Chromatography Skid Functionality and Design
1. Purpose and Scope
Chromatography skids are engineered process systems used for purification and separation of biological or chemical components based on differential interaction with a stationary phase and a mobile phase. In regulated pharmaceutical environments, chromatography skids are typically deployed for purification of active pharmaceutical ingredients, recombinant proteins, vaccines, monoclonal antibodies, and other biologically derived products.
This article focuses on functional design, engineering architecture, and operational principles of chromatography skids used in cGMP manufacturing. Column chemistry and lifecycle maintenance are addressed separately.
The objective is to define how the skid functions as an integrated process system and to identify the critical design elements that impact product quality, process control, and validation strategy.
2. System Overview
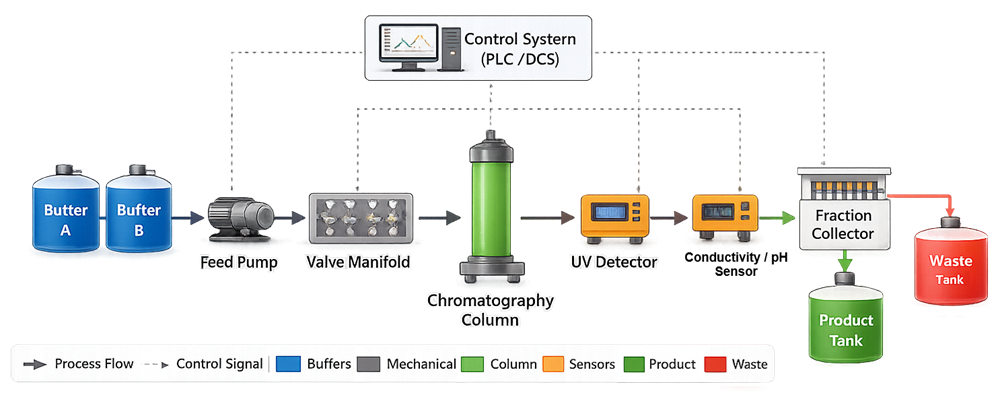
A chromatography skid is an integrated assembly of mechanical, fluidic, electrical, and control components designed to:
• Deliver and proportion mobile phase buffers
• Control flow rate and pressure
• Direct flow through selected columns
• Monitor process parameters in real time
• Collect product fractions
• Support cleaning and sanitization cycles
The skid serves as the control and fluid distribution platform, while the column provides the separation mechanism. In GMP environments, chromatography skids may be configured as:
• Single-column batch systems
• Multi-column switching systems
• Continuous chromatography platforms
• Single-use flow paths
• Stainless steel clean-in-place systems
Configuration selection depends on process throughput, product sensitivity, contamination control strategy, and facility infrastructure.
The diagram below illustrates the functional architecture of a typical chromatography skid used in GMP manufacturing. It depicts buffer sources, controlled pumping, automated valve routing, the separation column, inline detection, and fraction collection. The illustration defines system boundaries and clarifies how mobile phase, process monitoring, and product routing are integrated within a single purification platform.
The image below shows a real-world chromatography skid installed in a GMP manufacturing environment. Visible components typically include process pumps, stainless steel tubing, automated valves, instrumentation panels, and control interface. The physical layout demonstrates how fluid routing, sensor placement, and structural support are integrated into a compact purification platform.

3. Core Functional Subsystems
3.1 Buffer Delivery System
The buffer delivery subsystem provides controlled mobile phase flow to the column. It typically includes:
• Feed pumps, often diaphragm or piston type
• Inline pressure sensors
• Flow meters
• Degassing systems
• Pulse dampeners
Critical design considerations include:
• Flow accuracy and repeatability
• Pressure rating relative to column limits
• Chemical compatibility with buffers
• Dead leg minimization
Flow stability directly influences resolution, peak shape, and process reproducibility.
