Clean-in-Place (CIP) Utility Systems
Clean-in-Place (CIP) utility systems are engineered to deliver controlled, repeatable cleaning of product-contact equipment without disassembly. In GMP manufacturing, CIP systems are considered direct product-impact utilities because their performance directly affects equipment cleanliness, cross-contamination risk, and ultimately product quality. As such, CIP systems must be designed, qualified, and maintained under formal lifecycle control.
Typical CIP Utility System Architecture
Clean-in-Place utility systems are centralized cleaning utilities designed to supply controlled cleaning solutions to GMP process equipment through dedicated supply and return circuits. The CIP system boundary typically includes the CIP skid, associated tanks, pumps, heating elements, instrumentation, and control system, as well as defined interfaces to process equipment and drain or recovery systems.
The diagram below illustrates a representative CIP utility system architecture and its primary interfaces with GMP process equipment.
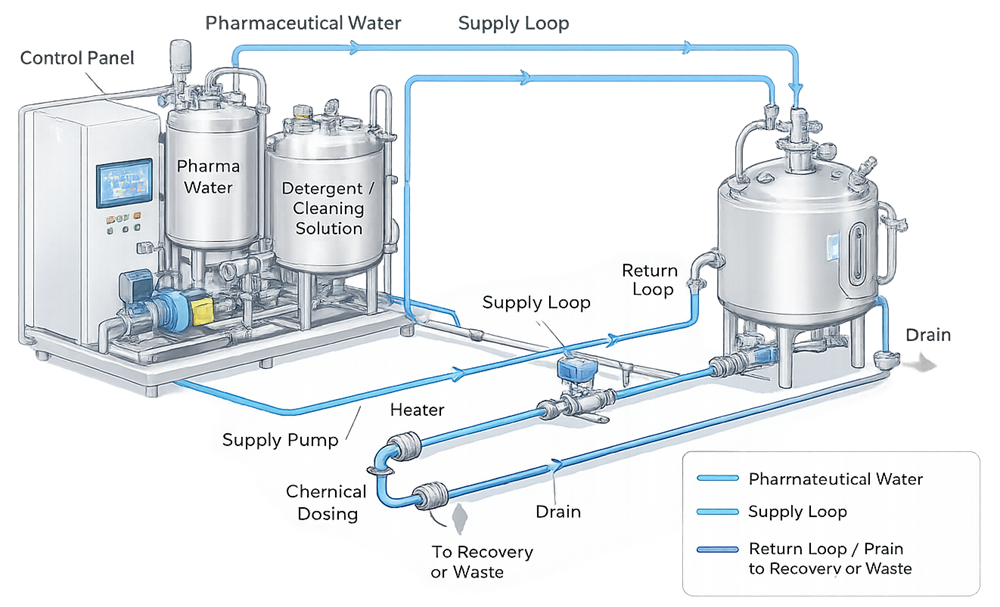
In this configuration, cleaning solutions are prepared and conditioned within the CIP skid and delivered to process equipment through controlled supply paths. Return flow is routed back to the CIP system or to drain, depending on the cleaning phase. System design must support complete coverage of product-contact surfaces, adequate hydraulic performance, and effective drainability to prevent residue accumulation or cross-contamination.
A representative CIP skid is shown below to illustrate a typical multi-tank, fully integrated Clean-in-Place utility system used in GMP manufacturing.

CIP System Design Considerations
Effective CIP performance begins with proper system design. The utility system must be capable of delivering cleaning solutions to all product-contact surfaces under defined and reproducible conditions.
Key design aspects include:
- Utility interfaces
Integration with pharmaceutical water systems, process equipment, drains, and return circuits. Supply and return paths must be designed to avoid dead legs and unintended hold-up volumes. - Hydraulic performance
Flow rate and pressure must be sufficient to achieve turbulent flow where required. Line sizing, pump selection, and spray device design are critical to cleaning effectiveness. - Temperature control
Heating capability must support defined cleaning temperatures and hold times. Temperature uniformity and control accuracy are expected to be demonstrated. - Chemical delivery and control
Detergent and cleaning agent dosing must be accurate, repeatable, and traceable. Concentration control is typically achieved through conductivity, flow-based dosing, or validated preparation tanks. - Material compatibility and drainability
All wetted materials must be compatible with cleaning agents and temperatures. Piping and equipment must be self-draining or demonstrably drainable to prevent residue carryover. - Automation and control
CIP systems are commonly automated. Control logic, sequence control, interlocks, and alarms must be defined and aligned with cleaning intent.
Qualification Strategy for CIP Utility Systems
CIP systems are qualified to demonstrate that they can consistently deliver the cleaning conditions required by the validated cleaning process. Qualification focuses on system capability rather than product-specific residue limits, which are typically addressed separately in cleaning validation.
Effective CIP performance depends on control of defined critical parameters, which must be monitored and verified during qualification.
