Coating Equipment
Coating equipment is used in pharmaceutical manufacturing to apply a uniform layer of coating material onto tablets, pellets, or capsules. The coating process improves product stability, modifies drug release characteristics, enhances appearance, and can provide protection from moisture, light, or oxygen.
Pharmaceutical coating operations are typically performed using automated coating systems that combine controlled airflow, spray application, and mechanical mixing of the dosage forms. Proper equipment design ensures uniform coating thickness, efficient solvent evaporation, and minimal product damage during processing.
Coating equipment must provide precise control of process parameters including spray rate, inlet air temperature, exhaust airflow, and drum rotation speed. Because coating performance directly influences product quality attributes such as dissolution behavior and tablet appearance, coating equipment must be carefully designed, qualified, and validated.
1. Purpose of Pharmaceutical Coating
The primary purpose of pharmaceutical coating is to apply a controlled layer of polymer or functional coating material onto the dosage form surface. Coating operations may serve several functions including:
- Protection of the drug product from environmental conditions
- Masking unpleasant taste or odor
- Improving product appearance and patient acceptability
- Controlling drug release characteristics
- Providing identification through color or printing
Coating thickness and composition must be carefully controlled to ensure consistent product performance.
2. Types of Coating Equipment
Several types of coating equipment are used in pharmaceutical manufacturing depending on dosage form characteristics and process requirements.
2.1 Perforated Pan Coaters
Perforated pan coaters are the most widely used coating systems for tablets and capsules.
In this design, dosage forms are placed in a rotating perforated drum where they tumble continuously during coating. Heated air is drawn through the perforations in the drum while coating solution is sprayed onto the moving tablet bed. The combination of mechanical tumbling and controlled airflow promotes uniform coating distribution and rapid solvent evaporation.
The illustration below shows a typical perforated pan coating machine used in pharmaceutical tablet coating. Tablets tumble inside a rotating perforated drum while coating solution is sprayed onto the moving bed and heated process air flows through the drum to remove solvent and dry the coating layer.

2.2 Fluid Bed Coaters
Fluid bed coaters are commonly used for coating pellets, granules, and multiparticulate dosage forms. In these systems, particles are suspended in an upward flow of heated air while coating solution is sprayed onto the fluidized particles.Fluid bed coating configurations include:
- Top-spray coating
- Bottom-spray coating (Wurster process)
- Tangential spray coating
Fluid bed coating allows efficient coating of small particles and provides excellent coating uniformity.
The illustration below shows a fluid bed coating system configured for bottom-spray coating using the Wurster process. Particles circulate through a central coating zone where coating solution is sprayed upward while heated air fluidizes the particles and promotes rapid drying.

2.3 Conventional Coating Pans
Traditional coating pans consist of rotating drums without perforations. Heated air is supplied externally while coating solution is sprayed onto the tumbling tablets. Although conventional pans are less efficient than perforated systems, they are still used in some manufacturing environments for specific coating applications.
The illustration below shows a conventional pharmaceutical coating pan in which tablets tumble within a rotating solid drum while coating solution is sprayed onto the moving tablet bed. Heated air introduced from an external air handling system supports evaporation of the solvent and drying of the coating layer during rotation.

3. Coating Process Overview
The coating process involves several coordinated steps that occur within the coating equipment. Typical coating operations include:
- Loading of tablets or pellets into the coating chamber
- Preheating of the product bed
- Spraying of coating solution
- Drying through heated airflow
- Cooling and discharge of coated product
During coating, tablets or pellets tumble continuously within the coating chamber while spray nozzles apply the coating solution. Heated process air removes solvent from the coating solution, allowing the coating film to solidify on the product surface.
The illustration below shows the sequence of operations during pharmaceutical tablet coating, including product loading, preheating, coating spray application, solvent evaporation, and final cooling before discharge.

Uniform mixing of the dosage forms is essential to ensure even coating distribution across the batch.
4. Major Components of Coating Equipment
Pharmaceutical coating equipment consists of several mechanical and process systems that control coating application and drying.
4.1 Coating Drum or Product Chamber
The coating drum is the primary vessel where tablets or pellets are tumbled during coating.
The drum may be perforated to allow process air to pass through the product bed and improve drying efficiency.
