Clean Steam Systems for GMP Applications
Purpose and Scope
Clean steam systems are GMP utilities intended for direct contact applications where steam or its condensate may contact product, product-contact surfaces, or critical process equipment. This article defines where clean steam is required, why it is required, and how its use is bounded in regulated pharmaceutical operations.
The objective here is to establish clear applicability, eliminate ambiguity with plant steam, and set defensible expectations consistent with long-standing industry practice.

What Clean Steam Is
Clean steam is steam generated from treated feed water using equipment and distribution systems designed to control chemical purity and prevent contamination. Its defining characteristic is not pressure or temperature, but condensate suitability for its intended GMP use.
Clean steam is a utility. It is expected to operate in a stable and predictable manner. Deviations from expected performance indicate a potential loss of control and require evaluation.
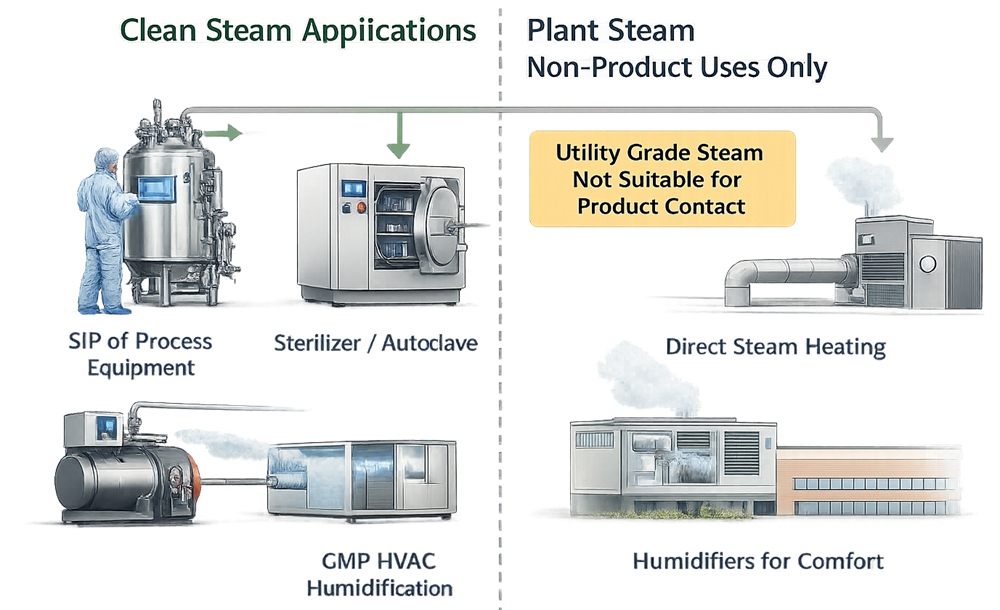
Clean Steam Application Boundaries
Clean steam is not applicable to:
- Plant or utility steam treated only by downstream filtration
- Steam used solely for non-contact space heating applications
- Steam systems intended to replace Water for Injection distribution
Clear definition of clean steam application boundaries is essential. Many compliance deficiencies originate from treating plant steam as functionally equivalent to clean steam without addressing differences in system design, materials of construction, and control strategy.

Typical GMP Applications
Clean steam is typically required for:
- Sterilization-in-place of product-contact equipment
- Autoclaves and sterilizers
- Humidification of classified spaces where steam contacts supply air
- Heating of product-contact surfaces where condensate formation is possible
If condensate can touch product, product-contact surfaces, or critical air streams, clean steam is in scope. There is no middle ground here.
Application Boundaries and Risk Perspective
Not every steam use in a GMP facility requires clean steam. Proper boundary definition is essential:
- Non-contact heating applications are usually suitable for plant steam
- Utilities isolated by heat exchangers may not require clean steam upstream
- Misclassification increases cost without improving compliance
The correct approach is risk-based but conservative, grounded in physical contact paths and failure modes rather than theoretical purity arguments.
Regulatory Expectations in Practice
Regulators expect facilities to:
- Clearly justify where clean steam is required and where it is not
- Demonstrate that clean steam quality supports its intended use
- Maintain control through design, operation, and lifecycle oversight
- Avoid informal substitutions or undocumented changes
There is no single prescriptive regulation for clean steam. Expectations are derived from general GMP principles and decades of inspection precedent. Facilities that treat clean steam as a first-class utility rarely struggle during inspections.
Relationship to Other Clean Utilities
Clean steam sits alongside other critical utilities:
- It is often generated from Purified Water or Water for Injection
- It supports sterilization and environmental control functions
- It interfaces directly with HVAC, process equipment, and sterilizers
Because of these interfaces, clean steam failures tend to cascade. That is why its applicability must be defined early and documented clearly.
Practical GMP Position
From a GMP perspective, clean steam systems shall be:
- Applied only where justified by intended use and risk
- Designed using conservative, proven engineering principles
- Operated within defined and controlled parameters
- Documented in a manner that supports lifecycle oversight
Excessive complexity increases the likelihood of failure. Clear intent and well-defined application boundaries support sustained control over the system lifecycle.
