Column Phases and Maintenance
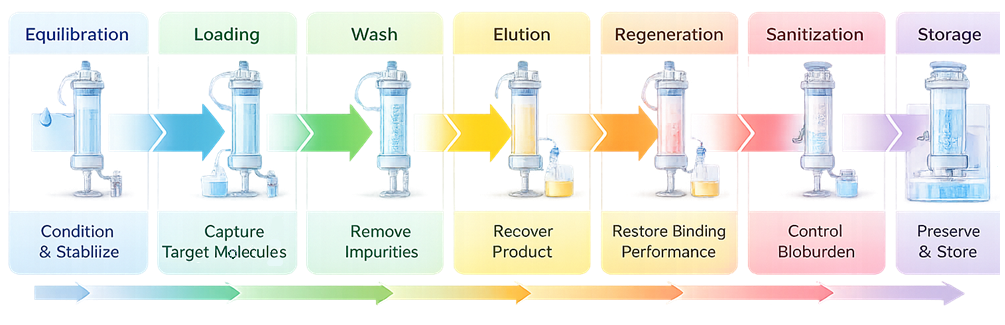
The diagram below illustrates the sequential operational phases of a chromatography column. Each phase performs a distinct functional role, from conditioning the resin and binding the target molecule to impurity removal, product recovery, and restoration of column performance. Although buffer chemistry varies by chromatography mode, this sequence reflects the standardized lifecycle of a production column.
1. Purpose and Scope
Chromatography columns are the primary separation element within a purification system. While the skid governs flow control and automation, the column contains the stationary phase media responsible for selective binding and resolution. This article addresses:
• Functional phases of column operation
• Media interaction principles
• Regeneration and sanitization
• Storage and lifecycle control
• Maintenance considerations affecting performance and validation
The focus is large-scale GMP production columns used in biopharmaceutical purification.
2. Column Functional Phases
Chromatography column operation follows defined, sequential phases designed to control molecular interaction between the mobile phase and the stationary phase. While buffer composition and operating limits depend on chromatography mode such as ion exchange, affinity, hydrophobic interaction, or mixed mode, the functional logic of each phase remains consistent.
Each phase has a specific purpose in conditioning the resin, capturing the target molecule, removing impurities, recovering product, and restoring column performance for reuse.
2.1 Equilibration
Equilibration prepares the column for product binding by establishing defined chemical and physical conditions within the packed bed. During storage, columns are typically held in preservative solutions that are incompatible with process binding conditions. Equilibration displaces the storage medium and saturates the resin with process buffer.
Functionally, equilibration:
• Sets the ionic strength and pH required for selective binding
• Establishes a stable conductivity baseline
• Allows the resin functional groups to adopt their operational charge state
• Stabilizes flow distribution and pressure profile
This phase ensures that resin binding sites are uniformly conditioned before exposure to feed. Inadequate equilibration may lead to inconsistent binding capacity or early breakthrough.
Equilibration volume is usually expressed in column volumes and must be validated to ensure complete buffer exchange and stable detector baselines.
2.2 Loading
Loading introduces the clarified process stream to the column under controlled flow and pressure. The target molecule interacts with functional ligands on the resin surface through defined mechanisms such as electrostatic attraction, affinity binding, or hydrophobic interaction.
During this phase:
• The target molecule binds selectively to the stationary phase
• Non-binding or weakly interacting impurities pass through the column
• UV or other inline detection monitors breakthrough behavior
Loading continues until a defined endpoint is reached, typically based on UV breakthrough or calculated dynamic binding capacity.
Critical functional aspects include:
• Mass transfer kinetics between bulk flow and pore structure
• Flow rate impact on residence time
• Resin saturation limits
• Bed height and diameter influence on capacity
If loading exceeds the dynamic binding capacity, the product begins to break through, reducing yield and potentially contaminating the flow-through fraction.
2.3 Wash
The wash phase improves product purity by removing weakly bound impurities while maintaining strong interaction between the target molecule and the resin.
Mechanistically, wash conditions are adjusted to:
• Increase stringency without disrupting target binding
• Remove nonspecific interactions
• Clear host cell proteins, DNA, or process contaminants
Control parameters may include moderate salt concentration shifts, pH adjustments, or addition of specific modifiers depending on resin chemistry.
Wash effectiveness is evaluated by monitoring UV and conductivity stability. Insufficient wash can compromise impurity clearance. Excessively aggressive wash conditions may partially elute the product, reducing recovery.
Wash design must balance purity improvement with yield retention.
2.4 Elution
Elution releases the bound product from the stationary phase by deliberately altering interaction forces between ligand and target molecule.
