Dispensing and Material Handling Equipment
Material dispensing and handling represent the first controlled mechanical stage in solid dosage manufacturing. At this point the process transitions from warehouse-controlled inventory to manufacturing-controlled materials. Accuracy of weighing, integrity of material identification, and prevention of cross-contamination must be established before the material enters downstream processing steps such as milling, blending, or granulation.
Although dispensing operations may appear operational rather than process-critical, errors introduced at this stage propagate throughout the entire batch. Incorrect weights, material mix-ups, or uncontrolled powder release can compromise blend uniformity, potency, and regulatory compliance. For this reason dispensing systems must be designed with both operational reliability and qualification requirements in mind.
Dispensing and material handling equipment is typically installed in dedicated dispensing suites or weigh rooms designed to control dust generation, manage material flow, prevent cross-contamination, and ensure safe operator interaction with powders. These areas commonly include raw material staging zones, weighing booths with integrated dust containment, operator workstations, and transfer interfaces that allow dispensed materials to move to downstream processing equipment. The equipment used at this stage may range from simple weighing stations to highly contained automated transfer systems used for potent compounds. Proper layout and integration of these elements support accurate weighing operations while minimizing the risk of powder release, material mix-ups, and contamination of adjacent manufacturing areas.
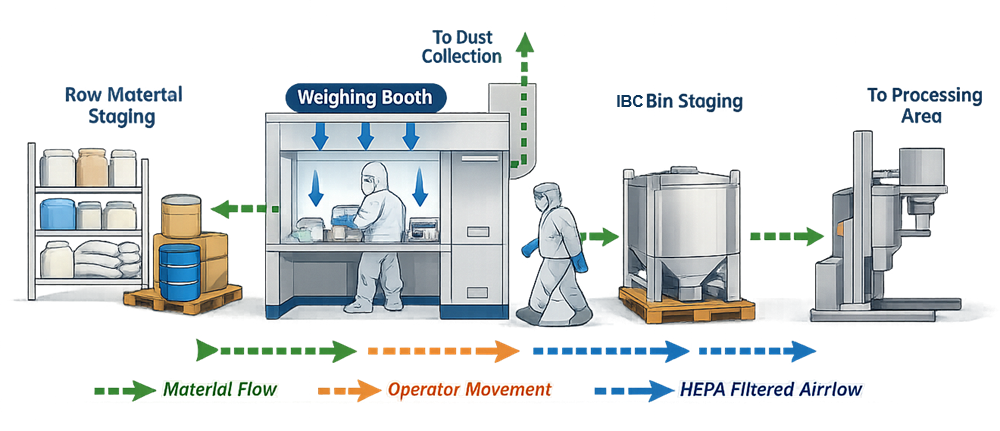
1. Equipment Types
Several categories of equipment are commonly used to support material dispensing and handling in solid dosage manufacturing.
1.1 Weighing and Dispensing Booths
Weighing booths provide a controlled environment for the accurate measurement of raw materials. These enclosures typically incorporate laminar airflow or negative pressure containment to capture airborne powder during dispensing operations. High-efficiency particulate air filtration is frequently used to prevent powder escape into surrounding manufacturing areas.
Dispensing booths often include integrated dust extraction systems and airflow monitoring to ensure containment performance remains within defined limits. The design must allow operators to safely access materials while preventing powder migration into adjacent areas.
Weighing and dispensing booths provide a controlled containment environment for measuring raw materials. These enclosures use directional airflow and filtration systems to capture airborne powder generated during dispensing operations. By maintaining controlled airflow patterns and dust extraction, dispensing booths help prevent powder escape into surrounding manufacturing areas while protecting both product quality and operator safety.

1.2 Intermediate Bulk Containers (IBC) and Material Bins
Intermediate bulk containers are widely used for bulk material storage and transport between processing steps. These stainless steel containers allow powders to be transferred from dispensing areas to blending or granulation equipment without direct operator contact.
IBC systems support closed transfer of materials, minimizing dust generation and reducing the risk of cross-contamination. Proper bin design must support smooth powder discharge, minimize hold-up areas, and allow effective cleaning between batches.
Intermediate bulk containers are commonly used for transporting powders between manufacturing steps without direct operator contact. These stainless steel containers allow materials dispensed in weigh rooms to be transferred safely to blending or granulation equipment. Proper bin design ensures complete discharge of materials while minimizing powder retention or contamination risk.

1.3 Bin Lifters and Transfer Interfaces
Bin lifters are mechanical systems designed to elevate and position IBC containers above downstream equipment such as blenders or granulators. These systems provide controlled discharge of material while reducing manual lifting and improving operator safety.
