Capsule Filling Equipment
Capsule filling equipment is used in pharmaceutical manufacturing to fill hard gelatin or hydroxypropyl methylcellulose (HPMC) capsules with powders, granules, pellets, or other solid formulations. The filling operation converts a prepared formulation into a finished oral dosage form by accurately dosing material into two-piece capsules and sealing them for downstream handling and packaging.
Capsule filling must ensure precise dose delivery, uniform fill weight, and consistent capsule integrity across the production batch. Because capsules are typically filled at high speeds in automated machines, the equipment must maintain tight control over capsule orientation, powder dosing, and capsule closure to ensure product quality and compliance with regulatory requirements.
Capsule filling equipment therefore plays a critical role in solid dosage manufacturing and must be properly designed, qualified, and integrated into the overall production process.
1. Purpose of Capsule Filling
The primary purpose of capsule filling is to accurately deliver the required quantity of formulation into individual capsules while maintaining consistent product quality. Capsule filling operations must ensure:
- Uniform fill weight
- Accurate active ingredient dose
- Consistent capsule body filling
- Reliable capsule closure
- Acceptable capsule appearance
Because capsules are typically used for powder formulations that cannot be easily compressed into tablets, the filling process must maintain uniform powder distribution and prevent segregation during feeding and dosing.
2. Types of Capsule Filling Equipment
Capsule filling machines used in pharmaceutical manufacturing generally fall into two main categories.
2.1 Manual and Semi-Automatic Capsule Fillers
Manual and semi-automatic machines are typically used for laboratory development, pilot-scale production, and small batches. These systems require partial operator involvement for tasks such as capsule loading, powder spreading, or capsule closing. Advantages include:
- Simple equipment design
- Lower equipment cost
- Flexibility for formulation development
However, manual systems have limited production capacity and are not suitable for large commercial manufacturing.
2.2 Automatic Capsule Filling Machines
Automatic capsule fillers are widely used in commercial pharmaceutical production.
These machines perform capsule separation, powder filling, capsule closing, and discharge in a continuous automated sequence. Capsules are processed through multiple stations mounted on a rotating turret or indexing platform. Automatic capsule fillers can achieve high production rates while maintaining consistent fill weight and capsule quality. Key advantages include:
- High production throughput
- Automated capsule orientation and separation
- Precise dosing systems
- Integration with in-process weight monitoring systems
The illustration below shows a typical automatic rotary capsule filling machine used in pharmaceutical manufacturing. Capsules are oriented, separated, filled, closed, and discharged as they move through sequential stations on a rotating turret.

3. Capsule Filling Process Overview
The capsule filling process involves several sequential mechanical steps. These include:
- Capsule feeding
- Capsule orientation
- Capsule separation
- Powder dosing
- Capsule closing
- Capsule discharge
Empty capsules are first introduced into the machine hopper and oriented so that the capsule bodies and caps are correctly positioned. The capsule halves are then separated, allowing the capsule body to receive the formulation. Powder or granules are dosed into the capsule body using a controlled dosing mechanism.
After filling, the capsule cap is repositioned and locked onto the body. The completed capsule is then discharged for downstream inspection and packaging.
The illustration below shows the basic sequence of operations during capsule filling, including capsule feeding, capsule separation, powder dosing, capsule closing, and capsule discharge.

4. Major Components of Capsule Filling Machines
Capsule filling machines consist of several key mechanical systems that control capsule handling, separation, dosing, and closure during the filling process.
Before describing these machine components, it is important to understand the structure of the capsule itself. Pharmaceutical hard capsules used in filling operations are two–piece shells composed of a longer capsule body and a shorter capsule cap. The capsule body serves as the container for the formulation, while the capsule cap functions as the closure that locks onto the body after filling.
The illustration below shows the anatomy of a typical two-piece pharmaceutical capsule. The capsule body is the longer section designed to receive the powder or granule formulation, while the capsule cap fits over the body to form the completed capsule. During the capsule filling process, the machine first separates these two components at the capsule separation station. The capsule body is held in the lower turret position where the formulation is introduced, while the capsule cap is temporarily retained above the body. After dosing is completed, the cap is returned and pressed onto the body during the capsule closing step to produce the finished capsule.
