Containment and Dust Collection Systems
1. Overview
Containment and dust collection systems are used in pharmaceutical manufacturing to control airborne particulate generated during material handling and solid dosage processing. Many operations such as powder dispensing, milling, blending, granulation, and tablet compression generate fine particulate that can become suspended in the air if not properly controlled.
These systems serve two primary objectives. First, they protect product quality by preventing cross-contamination between products and process areas. Second, they protect personnel and the surrounding environment from exposure to active pharmaceutical ingredients and excipient dust.
Containment systems are therefore an important component of facility design and process equipment integration in solid dosage manufacturing environments. Properly designed dust control systems support regulatory expectations for contamination control, operator safety, and environmental protection.
2. Sources of Dust Generation
Airborne particulate can be generated during several common pharmaceutical operations. Powder transfer and dispensing activities are often major sources because powders are exposed to the environment during handling. Other significant dust-generating processes include:
- Milling and particle size reduction
- Powder blending operations
- Granulation and drying
- Tablet compression
- Tablet dedusting
- Capsule filling
- Material discharge from bins or containers
During tablet compression, for example, fine particles can be released from the die table or during ejection of tablets. Dedusting equipment is typically used immediately downstream of the tablet press to remove excess powder from tablet surfaces.
The illustration below presents typical sources of airborne particulate generated during solid dosage manufacturing operations such as milling, blending, powder transfer, tablet compression, and capsule filling. These processes can release fine powder into the surrounding environment if effective containment and dust collection controls are not implemented.

If dust is not effectively captured, particles can disperse into the surrounding manufacturing area, increasing the risk of contamination and operator exposure.
3. Containment Strategies
Several engineering approaches are used to contain powder and dust during pharmaceutical processing. These approaches are often combined to achieve effective containment.
Local Containment
Local containment is achieved by enclosing dust-generating equipment or by installing capture devices close to the source of dust generation. Examples include enclosed tablet presses, glove boxes, isolators, and contained transfer systems. These solutions physically restrict the spread of powder into the surrounding environment.
The illustration below presents local containment and dust extraction applied to a tablet press. Dust generated during compression and tablet ejection is captured through dedicated extraction ports connected to a dust collection system, preventing airborne particulate from entering the surrounding manufacturing area.

Local Exhaust Ventilation
Local exhaust ventilation systems remove airborne particulate directly at the source using negative pressure airflow. Dust-laden air is captured through hoods or duct connections and transported to filtration equipment where particles are removed before air is returned or exhausted.
Local exhaust systems are commonly used with equipment such as tablet presses, mills, and powder handling stations.
Enclosed Material Transfer
Containment can also be improved by minimizing open material handling steps. Enclosed transfer systems such as vacuum transfer lines, split butterfly valves, and contained docking systems reduce the potential for dust release during material movement.
4. Dust Collection Systems
Dust collection systems are designed to capture airborne particulate generated during pharmaceutical processing and remove it from the air stream. Typical dust collection systems consist of several key components:
- Dust capture hoods or equipment connections
- Ductwork transporting contaminated air
- Filtration units such as cartridge or bag filters
- Fans generating airflow through the system
- Collection containers for captured particulate
Air containing suspended particles is drawn through the duct system by a fan. The air passes through filtration elements that trap particulate while allowing clean air to exit the collector. The illustration below presents a typical pharmaceutical dust collection system showing the airflow path from dust capture points through ductwork to the dust collector where particulate is removed by filtration before air is discharged or returned to the facility.
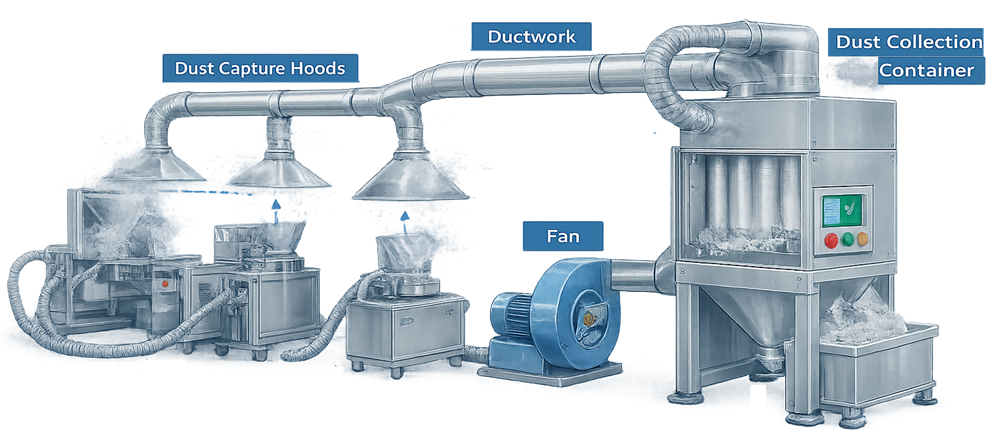
Pharmaceutical dust collectors are typically constructed of stainless steel or coated materials compatible with cleaning procedures and contamination control requirements.