The interface between bins and processing equipment must maintain containment and prevent material loss. Mechanical locking systems and docking mechanisms ensure correct positioning and prevent accidental release during transfer operations.
Bin lifters allow heavy bulk containers to be elevated and positioned above downstream processing equipment. These systems provide controlled lifting and secure docking of intermediate bulk containers, allowing powders to discharge safely into blenders or granulators. Mechanical locking mechanisms and precise positioning controls help ensure stable operation and prevent accidental material release during transfer

1.4 Vacuum Transfer Systems
Vacuum transfer systems are frequently used to transport powders between equipment units without manual handling. These systems use controlled negative pressure to move powders through sealed piping or flexible hoses.
The use of vacuum transfer reduces operator exposure and improves process cleanliness. However, system design must prevent material segregation during transport and avoid accumulation of residual powder in transfer lines.
Vacuum transfer systems provide a contained method for transporting powders between equipment units. These systems use controlled negative pressure to draw powders through sealed transfer lines, reducing manual handling and minimizing operator exposure to airborne particles. Proper design of vacuum transfer systems helps maintain clean manufacturing environments while ensuring reliable movement of materials.

1.5 Pneumatic Conveying Systems
Pneumatic conveying systems transport powders through enclosed pipelines using controlled airflow generated by a blower or vacuum pump. The airflow creates a pressure differential that moves air through the pipeline, entraining powder particles introduced at the feed point and carrying them through the conveying line toward the receiving vessel. These systems allow materials to be transferred over long distances within manufacturing facilities while maintaining closed containment and minimizing manual handling.
At the receiving point, the powder is separated from the air stream through gravity settling, filtration, or cyclonic separation before discharging into a hopper or downstream process equipment. Proper system design requires careful control of airflow velocity and pipeline geometry to keep particles suspended during transport while preventing segregation, degradation, or excessive accumulation in transfer lines. Powder characteristics such as particle size, density, and flow properties must also be considered to ensure consistent and reliable material movement.

1.6 Split Butterfly Valves and Contained Transfer Devices
Contained transfer systems are used when highly potent or hazardous compounds are handled. Split butterfly valves allow sealed connection between containers and processing equipment, preventing powder release during material transfer.
These systems are widely used in facilities that manufacture potent active pharmaceutical ingredients or highly active drug products. Proper mechanical alignment and seal integrity are essential to maintain containment performance.
Split butterfly valves are commonly used when handling potent or hazardous pharmaceutical powders. These devices allow sealed connection between containers and processing equipment, enabling materials to be transferred without releasing powder into the environment. Proper alignment and sealing between the active and passive valve halves are critical for maintaining containment performance.

1.7 Pre-Processing Sieving Equipment
Sieving equipment is often incorporated immediately after dispensing to remove agglomerates or foreign material from powders before blending. This step ensures consistent particle distribution and prevents large particles from affecting blend homogeneity.
Sieving systems must be designed to prevent powder leakage and allow easy inspection and cleaning.
Sieving equipment is frequently used immediately after dispensing to remove agglomerates or foreign particles from powders before blending. Vibratory sieves ensure consistent particle distribution and help prevent large particles from affecting downstream mixing or compression performance. These systems must be designed to prevent powder leakage and allow effective cleaning between batches.

2. Design Considerations
Dispensing and material handling equipment must support reliable operation while preventing contamination and material loss. Several key design factors influence equipment selection and layout.
2.1 Containment and Operator Protection
Powder handling operations generate airborne particulates that may pose both product contamination and occupational exposure risks. Equipment must therefore provide adequate containment through enclosed systems, airflow control, or dedicated dust collection. Facilities handling potent compounds may require specialized containment technologies capable of maintaining extremely low exposure limits.
Airflow control is a critical component of dispensing booth containment performance. Directional airflow introduced through HEPA filtration systems moves downward across the work surface and captures airborne powder at the rear extraction zone. This airflow pattern prevents dust from escaping into the surrounding manufacturing environment while protecting operators from exposure.

2.2 Cross-Contamination Control
Cross-contamination is a primary regulatory concern in solid dosage manufacturing. Equipment surfaces must be smooth, cleanable, and free of crevices where residual powder could accumulate. Transfer systems must be designed to prevent unintended product contact with surrounding equipment or facility surfaces.
Closed transfer systems significantly reduce cross-contamination risk.
2.3 Dust Management
Powder release during dispensing and transfer operations can compromise both product quality and facility cleanliness. Integrated dust collection systems capture airborne particles and maintain clean operating conditions.
Dust collectors must be properly sized and connected to dispensing booths or transfer equipment to maintain consistent airflow and capture efficiency.
