Depyrogenation Validation and Qualification
Depyrogenation validation demonstrates that a dry heat process consistently achieves the required reduction of endotoxin under defined operating conditions. Unlike sterility qualification, which evaluates microbial lethality, depyrogenation qualification verifies chemical destruction of endotoxin.
The objective is measurable and reproducible endotoxin reduction under worst-case conditions.
1. Validation Objective
The fundamental validation question is: Does the process achieve a defined log reduction of endotoxin across the validated load configuration?
Acceptance criteria are based on:
• Initial endotoxin challenge level
• Required minimum log reduction
• Worst-case load position
• Reproducibility across qualification runs
The process must demonstrate consistent performance at the coldest validated location within the load.
2. Regulatory Framework
Depyrogenation processes are supported by:
• 21 CFR 211 — process validation and equipment qualification
• USP <1228> Depyrogenation — depyrogenation principles
• USP <85> Bacterial Endotoxins Test — endotoxin testing methodology
Regulations do not prescribe cycle parameters. They require scientific validation and documented evidence of control.
3. Qualification Lifecycle
Depyrogenation qualification follows structured lifecycle phases:
Installation Qualification (IQ)
• Equipment installation verification
• Utilities confirmation
• Instrument calibration
• HEPA filter certification
Operational Qualification (OQ)
• Empty chamber temperature mapping
• Airflow verification
• Alarm and interlock testing
• Setpoint accuracy verification
Performance Qualification (PQ)
• Endotoxin challenge studies
• Worst-case load configuration
• Replicate qualification runs
• Verification of minimum log reduction
PQ is the critical phase for demonstrating endotoxin destruction capability.
4. Endotoxin Challenge Methodology
Depyrogenation validation uses standardized endotoxin preparations applied to representative materials. Typical approach:
• Known endotoxin concentration applied to carriers
• Drying under controlled conditions
• Placement at worst-case locations
• Exposure to validated cycle
• Post-process recovery and quantification
Worst-case locations typically include:
• Cold spots identified during mapping
• Areas with restricted airflow
• Maximum load density zones
Recovery efficiency must be evaluated to ensure accurate log reduction calculation.
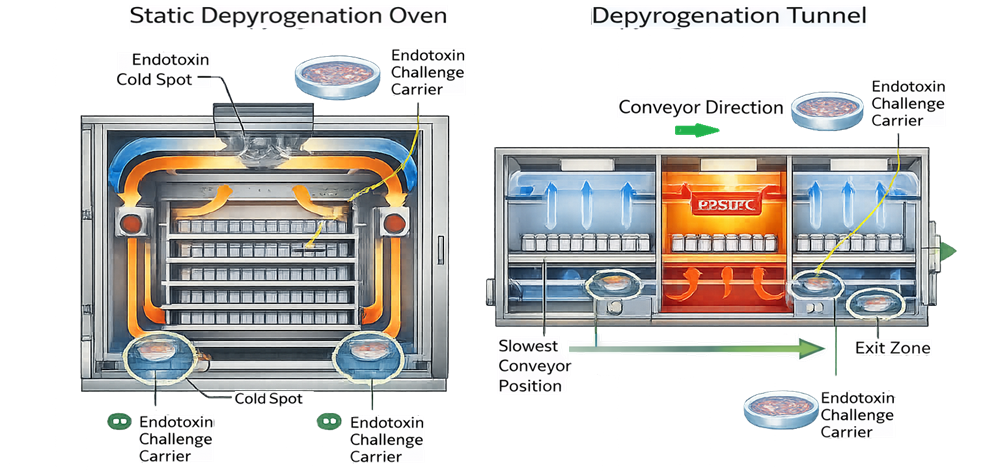
The schematic below illustrates the logic used for placement of endotoxin challenge carriers during depyrogenation performance qualification. Endotoxin is intentionally applied to representative carrier materials, dried under controlled conditions, and positioned at thermally worst-case locations identified during temperature mapping. In static ovens, carriers are placed at validated cold spots, typically at lower shelf corners, near door interfaces, and in high load density regions where airflow is restricted and heat penetration is least efficient. In continuous tunnel systems, carriers are positioned within the high-temperature dwell zone at the slowest validated conveyor speed and at edge positions most susceptible to reduced thermal exposure. Placement is not random; it is driven by prior mapping data and airflow analysis to ensure that validation challenges the process at its least favorable conditions.