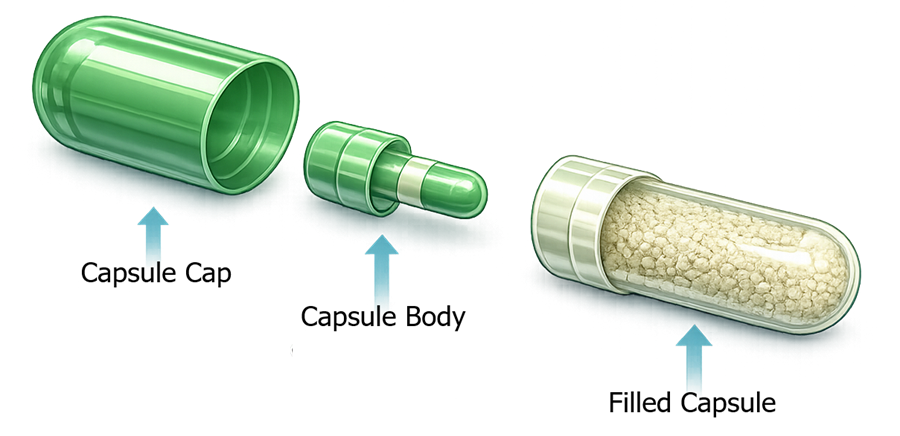
Understanding this capsule structure is essential because the entire capsule filling machine is designed to manipulate these two components during orientation, separation, dosing, and final closure.
4.1 Capsule Hopper and Feeding System
Empty capsules are stored in a hopper and delivered to the machine through a feeding system that ensures continuous supply. Capsules are oriented so that the capsule body and cap are properly aligned before entering the separation station.
4.2 Capsule Separation Station
At the capsule separation station, empty two-piece capsules are divided into their two components so that the capsule body can receive the formulation during the dosing step. Capsules arriving from the orientation system are positioned vertically within dedicated holders on the rotating turret. The capsule body is secured in the lower holder while the capsule cap is engaged by an upper holding mechanism.
Separation is typically achieved using a vacuum-assisted mechanism or a mechanical lifting system. When vacuum is applied to the upper holding ports, the capsule caps are pulled upward while the capsule bodies remain seated in the lower pockets. This controlled vertical movement separates the two capsule halves without damaging the capsule shell.
The illustration below shows a typical capsule separation station used in automatic capsule filling machines. Capsules are positioned within the turret pockets where the capsule bodies are held in the lower plate while the capsule caps are lifted upward by vacuum ports located in the upper plate. Arrows in the illustration indicate the upward motion of the capsule caps as the vacuum pulls them away from the capsule bodies. Once separated, the capsule bodies continue along the turret to the dosing station where powder or granules are introduced, while the caps remain temporarily retained in the upper holders until the capsule closing stage later in the process.

4.3 Dosing System
The dosing system determines how the formulation is introduced into the capsule body. Capsule filling machines must deliver a controlled quantity of powder or granules while maintaining consistent fill weight and avoiding segregation or material loss. Common dosing methods include:
- Dosator dosing
- Tamping pin dosing
- Auger dosing
- Pellet dosing
Each dosing method controls the quantity of material delivered to the capsule and influences fill weight accuracy, process stability, and machine speed.
The illustration below shows two widely used capsule dosing mechanisms: dosator dosing and tamping pin dosing, which are commonly implemented in automatic capsule filling machines to deliver a measured quantity of powder into the capsule body.
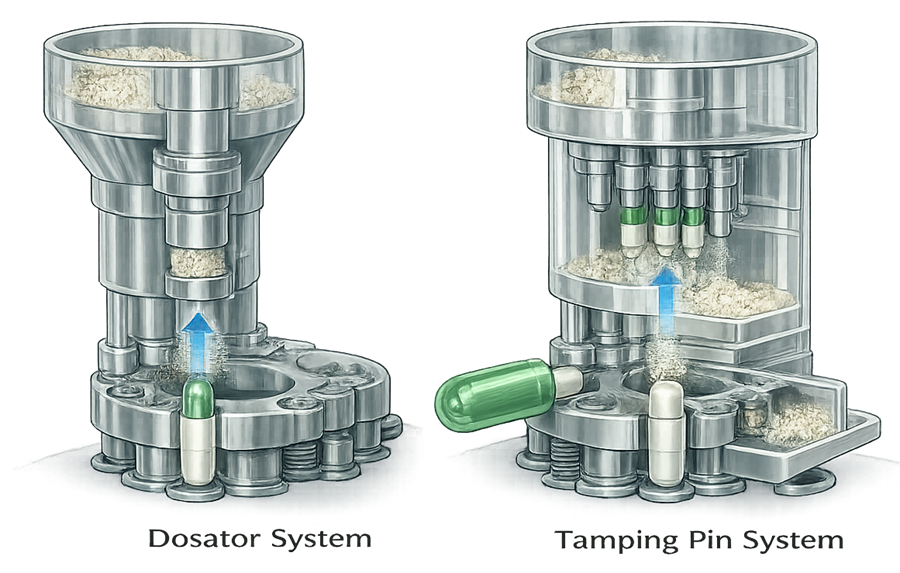
In the dosator system, a hollow dosing tube is inserted into the powder bed where vacuum or mechanical action draws powder into the tube to form a compact powder plug. The dosator then moves to the capsule filling station where a piston pushes the powder plug from the tube directly into the capsule body. The mass of the powder plug is controlled by the penetration depth of the dosator into the powder bed and the internal volume of the dosing tube.