2.4 Ergonomics and Operator Safety
Manual powder handling can create ergonomic challenges. Equipment must be designed to reduce operator strain, minimize lifting requirements, and provide safe access to materials.
Proper workstation height, bin lifting systems, and mechanical handling devices improve both safety and operational efficiency.
2.5 Hygienic Design and Cleanability
All product contact surfaces must be constructed from materials compatible with pharmaceutical cleaning and sanitation procedures. Stainless steel is commonly used because of its corrosion resistance and cleanability.
Equipment should minimize dead legs, crevices, and horizontal surfaces where powder may accumulate.
2.6 Integration with Downstream Equipment
Material handling equipment must interface reliably with downstream processing equipment. Transfer connections must align with blender charging ports, granulation feed systems, or compression hoppers without introducing contamination risk or material loss.
Proper mechanical alignment ensures consistent transfer performance.
3. Utility and Infrastructure Interfaces
Dispensing and material handling equipment relies on several facility utilities and supporting infrastructure systems. Proper integration of these services ensures reliable equipment performance, effective containment, and safe operator interaction with powders.
3.1 Dust Collection Systems
Dust collection systems capture airborne particles generated during dispensing, sieving, and material transfer operations. These systems typically connect directly to weighing booths, sieving stations, and transfer equipment. The system must maintain sufficient airflow to prevent powder escape while avoiding excessive turbulence that could disturb weighing accuracy. Filters and collectors must be sized appropriately for the expected powder load and designed to allow safe removal of collected dust.
3.2 Compressed Air Systems
Compressed air is commonly used for pneumatic conveying systems, valve actuation, and certain automated transfer devices. The air supply must meet pharmaceutical quality standards to prevent contamination of materials. In most facilities, compressed air used in material handling must be dry, oil-free, and filtered to remove particulates and condensate.
3.3 Electrical Power Supply
Electrical power supports weighing scales, bin lifting equipment, pneumatic control systems, transfer system motors, and dust collection units. Electrical installations must comply with facility safety standards, and equipment handling combustible powders may require explosion-rated electrical components.
3.4 Equipment Grounding and Static Control
Handling dry powders can generate electrostatic charge, particularly during pneumatic transfer or sieving operations. Equipment grounding systems must be installed to prevent static discharge that could ignite combustible dust. Grounding connections are typically required for containers, transfer lines, and dispensing stations.
3.5 Facility Layout and Room Design
Dispensing areas must be designed to support controlled material flow and contamination control. Dedicated dispensing rooms or suites are commonly used to isolate powder handling operations from other manufacturing areas. Layout considerations include personnel movement pathways, material staging areas, airflow patterns, and containment of airborne particles.
Proper integration of these utilities and infrastructure systems ensures that dispensing and material handling equipment operates safely, maintains containment performance, and supports consistent pharmaceutical manufacturing conditions.
4. Qualification Considerations
Qualification of dispensing and material handling equipment verifies that the systems are properly installed, operate within defined performance limits, and support controlled pharmaceutical manufacturing operations. Because these systems represent the first stage of material processing within manufacturing, qualification must confirm accurate weighing, reliable transfer, effective containment, and the ability to maintain hygienic conditions.
The qualification strategy typically includes installation verification, operational testing, containment verification, and cleaning capability assessment.
4.1 Installation Qualification
Installation Qualification confirms that dispensing and material handling equipment has been installed in accordance with approved engineering specifications, manufacturer documentation, and facility design requirements.
- Installation verification normally includes confirmation of equipment identification, model numbers, and serial numbers. Materials of construction must be verified against design documentation to ensure compatibility with pharmaceutical cleaning procedures and product contact requirements.
- Utilities required for equipment operation must also be verified. These may include electrical power connections, compressed air supplies, dust collection connections, and grounding systems for static discharge control. Utility connections must be correctly installed and capable of supporting normal equipment operation.
- Mechanical installation must be examined to confirm that equipment is properly anchored, aligned, and accessible for operation and maintenance. For systems such as bin lifters and transfer stations, docking interfaces must align correctly with upstream containers and downstream processing equipment.
- Safety features must also be verified during installation qualification. These may include emergency stop functions, mechanical guards, interlocks, and containment enclosures intended to protect operators and prevent accidental release of powders.
- Documentation generated during installation qualification establishes a verified baseline configuration for the equipment.
4.2 Operational Qualification
Operational Qualification demonstrates that dispensing and material handling equipment functions as intended throughout its defined operating range. Testing performed during this phase focuses on functional performance of weighing systems, mechanical transfer devices, and control interfaces.
