Dry Heat Qualification
Dry heat qualification demonstrates that a dry heat sterilization system consistently achieves the required level of microbial lethality under defined and controlled operating conditions. Qualification must establish:
- Uniform temperature distribution
- Control accuracy and stability
- Worst-case load effectiveness
- Reproducibility of defined cycle parameters
- Continued suitability for intended use
Dry heat systems present elevated risk for temperature non-uniformity. Cold spots represent the primary technical hazard and must be identified and justified through documented testing.
Qualification Lifecycle Framework
Dry heat qualification follows a structured lifecycle approach consistent with regulatory expectations and industry standards.
1. Design Review
The design phase confirms that the system is suitable for its intended sterilization purpose. Key elements include:
- Chamber volume and configuration
- Heating method and temperature range capability
- Air circulation design
- Control system architecture
- Alarm functionality
- Data recording capability
Design deficiencies cannot be corrected during OQ. They must be addressed before qualification begins.
2. Installation Qualification (IQ)
Installation Qualification verifies that the system is installed in accordance with approved specifications. Typical IQ elements:
- Equipment identification and documentation
- Utility connections verification
- Instrument calibration status
- Control panel configuration
- Sensor installation verification
- HEPA filter installation and integrity testing for tunnel systems
IQ confirms installation correctness. It does not evaluate performance.
3. Operational Qualification (OQ)
Operational Qualification verifies that the system operates within predefined limits under controlled conditions. Core OQ activities include:
- Empty chamber temperature mapping
- Heat-up time verification
- Setpoint accuracy verification
- Control stability assessment
- Alarm and interlock challenge testing
- Sensor accuracy confirmation
Mapping density must be sufficient to detect localized cold spots. Sensor placement must reflect chamber geometry and airflow characteristics.
Acceptance criteria must define allowable temperature variation across the chamber.
4. Performance Qualification (PQ)
Performance Qualification demonstrates that the system achieves required lethality under actual operating conditions. Key PQ components:
- Loaded chamber heat distribution studies
- Worst-case load configuration definition
- Biological indicator placement at validated cold spots
- Reproducibility across consecutive cycles
Worst-case load must be justified scientifically. Convenience-based load selection is not acceptable.

Equipment-Specific Qualification Considerations

Static Hot Air Ovens
Primary Risk:
- Natural convection stratification
- Localized cold spots near chamber door or corners
Qualification Emphasis:
- Increased sensor density during mapping
- Shelf-level temperature variation
- Door seal integrity
- Recovery time after door opening
These systems are simple but prone to spatial variation.
Forced-Air Circulation Ovens
Primary Risk:
- Airflow imbalance
- Fan failure
- Channeling effects
Qualification Emphasis:
- Verification of airflow pattern
- Uniformity improvement validation
- Fan interlock testing
- Alarm verification for airflow disruption
Air circulation reduces stratification but introduces mechanical dependency.
Continuous Dry Heat Tunnels
Primary Risk:
- Zone transition variability
- Conveyor speed influence
- Airflow pressure cascade failure
- Sterile boundary compromise
Qualification Emphasis:
- Multi-zone temperature mapping
- Conveyor speed validation
- Worst-case vial placement
- HEPA integrity verification
- Cooling zone contamination risk assessment
Tunnel systems require integrated system qualification, not chamber-only evaluation.
