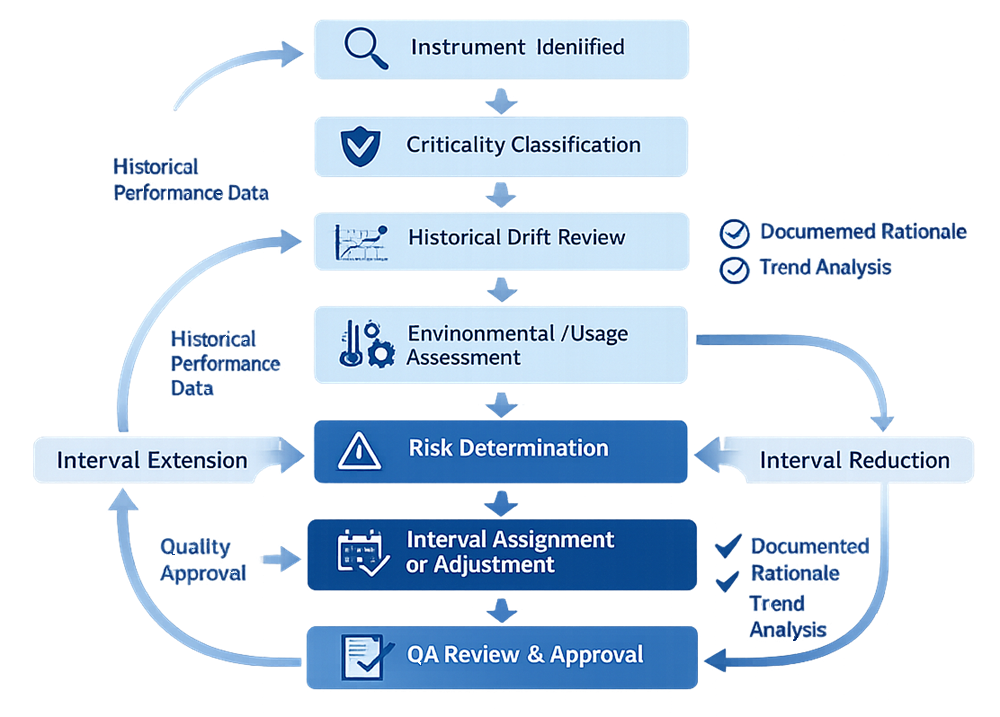
Out-of-Tolerance Handling and Impact Assessment
An out-of-tolerance condition represents a potential control failure and must be treated accordingly. When an instrument fails calibration:
- Determine magnitude and direction of error
- Identify the last acceptable calibration date
- Assess potential impact on product, utility, or environmental control
- Determine whether batch review is required
- Evaluate need for additional testing or corrective action
Impact assessment must consider:
- Duration of use since last acceptable calibration
- Process tolerance relative to measured drift
- Alarm or monitoring system performance
- Redundancy of measurement
- Historical stability of the instrument
The investigation must clearly answer whether product quality or system control may have been compromised.
Failure to perform documented impact assessment is frequently cited during regulatory inspections.
When an instrument is found out of tolerance, the response must extend beyond technical adjustment. A structured impact evaluation is required to determine whether product quality, utility performance, or environmental control may have been affected during the period of use.
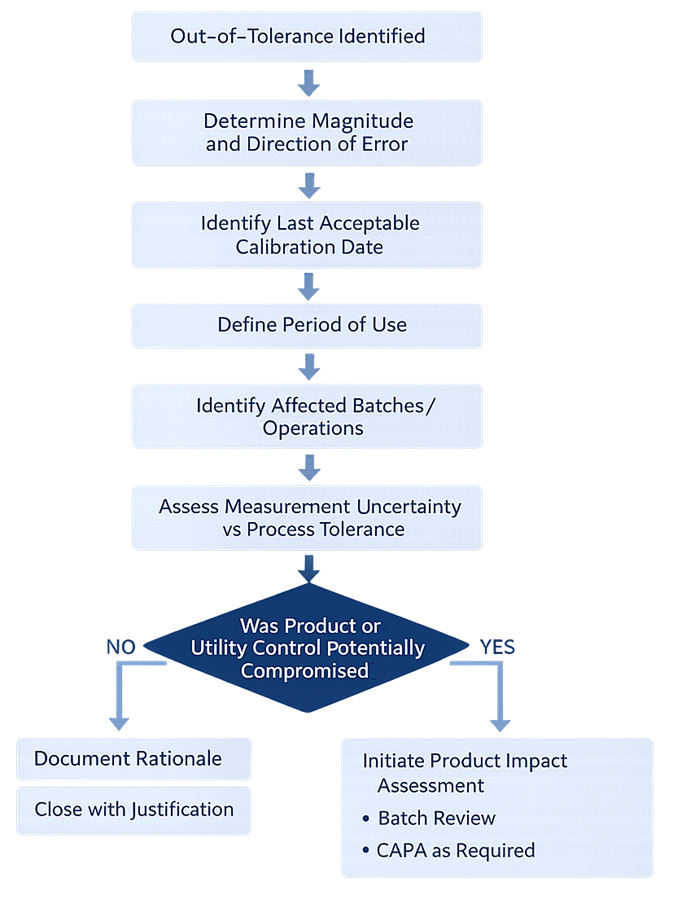
Preventive Maintenance Interface
Calibration and preventive maintenance must be coordinated. Activities such as sensor replacement, probe cleaning, component repair, or control system modification may invalidate previous calibration status. Maintenance procedures must define when recalibration or verification is required following intervention.
The calibration status of instruments must be verified prior to release of equipment for GMP use after maintenance. This interface prevents unintended return to service with uncontrolled instrumentation.
Integration with Qualification and Monitoring
Calibration underpins qualification integrity.
Installation Qualification confirms correct instrument installation. Operational Qualification relies on calibrated instruments to verify functional performance. Performance Qualification and environmental monitoring depend on measurement accuracy for ongoing control verification.
If instrumentation is not controlled, qualification data lacks credibility.
Calibration must therefore be considered a prerequisite condition for:
- Equipment release to GMP use
- Utility system operation
- Environmental monitoring programs
- Automated control system performance verification
Periodic Review and Program Effectiveness
A mature program includes periodic evaluation of overall calibration performance. Periodic review should evaluate:
- Out-of-tolerance frequency
- Drift trends
- Interval appropriateness
- Repeated failure patterns
- Vendor performance
- Instrument replacement needs
This review supports:
- Interval optimization
- Identification of unreliable instruments
- Systemic improvement opportunities
- Lifecycle risk management
Periodic review transforms calibration from reactive compliance into proactive measurement governance.
Lifecycle Control Summary
Effective measurement governance requires:
- Defined instrument classification
- Documented traceability
- Justified calibration intervals
- Structured out-of-tolerance handling
- Integration with maintenance and change control
- Periodic program review
Failure in any of these areas weakens qualification credibility and regulatory defensibility. When managed systematically, measurement control supports lifecycle reliability and sustained GMP compliance.