4.2 Spray System
The spray system delivers coating solution onto the moving product bed. Spray systems typically include:
- Solution tanks
- Peristaltic or diaphragm pumps
- Spray guns or atomizing nozzles
- Air pressure control systems
The illustration below shows a typical coating spray nozzle where coating solution is atomized by compressed air to form fine droplets. Proper droplet size distribution is critical for achieving uniform coating across the tablet bed.
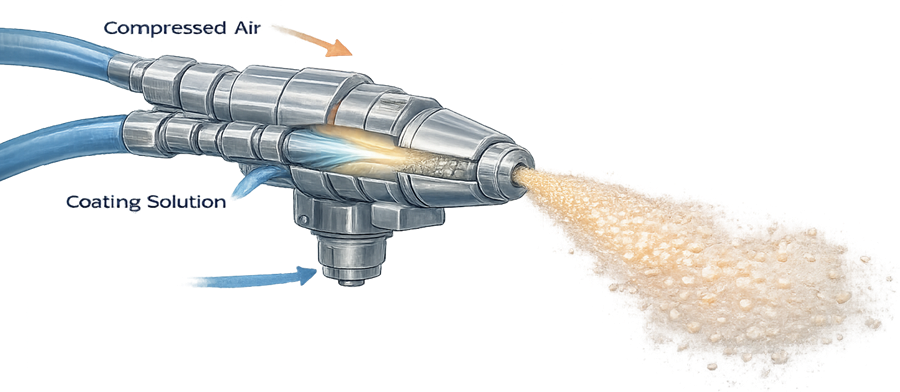
Spray droplet size and distribution significantly influence coating uniformity.
4.3 Air Handling System
The air handling system supplies conditioned process air required for solvent evaporation and drying during the coating process. The coating air handling system is specifically designed to deliver controlled airflow, temperature, and humidity to the coating chamber.
The primary function of the system is to provide heated, filtered air that passes through the tablet bed during coating. As coating solution is sprayed onto the tumbling tablets, the process air removes solvent through evaporation. The resulting moisture-laden air is then removed from the coating drum through the exhaust system. Important components of the coating air handling system typically include:
- Process air filters
- Inlet air heating system
- Air supply ducts to the coating drum
- Exhaust air ducts and exhaust blower
- Airflow and temperature control instrumentation
Precise control of inlet air temperature, airflow rate, and exhaust conditions is essential to maintain stable drying conditions and prevent coating defects such as sticking, rough surfaces, or incomplete film formation.
The illustration below shows the airflow pattern within a perforated coating drum. Heated inlet air enters the drum and passes through the tumbling tablet bed where solvent evaporation occurs. The humid exhaust air then exits the drum, carrying moisture and evaporated solvent away from the coating environment.

4.4 Exhaust and Solvent Recovery System
The exhaust system removes moisture and solvent vapors generated during the coating process. In some systems, solvent recovery equipment is used to capture organic solvents for safety and environmental compliance.
5. Critical Process Parameters
Successful tablet coating depends on precise control of several process parameters that govern coating application, solvent evaporation, and film formation. These parameters influence droplet formation, drying rate, tablet bed mixing, and coating uniformity. Improper control of any of these variables can disrupt the balance between spray deposition and drying, leading to coating defects. Key process parameters include:
Inlet Air Temperature
Inlet air temperature controls the drying capacity of the system. Air that is too cool may not provide sufficient energy for solvent evaporation, which can cause tablet surfaces to remain wet and promote sticking or picking. Excessively high inlet temperatures may cause rapid solvent evaporation at the droplet surface, producing rough coatings or defects such as orange peel.
Exhaust Air Temperature
Exhaust air temperature reflects the overall drying efficiency of the system and is often used as an indirect indicator of solvent evaporation. Low exhaust temperatures may indicate insufficient drying, while excessively high exhaust temperatures may indicate excessive drying or insufficient spray application.
Spray Rate
Spray rate determines how quickly coating solution is applied to the tablet bed. If the spray rate exceeds the drying capacity of the system, excess liquid accumulates on the tablet surface, leading to sticking, picking, or twinning. Conversely, spray rates that are too low may produce incomplete coating layers and poor film formation.
Atomization Air Pressure
Atomization pressure controls droplet size produced by the spray nozzle. Low atomization pressure generates larger droplets that can over-wet the tablet surface and cause localized coating defects. High atomization pressure produces very fine droplets that may dry prematurely before reaching the tablet surface, resulting in rough coatings or powdery film formation.