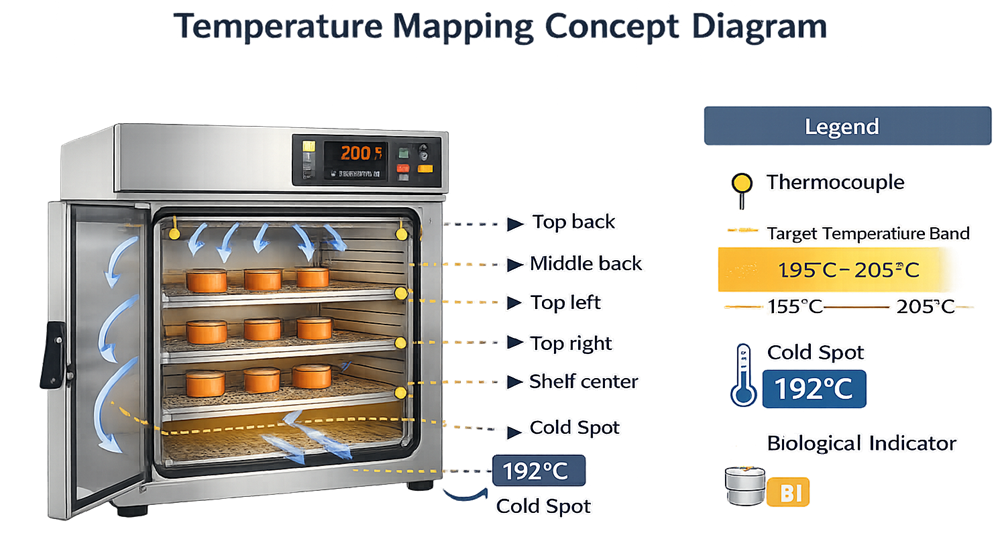
Temperature Mapping Strategy
Temperature mapping is the central technical element of dry heat qualification. Mapping should:
- Identify spatial temperature variation
- Confirm uniform heat distribution
- Establish validated cold spot locations
- Verify that minimum validated temperature is achieved for required exposure duration
Mapping configuration should reflect:
- Maximum and minimum load configurations
- Shelf placement variations
- Chamber volume utilization
Mapping results form the basis for biological indicator placement during PQ.
Acceptance Criteria
Acceptance criteria must be defined and approved prior to protocol execution. Criteria shall be scientifically justified, risk-based, and traceable to the intended sterilization objective.
At minimum, qualification acceptance criteria should address:
- Maximum allowable temperature deviation within the chamber or zone
- Minimum exposure time at validated target temperature
- Stability of temperature control during hold period
- Calibration tolerance of all critical sensors
- Biological indicator inactivation at validated cold spot locations
Criteria must demonstrate that the system consistently achieves the required sterility assurance objective under defined worst-case conditions.
Equipment-Specific Acceptance Considerations
While core criteria apply to all dry heat systems, certain limits must reflect equipment design characteristics.
Static Hot Air Ovens
- Defined maximum spatial temperature variation across shelves
- Confirmation that door-adjacent locations meet minimum temperature
- Recovery time limits following door disturbance
These systems require careful evaluation of stratification effects.
Forced-Air Circulation Ovens
- Uniformity limits reflecting validated airflow performance
- Acceptance criteria tied to verified fan operation
- Defined alarm response requirements for airflow interruption
Mechanical airflow introduces additional control dependencies that must be verified.
Continuous Dry Heat Tunnels
- Zone-specific minimum temperature criteria
- Defined conveyor speed tolerance during validated cycle
- Demonstration that coldest vial location achieves required exposure time
- HEPA integrity and airflow boundary confirmation
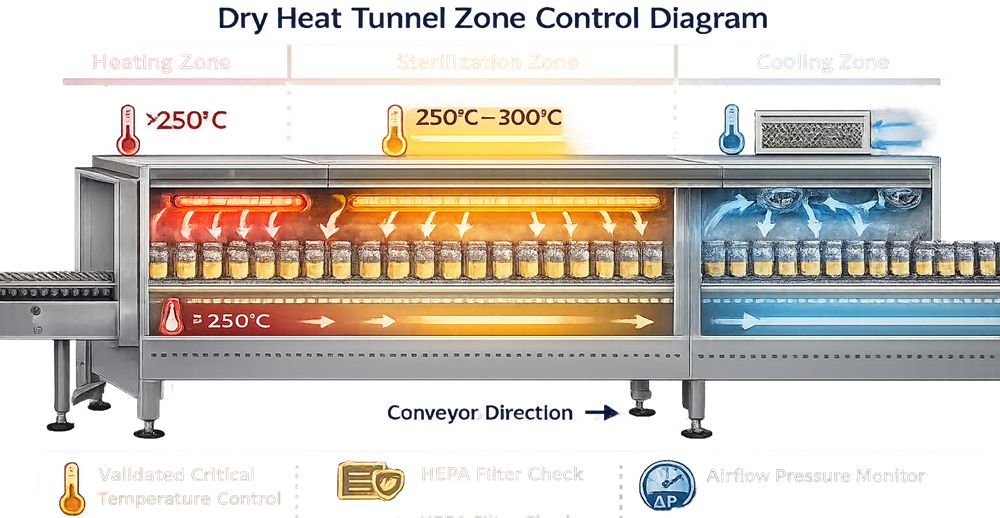
Tunnel qualification must address integrated system performance, not temperature alone.