5. Filtration Technologies
Dust collection systems commonly use several types of filtration technologies depending on the application. The illustration below presents the filtration principle used in pharmaceutical dust collectors where dust-laden air passes through cartridge or bag filters that capture particulate while allowing cleaned air to exit the system.

Cartridge Filters
Cartridge filters use pleated filter media with large surface area to capture fine particulate. These filters are widely used in pharmaceutical dust collection systems due to their high efficiency and compact design.
Cartridge Filters
Bag filters consist of fabric filter bags through which air passes while particulate accumulates on the outer surface of the bag. Periodic cleaning mechanisms remove collected dust from the filter surface.
HEPA Filtration
High Efficiency Particulate Air filters may be used in systems where high levels of filtration are required before air is returned to the manufacturing environment.
Filter selection must consider particle size distribution, airflow requirements, cleaning methods, and containment requirements.
6. Integration with Process Equipment
Dust collection systems are often directly connected to pharmaceutical processing equipment. Tablet presses, mills, granulators, and capsule filling machines frequently include dedicated dust extraction ports.
Proper integration ensures that airborne particulate generated at the source is immediately captured and transported to the dust collector. Design considerations include:
- Airflow balance across multiple equipment connections
- Proper duct sizing to maintain transport velocity
- Avoidance of powder accumulation in ductwork
- Isolation of equipment for maintenance
In some systems, dust collectors serve multiple pieces of equipment through a common duct network.
The illustration below presents integration of dust collection systems with multiple pieces of pharmaceutical processing equipment such as mills, blenders, and tablet presses connected to a common dust extraction network.

7. Qualification and Performance Verification
Containment and dust collection systems must be qualified to demonstrate that they operate as intended and effectively control airborne particulate generated during pharmaceutical processing. Qualification activities typically follow the lifecycle approach used for other GMP equipment and utilities, including Installation Qualification, Operational Qualification, and Performance Qualification.
7.1 Installation Qualification
Installation Qualification verifies that the dust collection system has been installed according to approved engineering design and manufacturer specifications. Because these systems include multiple mechanical and airflow components, installation verification must confirm correct configuration of the entire system. Typical IQ verification activities include:
- Equipment identification and model verification for dust collectors, fans, and filter housings
- Verification of ductwork routing and connection to process equipment
- Confirmation of duct diameter, materials of construction, and weld quality where applicable
- Verification of fan installation and rotation direction
- Confirmation of electrical connections and control panel configuration
- Verification of filter type, material, and installation orientation
- Inspection of dust collection bins or containment containers
- Verification of pressure gauges, differential pressure sensors, and monitoring devices
- Review of engineering drawings, airflow diagrams, and installation documentation
The objective of Installation Qualification is to confirm that the system has been assembled according to the approved design and is ready for operational testing.
7.2 Operational Qualification
Operational Qualification demonstrates that the dust collection system functions correctly across its defined operating range. This phase focuses on airflow performance, filter operation, and system control functions. Key OQ tests typically include:
- Verification of fan operation and airflow generation
- Measurement of airflow rates within ductwork and at equipment connection points
- Verification of duct transport velocity sufficient to prevent particulate accumulation
- Measurement of pressure drop across filters under normal operating conditions
- Verification of filter cleaning mechanisms such as pulse jet cleaning or shaker systems
- Verification of alarm functions for high differential pressure conditions
- Confirmation of proper operation of dust discharge systems or collection containers
Airflow testing may include measurement of capture velocity at equipment inlets and hood openings to ensure that airborne dust is effectively drawn into the system.
The illustration below presents typical airflow verification measurements performed during qualification of a pharmaceutical dust collection system. Measurements include capture velocity at extraction hoods and equipment ports, duct air velocity within the extraction network, and static pressure measurements across the dust collector filters. These measurements confirm that airborne particulate generated during processing is effectively captured and transported through the extraction system while maintaining proper filtration performance.