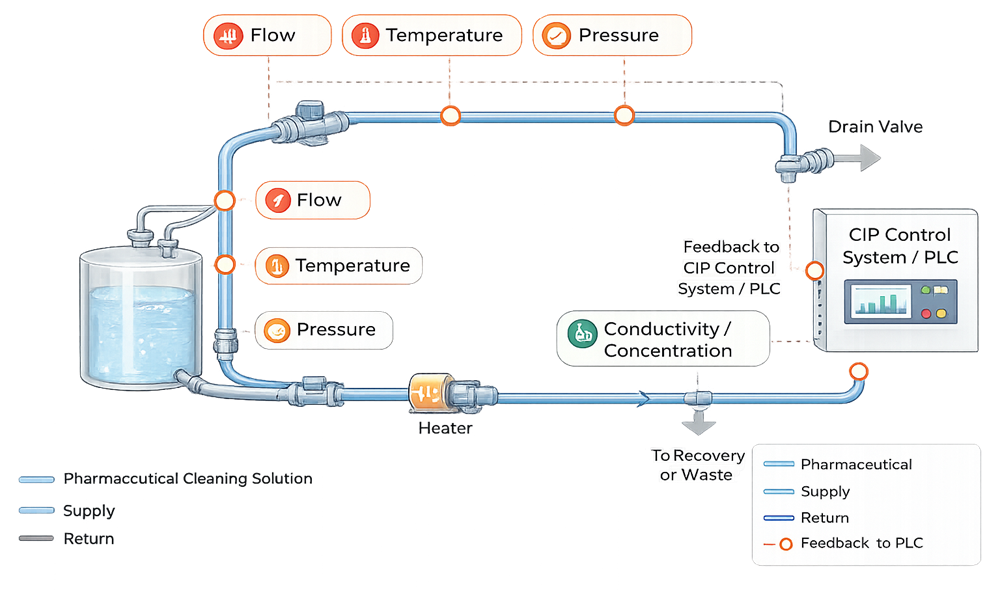
Installation Qualification (IQ)
IQ confirms that the CIP utility system is installed in accordance with approved design documentation and manufacturer specifications. Typical IQ elements include:
- Verification of system components, materials of construction, and piping configuration
- Confirmation of utility connections and instrumentation installation
- Review of P&IDs, wiring diagrams, and control hardware
- Calibration status of critical instruments such as flow, temperature, pressure, and conductivity sensors
Operational Qualification (OQ)
OQ demonstrates that the CIP system operates as intended across defined operating ranges. Testing typically includes:
- Verification of flow rates, pressures, and temperatures at representative points
- Control system functionality including sequences, interlocks, and alarms
- Chemical dosing accuracy and repeatability
- Verification of automated cycle execution and data recording
OQ testing is normally performed using water or surrogate solutions and focuses on system performance, not soil removal.
Performance Verification
Performance verification confirms that the CIP system can reproducibly deliver the defined cleaning parameters under routine conditions. This may include:
- Repeatability of critical parameters across multiple cycles
- Worst-case configuration challenges such as longest circuits or lowest flow paths
- Confirmation that monitoring and recording systems capture required data
Cleaning validation activities, including residue or bioburden evaluation, are typically executed using the qualified CIP system but are addressed in separate protocols.
CIP utility systems follow a defined qualification and lifecycle control model to ensure continued fitness for intended use.
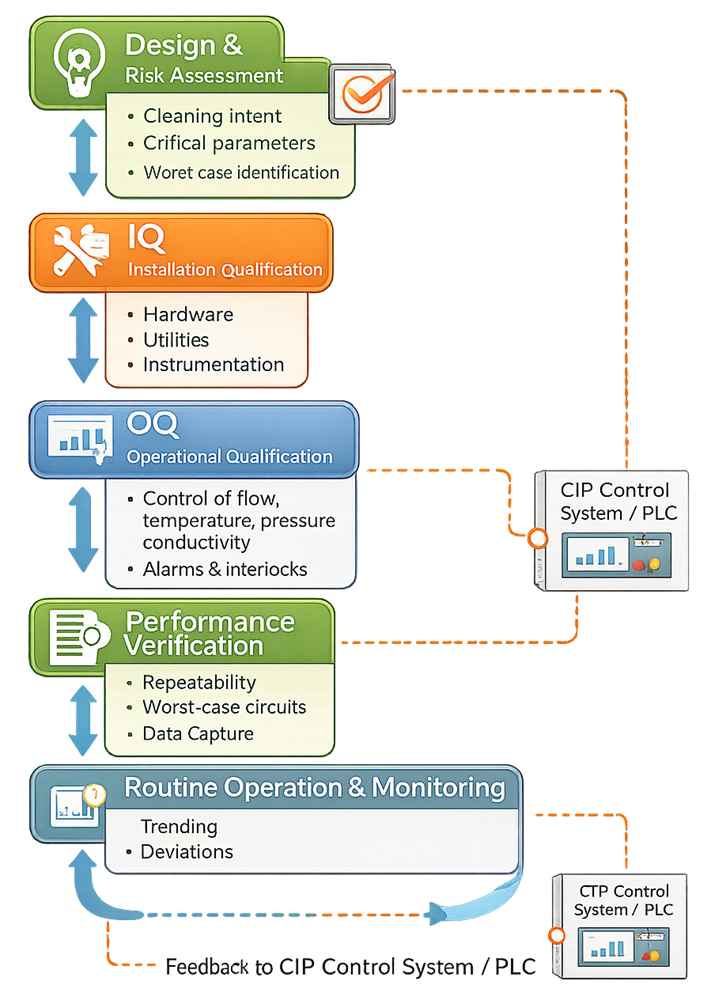
Ongoing Control and Lifecycle Management
Once qualified, CIP utility systems must remain under formal control to ensure continued suitability. This includes:
- Routine monitoring of critical parameters
- Review of alarms, deviations, and trend data
- Change control for equipment, software, utilities, or cleaning parameters
- Periodic review and requalification based on risk and performance history
Document review alone is acceptable only when performance history demonstrates stable control and no meaningful changes have occurred.
Summary
CIP utility systems are not ancillary services. They are GMP-critical utilities that must be deliberately designed, thoroughly qualified, and actively managed throughout their lifecycle. Proper control of CIP systems provides the foundation for effective cleaning validation, cross-contamination prevention, and sustained regulatory compliance.