- Weighing systems must demonstrate measurement accuracy within defined tolerances. Load cells, balances, and scale platforms must be calibrated and tested across their operating range to confirm that measurement accuracy remains within acceptable limits under expected operating conditions.
- Operational testing of material transfer equipment verifies proper functioning of vacuum transfer systems, pneumatic conveying lines, and gravity discharge systems. Testing must confirm that materials can be transferred reliably without significant loss, segregation, or blockages.
- Mechanical equipment such as bin lifters and docking systems must be evaluated to confirm stable lifting performance, secure container engagement, and controlled discharge of materials. Movement limits, positioning accuracy, and interlock functionality must be verified.
- Operational qualification also verifies correct functioning of equipment controls, including alarms, status indicators, and operator interface features.
4.3 Containment and Dust Control Verification
Powder containment is a critical performance attribute for dispensing and material handling systems.
- Qualification must verify that containment systems effectively control airborne particulate release during dispensing and transfer operations.
- Airflow testing may be performed to confirm that weighing booths maintain appropriate air velocity and directional airflow patterns. Smoke visualization studies are often used to demonstrate proper airflow containment and capture of airborne particles.
- Dust collection system performance must also be verified. Testing may include confirmation of airflow rates, filter installation integrity, and proper operation of dust extraction connections to dispensing booths or transfer equipment.
- In facilities handling potent compounds, containment performance testing may also include surrogate powder studies or particulate monitoring to confirm that operator exposure levels remain within defined occupational limits.
4.4 Cleaning Verification
Material handling equipment must support effective removal of product residues between batches in order to prevent cross-contamination.
- Cleaning verification confirms that equipment surfaces can be adequately cleaned using approved procedures.
- Cleaning studies typically evaluate equipment surfaces that may accumulate powder during normal operation. These include transfer lines, bin discharge valves, docking interfaces, and internal surfaces of containers or transfer equipment.
- Cleaning verification may involve visual inspection, surface sampling, or analytical residue testing depending on facility validation practices. Particular attention must be given to areas where powder hold-up could occur, such as crevices, seals, or horizontal surfaces.
- Equipment design must allow access to these areas for cleaning and inspection. Demonstration of effective cleanability during qualification supports subsequent cleaning validation activities performed during manufacturing operations.
Comprehensive qualification of dispensing and material handling equipment ensures that materials entering the manufacturing process are accurately weighed, safely transferred, and handled in a manner that maintains product integrity and regulatory compliance. These systems establish the first controlled mechanical interface between raw materials and pharmaceutical processing equipment, making their reliable performance essential to the overall manufacturing lifecycle.
5. Common Risk Points
Dispensing and material handling operations involve direct interaction between raw materials, operators, and mechanical transfer systems. Because these activities occur before materials enter controlled processing equipment, failures introduced at this stage can affect the entire batch and may remain undetected until later manufacturing stages or even finished product testing. Identification and control of common risk points are therefore essential to maintain reliable dispensing operations and prevent errors that could compromise product quality. Effective risk control relies on proper equipment design, environmental containment, clearly defined operator procedures, and qualification testing that verifies consistent system performance.
Two common risks encountered during powder handling are particle segregation and material hold-up. These phenomena can affect blend uniformity, batch consistency, and contamination control if not properly addressed in equipment design. Illustration below demonstrates these two mechanisms.
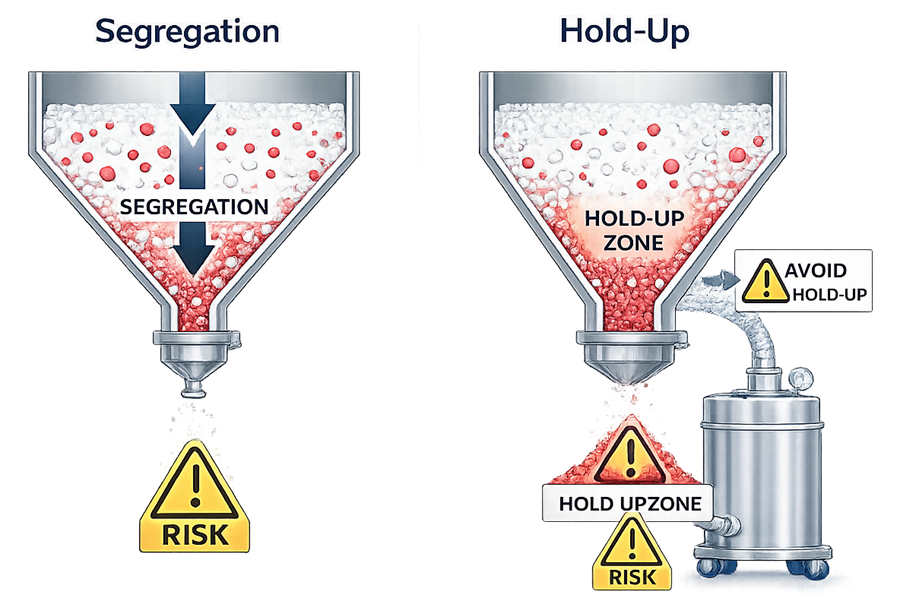
As shown in the illustration, segregation may occur during hopper discharge when powders composed of particles with different sizes, densities, or flow characteristics separate as they move through the discharge channel. Larger or denser particles may migrate differently than smaller particles, resulting in non-uniform composition of the material leaving the hopper.
