Change Impact on Calibration and Maintenance
Modifications affecting instrumentation, measurement systems, maintenance strategies, or reliability controls must be evaluated to ensure continued suitability, traceability, and validated state.
Change management within this domain is not a general facility activity. It specifically addresses how changes influence measurement accuracy, maintenance effectiveness, and lifecycle control integrity.
Purpose and Scope
Calibration and preventive maintenance programs are structured control systems. Any change to instruments, components, software, tolerances, intervals, or maintenance tasks may alter system performance or regulatory compliance. This article defines how change evaluation applies specifically to:
- Measurement instruments and transmitters
- Calibration standards and reference equipment
- Calibration intervals and tolerances
- Preventive maintenance tasks and frequencies
- Critical spare part substitutions
- Automation or firmware updates affecting measurement
- Maintenance procedures impacting validated state
The objective is to ensure that changes do not compromise traceability, reliability, or qualification status.
Regulatory and Quality Alignment
Change impact on measurement and maintenance systems aligns with:
- 21 CFR 211.68 – Equipment performance controls
- 21 CFR 211.67 – Maintenance of equipment
- 21 CFR 211.160 – Scientifically sound laboratory controls
- 21 CFR 820.72 – Control of inspection, measuring, and test equipment
These regulations require that systems remain suitable for their intended use following modification.
Types of Changes Affecting Calibration Programs
Modifications to instrumentation or maintenance strategy frequently interact with calibration controls, reliability assumptions, and qualification status. The following diagram illustrates how change management interfaces with these control systems.
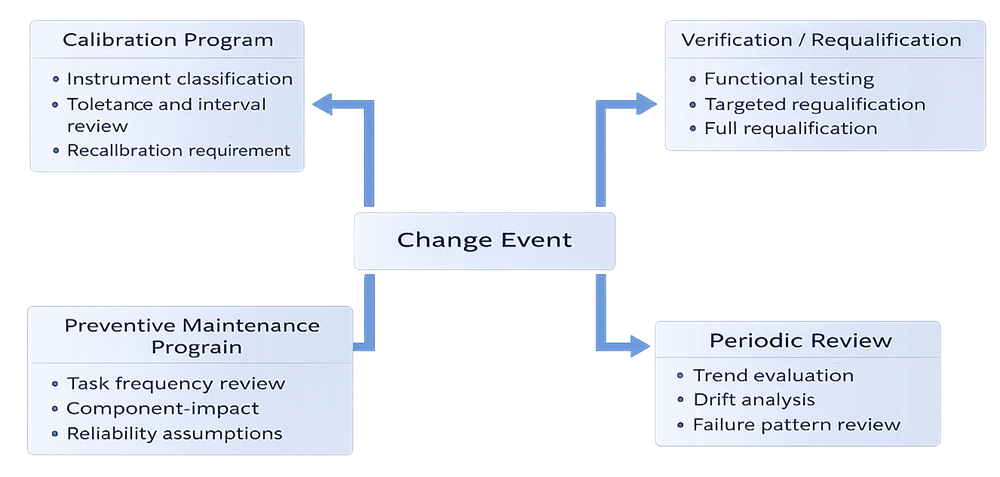
Instrument Replacement
Replacement of sensors, transmitters, or gauges must evaluate:
- Equivalent range and accuracy
- Compatibility with validated operating limits
- Alarm setpoint alignment
- Need for recalibration
- Impact on historical drift trending
Even like-for-like replacements require documented evaluation.
Tolerance or Range Modification
Changes to acceptance criteria or instrument range may affect:
- Process capability
- Alarm performance
- Risk classification
- Qualification acceptance criteria
Such changes typically require risk assessment and may require requalification testing.
Calibration Interval Adjustment
Proposed interval extension or reduction must be supported by:
- Historical drift data
- Out-of-tolerance frequency review
- Risk evaluation
- Quality approval
Interval changes are controlled modifications, not administrative edits.
Calibration Standard Changes
Replacement of reference standards or external calibration vendors must evaluate:
- Traceability documentation
- Uncertainty values
- ISO/IEC 17025 compliance where applicable
- Impact on measurement uncertainty
Changes at the top of the metrology hierarchy may affect all downstream instruments.
