Compressed Air Systems for GMP Manufacturing
1. Role of Compressed Air in GMP Manufacturing
Compressed air is one of the most widely used utilities in GMP-regulated facilities and one of the most frequently underestimated. It is routinely used to operate equipment, control valves, actuate instruments, and support manufacturing operations. In many facilities, compressed air also comes into indirect or direct contact with product-contact surfaces.
Compressed air is generated on site and conditioned through multiple mechanical and filtration stages. Each stage introduces potential failure modes that can affect air quality. As a result, compressed air is treated as a critical utility whose design must be appropriate for its intended use and risk to product quality.
Regulatory observations related to compressed air systems are common and typically trace back to design shortcomings rather than isolated operational failures.
2. Intended Use and Risk Categorization
Compressed air quality expectations increase with proximity to product and are defined by intended use rather than by a single system-wide specification.
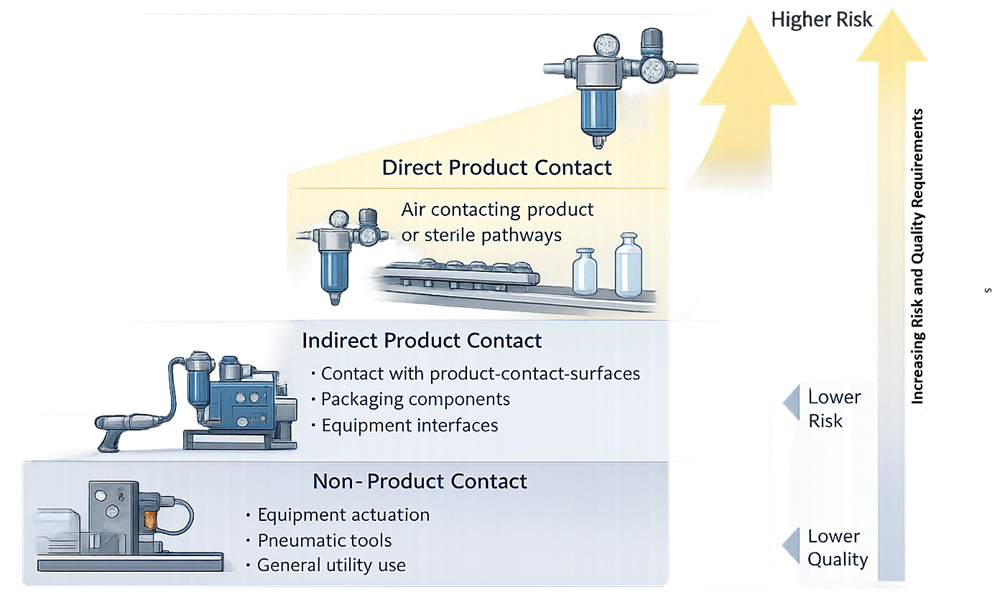
The design of a compressed air system must begin with a clear understanding of how the air is used. Intended use drives quality expectations, system complexity, and downstream controls.
Compressed air applications generally fall into three categories:
- Non-product contact
Air used for equipment operation, utilities, or general facility functions with no reasonable pathway to product or product-contact surfaces. - Indirect product contact
Air that does not contact product directly but may contact product-contact surfaces, packaging components, or critical equipment interfaces. - Direct product contact
Air that contacts product, product-contact surfaces, or sterile pathways during manufacturing operations.
Each category carries a different risk profile and requires different design controls. Applying a single compressed air quality target across all uses is neither necessary nor appropriate.
3. Compressed Air System Architecture
A typical GMP compressed air system consists of multiple conditioning stages designed to progressively reduce contamination and stabilize air quality before use.