Drum Rotation Speed
Drum speed influences tablet bed mixing and coating distribution. Insufficient drum speed can result in poor mixing and uneven coating application, while excessively high speeds may increase mechanical stress and cause tablet chipping or coating damage.
Product Bed Temperature
The temperature of the tablet bed determines the rate at which solvent evaporates from the applied coating droplets. Bed temperatures that are too low may lead to incomplete drying and sticking, while excessively high bed temperatures can produce brittle coatings or surface roughness.
The interaction between these parameters determines the balance between coating application and drying. Deviations from optimal conditions can produce visible coating defects that indicate improper process control.
The illustration below shows examples of common tablet coating defects that may occur when coating process parameters are not properly controlled. These include sticking, where tablets adhere to each other due to excessive wetting; picking, where portions of the coating are removed from the tablet surface; peeling, where the coating film separates from the tablet core; rough surfaces or orange peel, caused by rapid solvent evaporation; twinning, where two tablets adhere together during coating; and uneven coating distribution, which results from inadequate mixing or improper spray conditions.
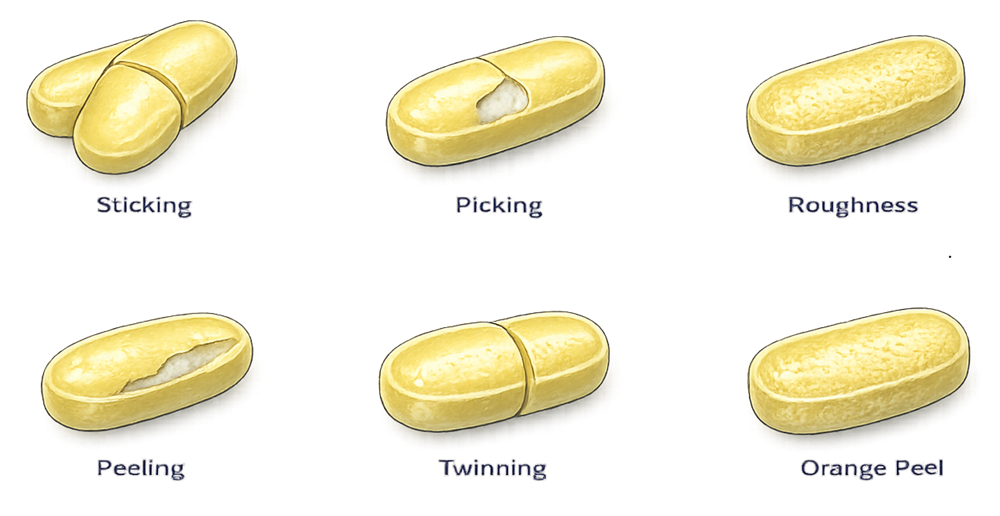
6. Qualification of Coating Equipment
Coating equipment must undergo formal qualification to demonstrate that the system is properly installed, operates within defined limits, and consistently produces coated products meeting quality specifications. Qualification typically includes:
- Design Qualification
- Installation Qualification
- Operational Qualification
- Performance Qualification
6.1 Design Qualification
Design Qualification verifies that the coating equipment meets the requirements defined in the User Requirements Specification. Design evaluation typically includes:
- Drum capacity and size
- Air handling system capacity
- Spray system configuration
- Control system capabilities
- Materials of construction and cleanability
6.2 Installation Qualification
Installation Qualification verifies that the equipment has been installed according to manufacturer specifications and engineering documentation. Typical IQ activities include:
- Verification of equipment identification and model number
- Verification of mechanical installation
- Verification of electrical connections
- Calibration of instrumentation
- Verification of documentation and manuals
6.3 Operational Qualification
Operational Qualification verifies that the coating equipment operates correctly across its defined operating ranges. Operational tests typically include:
- Drum rotation speed verification
- Airflow control testing
- Spray system operation
- Temperature control verification
- Alarm and safety system testing
These tests confirm that the coating system functions properly under controlled conditions.
6.4 Performance Qualification
Performance Qualification demonstrates that the coating process consistently produces coated dosage forms meeting predefined quality specifications. PQ studies typically evaluate:
- Coating weight gain
- Coating uniformity
- Tablet appearance
- Dissolution performance
- Batch-to-batch reproducibility
Multiple PQ batches are typically executed to demonstrate process stability.