In the tamping pin system, powder is first distributed into small dosing cavities on a rotating dosing disc. A series of tamping pins sequentially compress the powder within these cavities to form a compact plug. Once the plug reaches the required density and volume, it is transferred from the dosing disc into the capsule body positioned below. Multiple tamping stages improve plug uniformity and help control fill weight consistency.
Both dosing mechanisms are designed to produce a reproducible powder plug before insertion into the capsule body. Accurate dosing depends on powder flow characteristics, formulation density, and proper machine adjustment. The selection of dosing mechanism therefore depends on formulation properties, desired production speed, and required dosing accuracy.
4.4 Capsule Closing Station
After filling, the capsule cap is repositioned onto the filled capsule body.
Mechanical pressure locks the two capsule halves together to produce the finished capsule.
4.5 Capsule Discharge System
Completed capsules are discharged from the machine and transferred to downstream handling equipment such as capsule polishers, metal detectors, or inspection systems.
5. Critical Design Features
Capsule filling equipment must incorporate design features that support accurate dosing and reliable capsule handling. Important design considerations include:
- Precise capsule orientation mechanisms
- Reliable capsule separation systems
- Accurate dosing control
- Minimal powder loss or dust generation
- Consistent capsule closing pressure
- Ease of cleaning and changeover
The machine must also minimize capsule damage such as splitting or deformation during processing.
6. Process Monitoring and Control
Modern capsule filling machines incorporate sensors and control systems that monitor critical process parameters. Typical monitored parameters include:
- Fill weight or dosing volume
- Capsule orientation and presence
- Machine speed
- Powder level in dosing stations
- Capsule rejection rates
Some systems include in-process weight monitoring to detect fill weight deviations during production. Automated control systems can trigger alarms or stop the machine if process conditions fall outside predefined limits.
7. Qualification of Capsule Filling Equipment
Capsule filling machines must undergo formal qualification to demonstrate that the equipment is properly installed, operates within defined limits, and consistently produces capsules meeting product quality requirements. Qualification typically includes:
- Design Qualification
- Installation Qualification
- Operational Qualification
- Performance Qualification
7.1 Design Qualification
Design Qualification verifies that the capsule filling machine meets the requirements defined in the User Requirements Specification. Design evaluation typically includes:
- Production capacity
- Capsule size compatibility
- Dosing system performance
- Control system functionality
- Dust containment design
- Materials of construction and cleanability
7.2 Installation Qualification
Installation Qualification confirms that the equipment has been installed according to manufacturer specifications and engineering documentation. IQ activities typically include:
- Verification of equipment identification and model number
- Verification of mechanical installation
- Confirmation of electrical connections
- Calibration of sensors and instrumentation
- Verification of documentation and manuals
7.3 Operational Qualification
Operational Qualification verifies that the capsule filling machine operates correctly within its defined operating ranges. Operational testing typically includes:
- Capsule feeding and orientation
- Capsule separation performance
- Dosing system functionality
- Capsule closing operation
- Machine speed control
- Alarm and safety system testing
These tests demonstrate that the equipment performs correctly under controlled conditions.
7.4 Performance Qualification
Performance Qualification demonstrates that the capsule filling process consistently produces capsules meeting predefined product specifications during routine manufacturing. PQ studies typically evaluate:
- Capsule fill weight uniformity
- Content uniformity
- Capsule integrity
- Capsule rejection rate
- Capsule appearance
Multiple PQ batches are typically evaluated to demonstrate reproducibility and process stability.
7.4.1 Sampling Strategy
Sampling during PQ should represent capsules produced throughout the filling run. Samples are typically collected at defined time intervals during the batch to evaluate potential variability in fill weight or capsule quality. Samples may be collected at the beginning, middle, and end of the batch, or at fixed time intervals depending on the production run length.
The illustration below shows a typical sampling strategy used during Performance Qualification of capsule filling equipment. Capsule samples are collected at defined intervals during the filling run to verify consistent fill weight and capsule integrity.