The mechanism depends on chromatography type:
• Ion exchange: increase ionic strength or shift pH to neutralize charge interactions
• Affinity: introduce competitive ligand or modify pH
• Hydrophobic interaction: reduce salt concentration
• Reverse phase: increase organic solvent concentration
Functionally, elution:
• Disrupts binding equilibrium
• Releases product into the mobile phase
• Generates a defined elution peak profile
Elution parameters directly affect:
• Peak symmetry
• Product concentration
• Fraction collection strategy
• Downstream processing efficiency
Gradient slope, flow rate, and buffer composition must be controlled to ensure reproducible elution behavior.
2.5 Regeneration
Regeneration removes tightly bound impurities and residual process contaminants that were not displaced during elution. Over time, foulants accumulate on the resin surface and within pores, reducing available binding capacity.
Regeneration typically involves:
• High salt solutions
• Extreme pH exposure
• Chaotropic agents
• Stripping solutions specific to resin chemistry
The objective is to restore the resin to a condition equivalent to pre-load functionality without damaging ligand integrity.
Regeneration effectiveness influences:
• Long-term capacity retention
• Pressure stability
• Consistency of subsequent cycles
Insufficient regeneration results in gradual performance decline.
2.6 Sanitization
Sanitization is distinct from regeneration. Its purpose is microbial control rather than impurity removal.
Repeated process cycles introduce potential bioburden from feed streams and environmental exposure. Periodic sanitization:
• Reduces microbial contamination
• Prevents biofilm formation
• Maintains column hygienic status
Sodium hydroxide is commonly used in ion exchange systems due to its microbial inactivation capability and resin compatibility.
Sanitization conditions must be validated to ensure:
• Microbial reduction effectiveness
• No degradation of ligand functionality
• Acceptable resin lifetime impact
Overexposure to sanitants accelerates resin aging.
2.7 Storage
When columns are idle, controlled storage conditions prevent microbial growth and chemical degradation.
Storage solutions typically:
• Contain ethanol or antimicrobial agents
• Maintain low pH where compatible
• Preserve ligand structure
Key controls include:
• Sealing integrity to prevent evaporation
• Temperature control
• Verification of preservative concentration
Improper storage may lead to:
• Microbial contamination
• Resin drying or cracking
• Ligand denaturation
• Reduced dynamic binding capacity
Storage strategy is part of overall lifecycle management and must align with reuse validation.
3. Column Performance Attributes
Column performance is defined by measurable hydraulic and chromatographic parameters that reflect both mechanical bed integrity and chemical functionality of the stationary phase. These metrics are monitored to evaluate performance consistency across cycles and support lifecycle control.
Backpressure
Backpressure is the hydraulic resistance across the packed bed at a defined flow rate and buffer condition.
What it indicates
• Bed packing density
• Resin swelling state
• Fouling or blockage
• Flow distribution uniformity
Phase association
• Established during equilibration as baseline
• May increase during loading due to fouling
• Should stabilize after effective regeneration
• May shift after repeated sanitization
Progressive pressure increase suggests fouling, compression, or channel restriction.
Asymmetry Factor
Asymmetry describes elution peak shape and symmetry. Ideal value is near 1.0.
What it indicates
• Uniform flow distribution
• Proper packing
• Absence of channeling
• Controlled mass transfer
The illustration below compares uniform flow distribution within a properly packed column to channel formation resulting from mechanical disturbance or bed compression. Uniform flow ensures consistent mass transfer and peak symmetry, while channeling creates preferential pathways that reduce efficiency, distort peak shape, and decrease separation performance.

Phase association
• Observed during elution
• Influenced by loading conditions
• Degrades if regeneration is incomplete
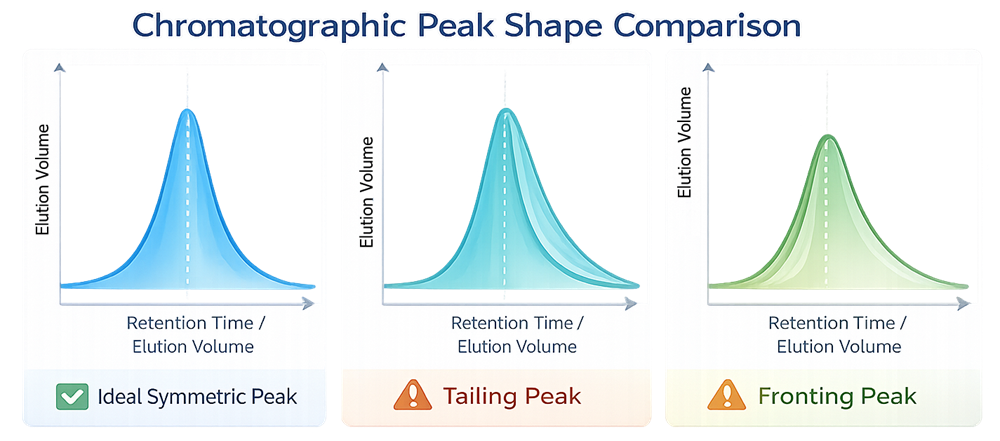
Peak tailing often signals fouling or ligand interaction changes. The graphic below illustrates typical chromatographic peak shapes. A symmetrical peak indicates uniform mass transfer and stable packing. Tailing peaks often reflect secondary interactions or fouling, while fronting peaks may indicate column overloading or packing irregularities. Peak morphology provides a rapid visual indicator of column health.