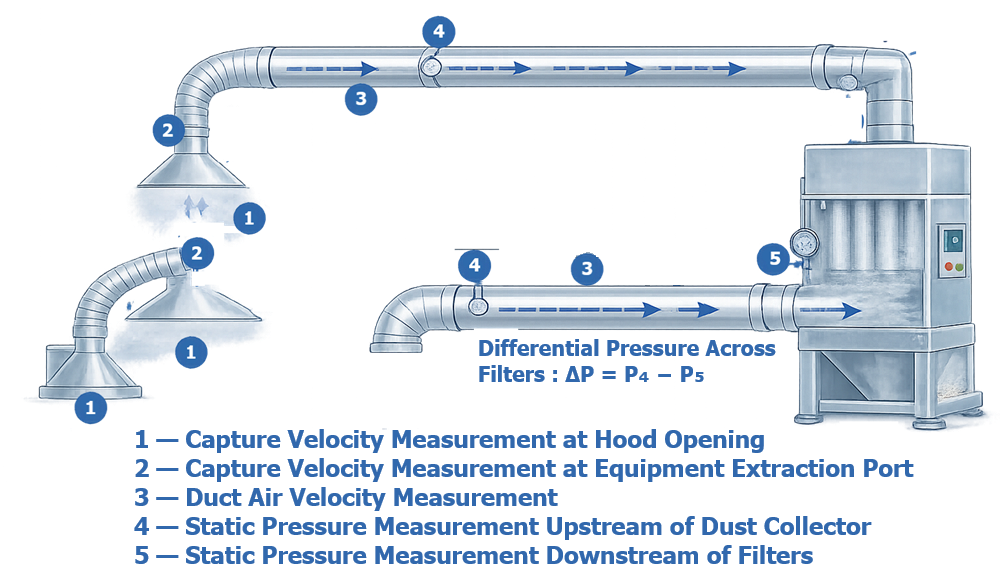
Operational Qualification also confirms that the system maintains negative pressure relative to surrounding areas where required to prevent dust escape.
7.3 Performance Qualification
Performance Qualification verifies that the containment and dust collection system effectively controls airborne particulate during actual manufacturing operations.
PQ testing typically occurs while processing representative materials under normal production conditions. During this phase, the system must demonstrate the ability to capture dust generated during operations such as powder transfer, milling, blending, or tablet compression. Typical PQ evaluation methods may include:
- Observation of visible dust release during operation
- Airborne particulate monitoring within the process area
- Surface contamination monitoring around equipment
- Verification that dust collectors maintain stable differential pressure across filters during operation
- Evaluation of dust accumulation within the collector and discharge mechanisms
Where containment performance is critical, surrogate powder testing or containment verification studies may be performed to confirm that airborne particulate levels remain within predefined limits.
7.4 Airflow Verification and Balance
Because dust collection systems rely on airflow for effective operation, airflow balance across the system must be verified during qualification. Testing may include:
- Measurement of airflow at individual equipment connections
- Verification of airflow distribution across multiple process equipment connections
- Adjustment of dampers or balancing devices to achieve proper airflow distribution
- Verification that airflow changes caused by opening or closing equipment connections do not compromise system performance
Proper airflow balance ensures that each connected process device receives sufficient extraction capacity to capture generated dust.
7.5 Filter Integrity and Maintenance Verification
Filters are critical components of dust collection systems and must be verified to ensure proper performance. Qualification testing typically confirms:
- Correct filter installation
- Integrity of filter seals and mounting
- Differential pressure across filters within acceptable operating ranges
- Proper operation of automatic filter cleaning systems
Filter monitoring programs are typically established following qualification to ensure that filter loading and replacement intervals are properly managed.
7.6 Continued Performance Verification
After qualification is complete, routine monitoring ensures that the dust collection system continues to perform effectively throughout its operational lifecycle. Typical monitoring activities include:
- Routine differential pressure monitoring across filters
- Inspection of dust discharge systems
- Verification of airflow indicators or airflow alarms
- Periodic inspection of ductwork for dust accumulation
- Preventive maintenance of fans, motors, and control systems
Trending of system performance parameters helps detect gradual performance degradation and supports timely maintenance interventions.
8. Summary
Containment and dust collection systems play a critical role in pharmaceutical solid dosage manufacturing by controlling airborne particulate generated during powder handling and processing operations. These systems protect product quality, prevent cross-contamination, and reduce operator exposure to active pharmaceutical ingredients and excipient dust.
Qualification of containment and dust collection systems demonstrates that the system is properly installed, operates as designed, and effectively captures process-generated particulate during manufacturing activities. Installation verification, airflow testing, operational checks, and performance evaluation under representative operating conditions confirm that the system provides effective containment and dust removal.
Through proper engineering design, integration with process equipment, and systematic qualification, dust collection systems support safe and controlled manufacturing environments. Routine monitoring, maintenance, and performance verification ensure that containment effectiveness is maintained throughout the operational lifecycle of the system.