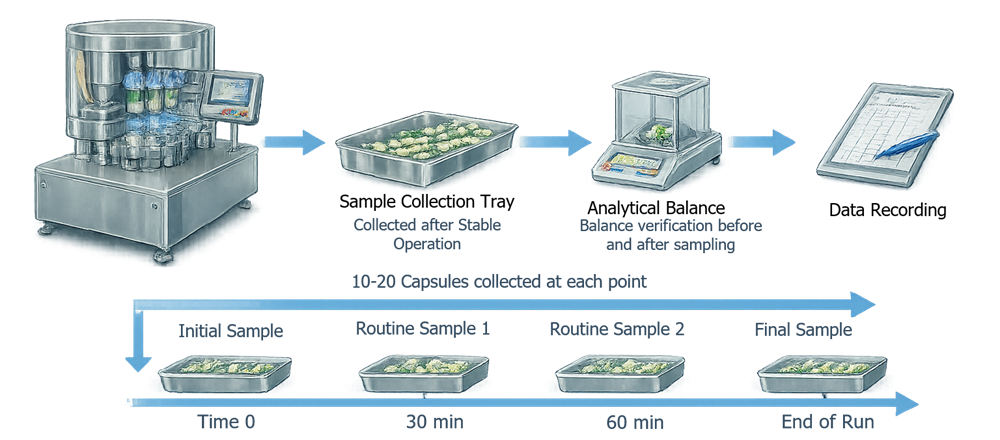
7.4.2 Statistical Evaluation
Statistical evaluation of PQ data is used to confirm that fill weight and capsule quality remain consistent throughout production. Typical statistical evaluations include:
- Mean fill weight
- Standard deviation
- Relative standard deviation
- Trend analysis across sampling intervals
These analyses confirm that the capsule filling process operates within the validated operating range.
7.4.3 Acceptance Criteria
Acceptance criteria must be defined before PQ execution. Typical acceptance criteria include:
- Fill weight within specification limits
- Capsule integrity without damage or separation
- Acceptable capsule rejection rates
- Content uniformity meeting pharmacopeial requirements
Successful completion of Performance Qualification confirms that the capsule filling equipment consistently produces capsules meeting quality specifications.
8. Integration with Downstream Processing
Capsule filling equipment operates as part of the broader solid dosage manufacturing process. Blended powder or granules are transferred to the capsule filler where capsules are filled and closed. After filling, capsules are typically transferred to downstream systems including:
- Capsule polishers
- Metal detectors
- Capsule inspection systems
- Packaging equipment
The illustration below shows how capsule filling equipment integrates within the solid dosage manufacturing process. Blended powders or granules are transferred to the capsule filler and filled capsules are subsequently inspected, polished, and packaged.
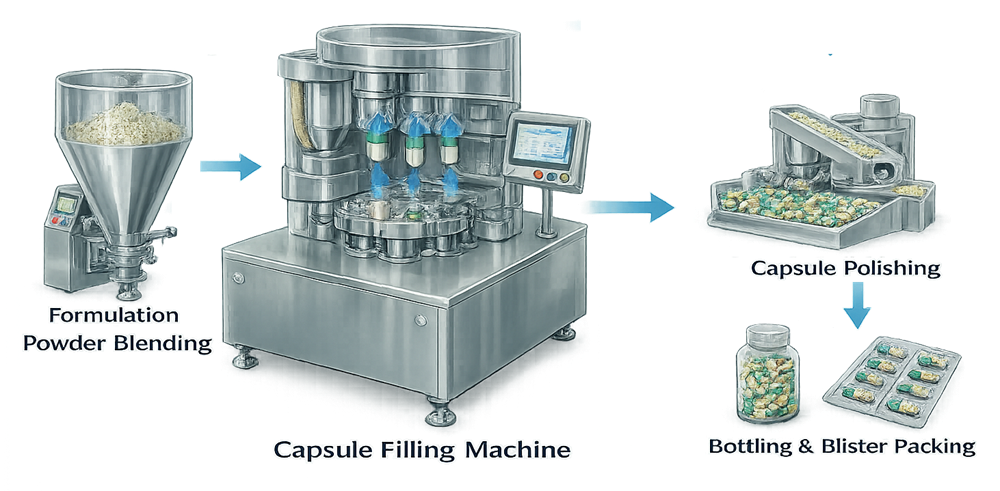
Proper integration between blending, capsule filling, inspection, and packaging ensures continuous material flow and reliable product quality. Capsule filling therefore represents a critical final manufacturing step in capsule-based solid dosage production.