6.4.1 Sampling Strategy
Sampling during Performance Qualification is designed to demonstrate that the coating process produces tablets with consistent coating weight and uniform film distribution throughout the batch. Because tablets continuously tumble and circulate within the coating drum, samples must represent material produced across the full duration of the coating run.
Sampling typically includes collecting coated tablets at predefined time intervals during the coating process. These samples allow evaluation of coating weight gain progression and verification that the coating layer develops uniformly as the process proceeds. Samples are usually withdrawn from the coating pan after the process has stabilized and then periodically throughout the run until the target coating weight is achieved.
At each sampling interval, a defined number of tablets are removed and weighed to determine coating weight gain relative to the uncoated tablet core weight. In addition to weight measurements, samples may also be visually inspected to evaluate coating appearance, surface uniformity, and the absence of coating defects.
Sampling intervals are typically defined based on process duration and coating rate, for example at the start of stable coating conditions and then at regular intervals throughout the coating cycle. This approach allows verification that coating deposition progresses in a controlled and predictable manner.
The illustration below shows a typical sampling strategy used during Performance Qualification of coating equipment. Tablet samples are collected at defined intervals during the coating process and evaluated to confirm consistent coating weight gain and coating uniformity across the batch.
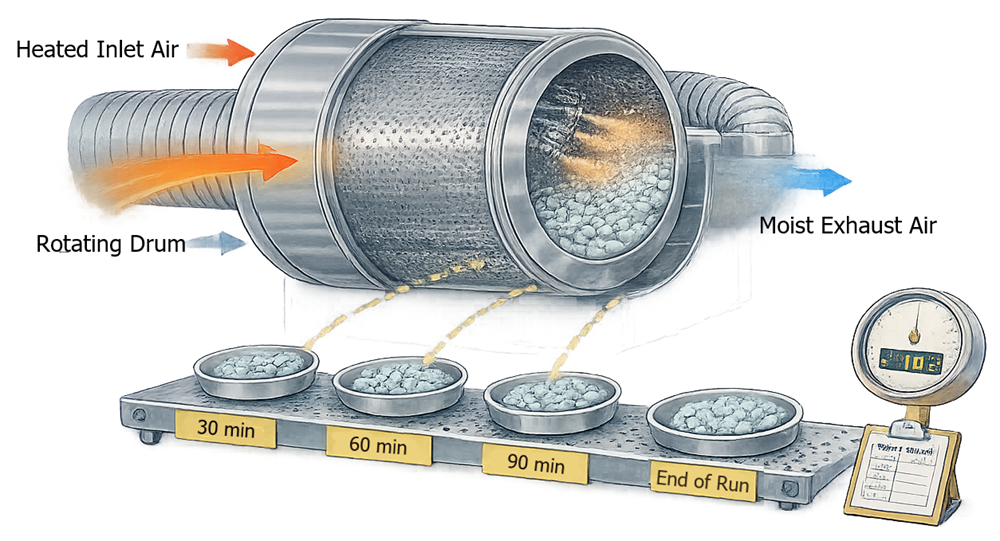
6.4.2 Statistical Evaluation
Statistical evaluation of PQ data is used to confirm that coating thickness and product quality remain consistent across the batch. Typical statistical evaluations include:
- Mean coating weight gain
- Standard deviation
- Relative standard deviation
- Trend analysis across sampling intervals
6.4.3 Acceptance Criteria
Acceptance criteria must be established before PQ execution. Typical acceptance criteria include:
- Coating weight gain within specification limits
- Uniform coating appearance
- Absence of defects such as picking, peeling, or roughness
- Acceptable dissolution performance
Successful completion of Performance Qualification confirms that the coating equipment consistently produces coated dosage forms meeting product quality specifications.
7. Integration with Solid Dosage Manufacturing
Coating equipment operates as part of the broader solid dosage manufacturing process. Tablets produced during compression or capsules produced during filling are transferred to the coating system where functional or protective coatings are applied.
After coating, products are typically transferred to downstream processes including:
- Inspection systems
- Metal detection
- Packaging operations
Proper integration between compression or filling equipment, coating systems, and packaging lines ensures continuous production and consistent product quality.