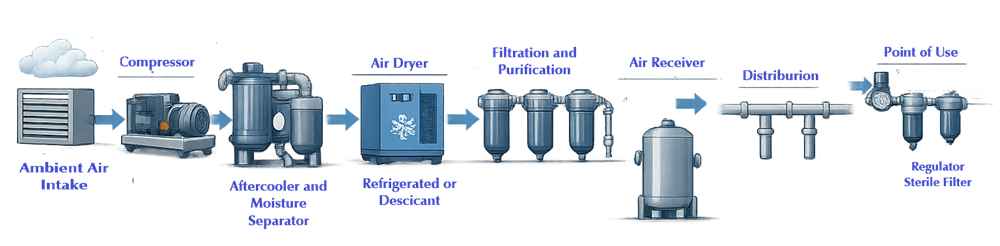
A typical GMP compressed air system consists of the following functional elements:
- Ambient air intake
- Compression equipment
- Drying systems
- Filtration and purification stages
- Storage and distribution
- Point-of-use controls
System architecture should be designed as an integrated whole. Upstream components reduce bulk contamination and load, while downstream controls provide final protection at the point of use. Redundancy, accessibility for maintenance, and system stability under varying demand conditions are key architectural considerations.
In practice, GMP compressed air systems consist of multiple interconnected components distributed across utility spaces and mechanical rooms, each representing a potential point of failure if not properly designed and maintained.

4. Quality Attributes and Design Expectations
Compressed air quality is defined by a small number of critical attributes. These attributes must be controlled to a level appropriate for intended use and system risk.
Particulates
Particulate contamination can originate from ambient intake air, compressor wear, corrosion, or distribution piping. Excessive particulates can damage equipment and contaminate product-contact surfaces.
International standards such as ISO 8573-1 are commonly used to classify particulate content in compressed air. In GMP environments, these classifications are applied as reference benchmarks and interpreted based on use and risk. Final filtration at the point of use remains the primary control for particulate risk.
Oil and Hydrocarbons
Oil contamination may be present as aerosols or vapors and can originate from compressor lubricants or intake air hydrocarbons.
ISO 8573-1 provides a structured approach to oil classification, including both aerosol and vapor components. Oil-free compressors reduce risk but do not eliminate the need for downstream filtration and adsorption. Claims of oil-free air without downstream controls are not considered adequate for GMP manufacturing.
Moisture and Dew Point
Moisture is a primary driver of corrosion, filter degradation, and microbial growth. Dew point control is therefore a fundamental design requirement.
ISO 8573-1 dew point classes are commonly referenced, but the required dew point is determined by system distribution, ambient conditions, and point-of-use risk. Excessively aggressive dew point targets are not inherently beneficial and may compromise system reliability.
Microbiological Considerations
Compressed air is not inherently sterile. ISO compressed air standards do not define microbiological limits.
Microbial risk is controlled through dryness, filtration, system integrity, and point-of-use sterile filtration where air contacts product or product-contact surfaces. Expectations for microbial control are driven by GMP principles and intended use rather than by ISO classification alone.
5. Design Considerations Driven by Risk
Several design decisions have a direct impact on compressed air quality and system robustness:
- Compressor selection
Oil-lubricated and oil-free compressors each have advantages and limitations. Selection should be based on risk, maintainability, and lifecycle considerations rather than marketing claims. - Drying technology
Refrigerated, desiccant, and membrane dryers provide different levels of moisture control and stability. Selection should consider required dew point, energy use, and system resilience. - Filtration strategy
Multi-stage filtration should be used to progressively remove contaminants and protect downstream components. Final filtration at point of use is essential for higher-risk applications. - Materials of construction
Distribution piping materials should be selected to minimize corrosion, particulate generation, and degradation over time. - Drainage and condensate management
Poor condensate management is a common source of system contamination and performance drift.
Design decisions should be documented and justified based on risk and intended use rather than defaulting to generic industry configurations.
6. Design and Lifecycle Boundary
This article addresses the design expectations for compressed air systems used in GMP manufacturing. Design decisions directly determine the scope and depth of qualification, monitoring, and requalification activities, which are addressed separately as part of gas utility lifecycle control.