Theoretical Plates
Theoretical plates measure column efficiency and separation sharpness based on peak width and retention behavior.
What it indicates
• Bed uniformity
• Dispersion control
• Effective mass transfer
Phase association
• Calculated during elution
• Reduced by overloading during loading
• Declines with resin aging
Loss of efficiency suggests mechanical or structural degradation.
Binding Capacity
Binding capacity is the quantity of target molecule the resin can bind under defined dynamic conditions.
What it indicates
• Ligand density and functionality
• Resin surface activity
• Chemical integrity
Phase association
• Determined during loading
• Influenced by regeneration effectiveness
• Reduced by excessive sanitization
Declining capacity typically reflects ligand degradation or irreversible fouling.
Resolution
Resolution measures separation quality between adjacent peaks.
What it indicates
• Combined effect of efficiency and selectivity
• Integrity of resin chemistry
• Gradient control
Phase association
• Most evident during elution
• Affected by loading strategy
• Degrades if fouling alters selectivity
Reduced resolution often precedes measurable capacity loss.
Lifecycle Monitoring
Each parameter reflects a different aspect of column health:
Backpressure → mechanical condition
Asymmetry and plates → packing integrity and efficiency
Binding capacity → chemical functionality
Resolution → overall separation performance
Trending these metrics across cycles supports:
• Reuse justification
• Regeneration validation
• Early detection of degradation
• End-of-life determination
Integrated monitoring provides objective evidence that the column remains fit for intended use.
4. Mechanical Considerations
Column integrity depends on:
• Packing uniformity
• Bed height consistency
• Distributor performance
• Absence of channeling
• Seal integrity
Mechanical disturbances such as pressure spikes or rapid flow changes may cause:
• Bed compression
• Channel formation
• Reduced efficiency
Packing quality is foundational to reproducible separation. The image below shows a large-scale production chromatography column used in GMP biopharmaceutical manufacturing. The column contains the packed stationary phase media responsible for molecular separation, while inlet and outlet distributors ensure controlled flow distribution across the bed. Mechanical integrity, packing quality, and seal stability directly influence column efficiency and reproducibility.

5. Media Lifecycle and Reuse
Resin lifetime depends on:
• Cleaning exposure frequency
• Sanitization chemistry
• Process load
• Feed impurity profile
Manufacturers typically define maximum cycle limits, but actual lifecycle must be supported by:
• Performance trending
• Cleaning validation
• Periodic capacity testing
Reuse strategy must be scientifically justified.
6. Maintenance Activities
Column maintenance includes:
• Visual inspection
• Seal and gasket inspection
• Distributor cleaning
• Flow distribution verification
• Pressure limit verification
For prepacked disposable columns, maintenance is limited to:
• Integrity verification
• Lot traceability
• Storage condition control
7. Failure Modes
Common failure indicators include:
• Increased backpressure
• Peak tailing
• Reduced binding capacity
• Channeling
• Leakage
Root causes may involve:
• Fouling
• Resin degradation
• Mechanical compression
• Improper cleaning
Failure impact must be evaluated under change control.
8. Validation Considerations
From a qualification perspective, columns impact:
• Process performance qualification
• Cleaning validation
• Reuse validation
• Change management
Critical documentation typically includes:
• Resin lot traceability
• Packing records
• Performance trending data
• Cleaning cycle validation evidence
Columns are direct-impact components and require defined lifecycle oversight.
9. Conclusion
The chromatography column is the functional core of the purification process. Its operational phases, mechanical integrity, and maintenance strategy directly influence product purity, yield, and regulatory compliance. Effective lifecycle management requires structured monitoring, scientifically justified cleaning practices, and controlled reuse strategy within the validated process framework.