Types of Changes Affecting Maintenance Programs
Maintenance Task Modification
Adjusting preventive maintenance scope or frequency must consider:
- Failure history
- Reliability trends
- Consequence of failure
- Interaction with qualification status
Elimination of maintenance tasks requires documented technical justification.
Component Substitution
Substitution of critical parts such as:
- Motors
- Pumps
- Control boards
- Sensors
- Actuators
Must evaluate:
- Performance equivalency
- Impact on control stability
- Effect on validated operating parameters
Not all substitutions are neutral.
Software or Firmware Updates
Updates affecting:
- Alarm logic
- Control parameters
- Data acquisition
- Monitoring resolution
May require:
- Functional verification
- Targeted requalification
- Documentation revision
Software changes affecting measurement are not minor events.
Impact Evaluation Framework
When a change affecting calibration or maintenance is proposed, evaluation should determine:
- Does the change alter measurement accuracy or uncertainty?
- Does it affect traceability?
- Does it modify validated operating limits?
- Does it alter alarm functionality?
- Does it change maintenance frequency or reliability assumptions?
- Does it require recalibration or verification testing?
If the validated state may be affected, requalification scope must be defined.
Outcomes may include:
- Documentation update only
- Verification testing
- Targeted requalification
- Full requalification
The rationale must be documented and technically defensible. Changes affecting instrumentation or maintenance strategy must undergo structured evaluation to determine whether validated state, traceability, or reliability assumptions are affected. The following model illustrates the impact assessment pathway specific to calibration and maintenance systems.
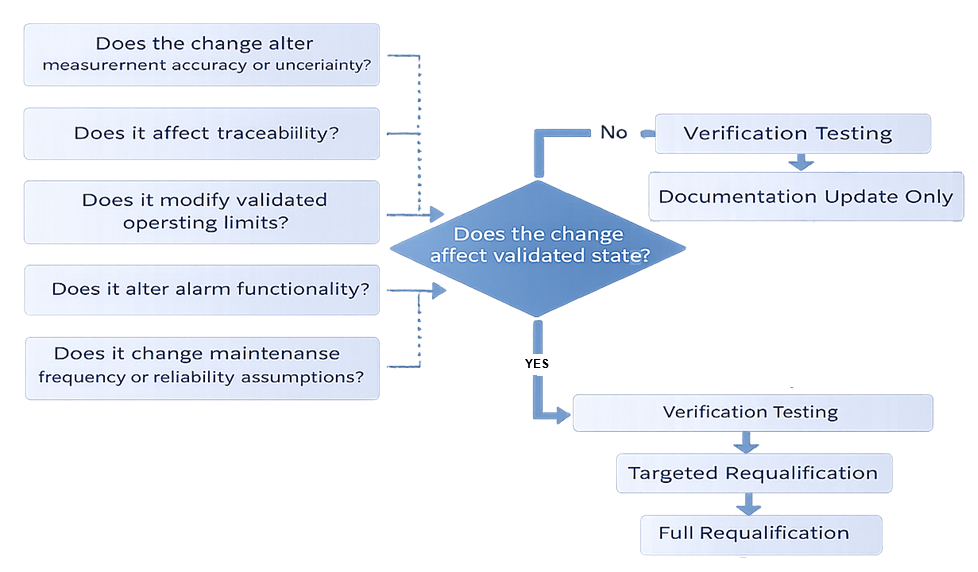
Integration with Periodic Review
Change activity should be considered during periodic program review.
Frequent interval changes, repeated instrument replacements, or recurring maintenance adjustments may indicate:
- Design instability
- Inadequate initial classification
- Improper interval justification
- System reliability weaknesses
Change trends provide valuable reliability insight.
Lifecycle Control Summary
Effective governance of calibration and maintenance programs requires:
- Formal evaluation of instrument and maintenance-related changes
- Risk-based assessment of measurement and reliability impact
- Defined verification or requalification when required
- Alignment with metrology traceability and maintenance strategy
- Documentation supporting defensible decisions
Changes affecting measurement systems or reliability controls must never bypass structured evaluation. Measurement integrity and system stability depend on disciplined change management.