3.2 Valve and Flow Path Network
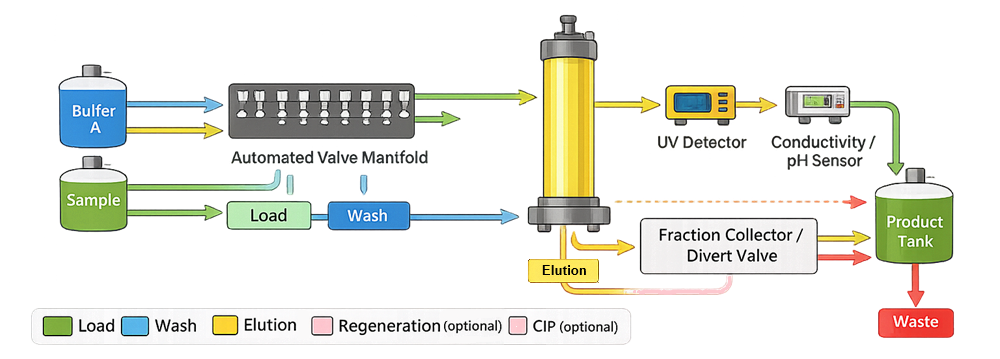
Automated valves direct fluid through selected pathways, including:
• Load
• Wash
• Elution
• Regeneration
• Cleaning
Valve design must ensure:
• Minimal internal volume
• Low hold-up volume
• Chemical compatibility
• Reliable actuation under process pressure
Flow path architecture must prevent unintended cross-contamination and allow validated cleaning between campaigns when applicable.
The following schematic illustrates simplified valve routing logic within a chromatography skid. It demonstrates how automated valves redirect flow between load, wash, elution, regeneration, and cleaning pathways. Proper valve sequencing prevents cross-contamination and ensures defined process phase transitions.
3.3 Column Interface and Support
The skid provides structural and fluidic interface to the chromatography column. Design considerations include:
• Pressure rating of fittings and tubing
• Secure column mounting
• Controlled compression where applicable
• Prevention of air ingress
The skid must prevent pressure spikes that could damage column packing or affect separation efficiency.
The image below shows a packed chromatography column used for large-scale purification. The column contains the stationary phase media responsible for separation, while the skid controls flow, pressure, and process conditions. Column diameter, height, and packing integrity directly influence resolution and product recovery.

3.4 Instrumentation and Monitoring
Typical instrumentation includes:
• Pressure transmitters
• Flow sensors
• Conductivity probes
• UV detectors
• pH sensors
• Temperature sensors
Instrumentation supports:
• Real-time monitoring of critical process parameters
• Detection of breakthrough or elution profiles
• Process endpoint determination
• Alarm generation for excursions
Sensor accuracy and response time directly affect batch control and release decisions.
3.5 Fraction Collection and Product Routing
Post-column flow is directed to:
• Waste
• Intermediate collection vessels
• Product hold tanks
Automated fraction collection is controlled by:
• UV signal thresholds
• Time-based switching
• Volume-based criteria
Design must ensure traceability of collected fractions and prevent misrouting events.
3.6 Cleaning and Sanitization Integration
Chromatography skids may support:
• Clean-in-place cycles
• Sanitization cycles
• Storage buffer circulation
For stainless steel systems, CIP integration requires:
• Defined spray or flow coverage
• Validated cleaning agent concentration
• Controlled contact time
• Verified drainability
For single-use systems, cleaning validation is replaced by disposable path control and component integrity verification.
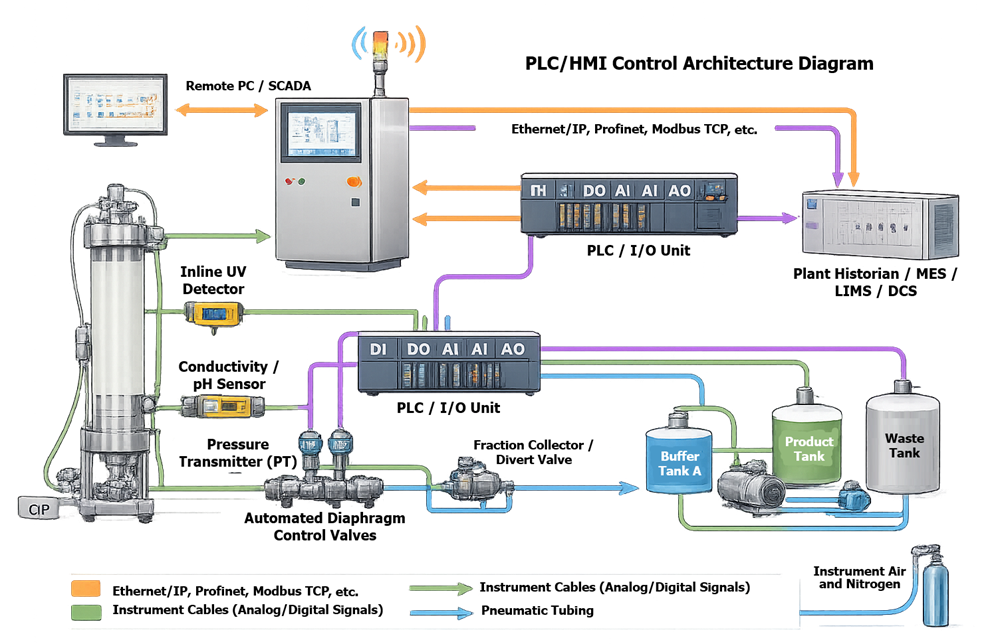
4. Control System Architecture
Modern chromatography skids incorporate PLC- or DCS-based control systems with:
• Recipe management
• Parameter setpoint control
• Alarm management
• Electronic batch recording
• Data logging and trending
In regulated environments, control systems must support:
• Access control
• Audit trails
• Electronic signatures where applicable
• Secure data storage
Software functionality influences validation scope and Part 11 compliance requirements.