Requalification Strategy
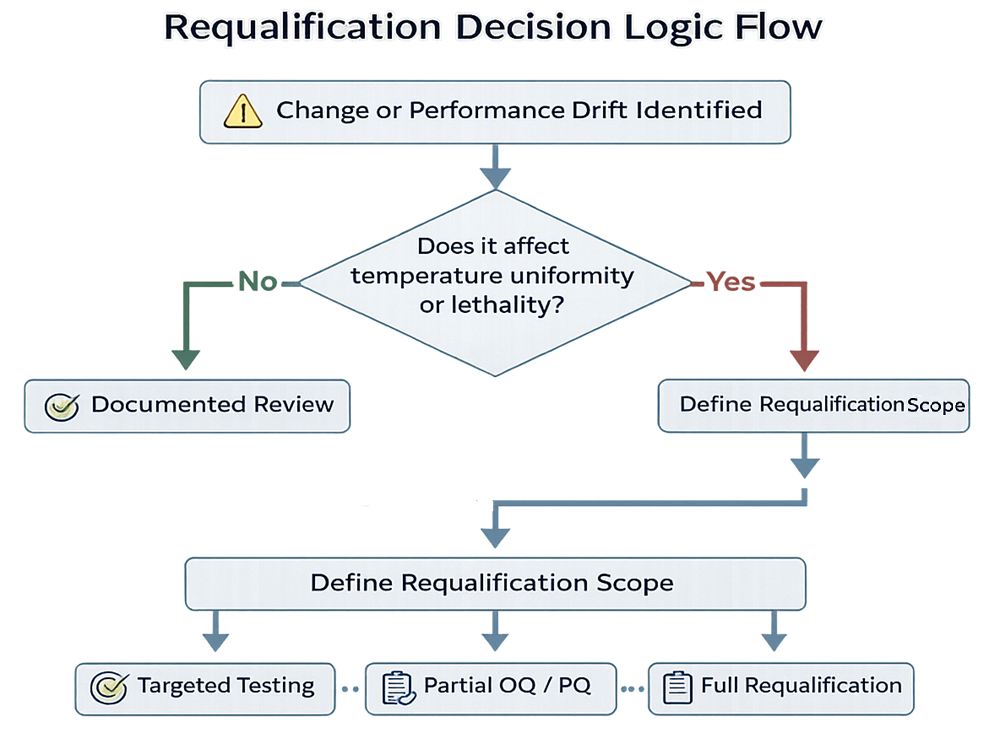
Requalification scope must reflect risk and system history. Triggers may include:
- Heating element replacement
- Control system modification
- Sensor relocation
- Significant maintenance
- Repeated temperature excursions
- Load configuration changes
Requalification may consist of:
- Targeted verification testing
- Partial requalification
- Full OQ and PQ repetition
Documentation review alone is acceptable only when risk is demonstrably low and supported by performance history.
Regulatory Alignment
Dry heat qualification aligns with requirements such as:
- 21 CFR 211.113(b) — validated sterilization processes
- 21 CFR 211.63 — equipment suitability
- EU GMP Annex 1 — sterilization validation and control
- ISO 20857 — dry heat sterilization
Regulators evaluate not only initial data but the robustness of ongoing lifecycle control.