5. Log Reduction Requirement
Depyrogenation performance is typically expressed as: Minimum validated log reduction of endotoxin.
The required log reduction must be:
• Scientifically justified
• Aligned with product risk
• Consistent with compendial expectations
The validated process must consistently achieve this reduction at the coldest qualified point.
The diagram below illustrates the logarithmic reduction principle underlying depyrogenation validation. A defined initial endotoxin challenge is applied to representative carriers and exposed to the validated thermal cycle. Endotoxin destruction follows a logarithmic decline, meaning each log reduction represents a tenfold decrease in endotoxin concentration. Validation demonstrates that the process consistently achieves a minimum required log reduction under worst-case conditions.
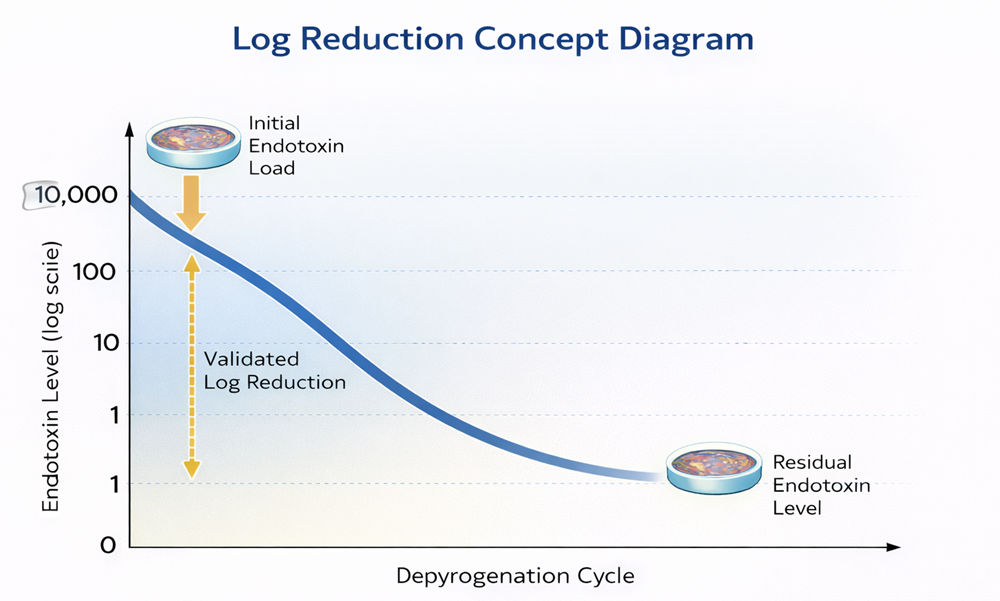
In practical terms, a 3-log reduction represents a 1,000-fold decrease in endotoxin level, while a 6-log reduction represents a 1,000,000-fold decrease. The required reduction is defined during process development and must be consistently demonstrated at the coldest validated location. Log reduction capability, not absolute starting concentration, is the validated performance metric.
6. Critical Parameters
Depyrogenation capability is driven by:
• Chamber temperature
• Exposure time
• Air velocity
• Conveyor speed (for tunnels)
• Load density
During validation, acceptable operating ranges must be defined. Process capability must remain within these ranges.
7. Requalification Triggers
Requalification may be required following:
• Heating element replacement
• Airflow system modification
• Control system changes
• Load configuration modification
• Repeated deviations
• Preventive maintenance affecting thermal performance
Change evaluation must assess potential impact on endotoxin reduction capability.
8. Documentation Requirements
Validation documentation must include:
• Defined acceptance criteria
• Mapping data
• Endotoxin recovery calculations
• Raw data traceability
• Deviations and investigations
• Approved validation report
Data integrity controls must comply with electronic record requirements where applicable.
Core Validation Principle
Depyrogenation validation demonstrates consistent endotoxin reduction under worst-case conditions, supported by thermal mapping, controlled parameters, and documented evidence.
The validated state must be maintained through lifecycle oversight, defined operating ranges, and structured change management.