The diagram below illustrates the instrumentation and control structure of a chromatography skid. Process sensors such as pressure, flow, conductivity, UV absorbance, and pH transmit signals to the control system, which executes recipe logic, manages alarms, and records batch data. This architecture defines the validation scope for software and electronic records compliance.
5. Pressure and Flow Dynamics
Chromatography performance is sensitive to:
• Flow rate stability
• Pressure gradients
• Column backpressure
• System dead volume
The skid must be designed to:
• Prevent cavitation
• Avoid excessive pressure drop
• Maintain laminar flow within defined operating range
• Protect the column from overpressure conditions
Pressure interlocks are typically implemented to protect both column integrity and product quality.
6. Single-Use Versus Stainless Steel Designs
Stainless Steel Systems
• Designed for repeated use
• Require validated CIP and sanitization
• Higher initial capital investment
• Greater mechanical durability
Single-Use Systems
• Disposable fluid pathways
• Reduced cleaning validation burden
• Lower cross-contamination risk
• Dependency on supplier quality and integrity testing
The comparison below highlights structural and operational differences between stainless steel chromatography systems and single-use flow path configurations. Stainless steel systems rely on validated CIP cycles and fixed piping, while single-use platforms utilize disposable tubing sets and modular assemblies to reduce cleaning burden and cross-contamination risk.

Selection impacts lifecycle control strategy and qualification depth.
7. Integration with Upstream and Downstream Operations
Chromatography skids typically interface with:
• Bioreactor harvest systems
• Depth filtration units
• Ultrafiltration/diafiltration systems
• Buffer preparation systems
Proper integration requires:
• Pressure compatibility
• Flow synchronization
• Transfer line validation
• Defined material traceability
The skid must function as part of a controlled process train rather than as a standalone unit.
The diagram below illustrates the position of the chromatography skid within a typical biopharmaceutical process train. It receives clarified harvest from upstream processing and transfers purified product to downstream concentration or formulation systems. Defining these interfaces is essential for impact assessment and validation boundary determination.

8. Critical Design Attributes Affecting Validation
From a validation perspective, key design attributes include:
• Defined maximum operating pressure
• Flow control accuracy
• Sensor calibration accessibility
• Software configuration control
• Cleanability or disposability strategy
• Alarm and interlock robustness
These attributes determine qualification depth, risk assessment outcomes, and lifecycle control requirements.
9. Lifecycle Considerations
Although detailed qualification is addressed separately, skid functionality directly influences:
• Installation qualification scope
• Operational qualification parameter challenges
• Performance qualification reproducibility
• Change management impact assessment
• Periodic review requirements
Engineering design decisions made at procurement stage have long-term regulatory implications.
10. Conclusion
A chromatography skid is not simply a pump-and-valve assembly. It is a controlled process platform that governs fluid dynamics, process monitoring, and product fractionation. Its design must support consistent separation performance, prevent cross-contamination, protect column integrity, and ensure traceable, compliant operation within a GMP environment.
Understanding skid functionality and engineering design is foundational before addressing column chemistry, maintenance strategy, and formal qualification activities.