The illustration also shows material hold-up, where powder becomes trapped within equipment geometry such as hopper corners, valves, elbows, or transfer lines. Retained material may remain in the system after discharge and later enter subsequent batches, creating risks of cross-contamination or inconsistent material transfer.
Effective powder handling system design therefore focuses on minimizing particle segregation and eliminating equipment dead zones to ensure consistent material movement and complete discharge.
5.1 Weighing Errors
Accurate weighing of raw materials is fundamental to batch formulation. Errors in weighing can result in incorrect ingredient ratios, which may affect potency, stability, dissolution, or other critical quality attributes of the final product.
Weighing errors may occur due to scale miscalibration, drift in load cell performance, or environmental factors such as vibration, airflow disturbance, or static charge affecting scale readings. Operator-related errors can also occur during manual dispensing, particularly when multiple containers or partial quantities are involved.
Control measures typically include regular calibration of balances, environmental control within dispensing rooms, use of electronic batch record verification, and procedural checks requiring independent verification of critical weighings.
5.2 Material Misidentification
Handling multiple raw materials within the same dispensing area introduces the risk of material mix-ups. If containers are improperly labeled, staged incorrectly, or returned to the wrong location, incorrect ingredients may be dispensed into a batch.
Material misidentification is particularly critical in pharmaceutical manufacturing because substitution of even a single component may significantly alter the safety or effectiveness of the product.
Risk mitigation typically involves barcode-based material verification, strict labeling controls, segregated staging areas for materials awaiting dispensing, and procedural requirements for material identity confirmation prior to weighing.
5.3 Powder Retention and Equipment Hold-Up
Powders handled during dispensing and transfer operations may accumulate within equipment surfaces, transfer lines, valves, or container discharge points. This retained material may subsequently dislodge during later operations, introducing unintended material into another batch.
Powder retention is commonly observed in poorly designed transfer lines, areas with horizontal surfaces, dead legs within piping, or complex valve assemblies. Accumulated material may also create difficulties during cleaning operations.
Equipment design should minimize areas where powder can accumulate, and transfer systems should allow complete discharge of materials during normal operation.
5.4 Segregation During Transfer
Powder segregation can occur when materials with different particle sizes, densities, or flow characteristics are transported through pneumatic conveying systems or long transfer lines. During transfer, lighter or smaller particles may separate from larger particles, resulting in non-uniform composition of the transferred material.
Segregation at the dispensing stage may compromise downstream blend uniformity, particularly when powders are transferred directly to blending equipment or intermediate containers. Equipment design and transfer velocities must therefore be carefully controlled to minimize conditions that promote particle separation.
5.5 Dust Release and Environmental Contamination
Dispensing and material transfer operations frequently generate airborne powder. If containment systems are inadequate or airflow control is poorly designed, powder may escape into surrounding manufacturing areas.
Airborne dust may contaminate other materials staged within the room, deposit on equipment surfaces, or create occupational exposure risks for operators. In facilities producing multiple products, uncontrolled dust release may also create cross-contamination concerns.
Effective dust capture systems, properly designed dispensing booths, and controlled airflow patterns are necessary to maintain clean operating conditions.
5.6 Operator Handling Errors
Manual material handling remains a significant contributor to risk during dispensing operations. Improper handling of containers, incorrect sequencing of dispensing steps, or incomplete transfer of materials from containers to receiving equipment may introduce variability in the process.
Operator fatigue, complex batch formulations, and poorly organized dispensing workstations can increase the likelihood of such errors.
Training programs, ergonomic workstation design, and clearly defined dispensing procedures are essential to minimize operator-related risks.
Dispensing and material handling equipment establishes the initial mechanical control of pharmaceutical materials within the manufacturing environment. Proper design, containment, and qualification of these systems ensures that materials enter downstream processing stages under controlled and verified conditions, providing a reliable foundation for all subsequent solid dosage manufacturing operations.
