Change Control Impact on Validation
Change control is one of the most critical control mechanisms in regulated pharmaceutical and medical device environments. Its primary purpose is to ensure that changes to validated systems do not compromise the validated state.
Any change that may affect a process, system, piece of equipment, utility, facility, or computerized system must be formally evaluated for its impact on validation. Failure to do so is not a procedural gap; it is a compliance risk.
Role of Change Control in Validation
Validation establishes that a system is fit for its intended use. Change control ensures that this state is maintained over time.
Change control applies to, but is not limited to:
- Equipment modifications or replacements
- Software updates or configuration changes
- Process parameter changes
- Utility or facility alterations
- Material, component, or supplier changes
- Procedural or operational changes affecting validated systems
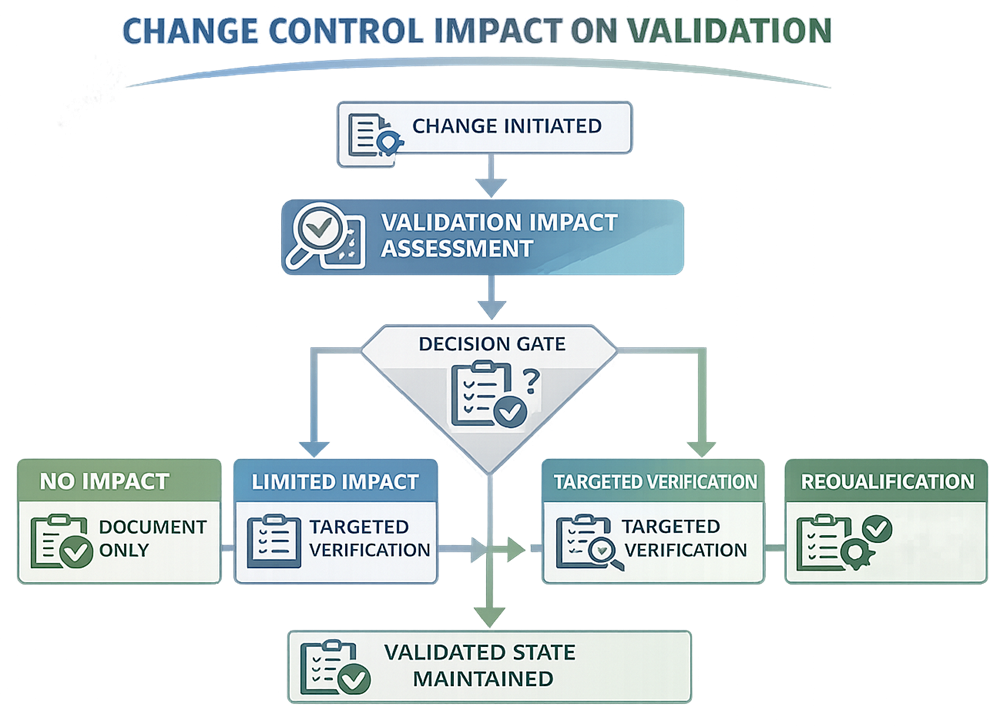
Not every change requires requalification, but every change requires impact assessment.
Impact Assessment
When a change is proposed, a documented impact assessment must be performed to evaluate its potential effect on the validated state.
A proper validation impact assessment considers:
- Impact on product quality and patient safety
- Impact on critical process parameters and controls
- Impact on data integrity
- Impact on regulatory compliance
- Interaction with previously qualified or validated elements
The outcome of the impact assessment determines whether:
- No validation action is required
- Limited verification or targeted testing is sufficient
- Partial or full requalification is necessary
Strong validation programs justify decisions based on risk and rationale, not convenience.
Change Evaluation and Classification
Changes are evaluated to determine their significance and classification. This step establishes whether the change is:
- Minor and administrative
- Technical but non-impacting
- Potentially impacting the validated state
- Major and requiring formal requalification
Changes with any potential impact on validated systems must proceed through formal change control, including documented review and approval prior to implementation.
Change Control Process
A robust change control process defines the required steps to manage change in a controlled and compliant manner. At minimum, this includes:
- Change request initiation
- Impact assessment
- Technical and quality review
- Approval by appropriate stakeholders
- Controlled implementation
- Post-implementation verification as required
Change control is not an approval exercise. It is a risk management activity.
Requalification and Revalidation Considerations
Changes that impact validated functions may require requalification activities to demonstrate that acceptance criteria continue to be met.
Depending on the nature of the change, this may include:
- Design review or Design Qualification updates
- Installation verification
- Targeted Operational Qualification testing
- Performance verification under routine conditions
The scope of requalification must be justified, traceable, and proportional to the change.
Over-validation is a sign of weak change control, not strong compliance.
Documentation and Traceability
All change-related validation activities must be documented to maintain traceability and inspection readiness.
This may involve updates to:
- Validation protocols or test scripts
- Validation reports or summaries
- Risk assessments
- Drawings, specifications, or configuration records
- SOPs or operating procedures
Documentation must clearly demonstrate the system’s validated state before and after the change.
Regulatory Expectations
Regulatory authorities expect organizations to:
- Identify changes that may affect validated systems
- Assess impact before implementation
- Justify validation decisions based on risk
- Maintain documentation supporting continued control
Weak change control is one of the most common contributors to validation-related inspection findings.
Key Principle
Change control does not exist to slow progress. It exists to ensure that progress does not undermine control.
A well-designed change control process protects the validated state while allowing systems and processes to evolve in a controlled, compliant manner.
