E-Beam Sterilization Process
Electron beam sterilization is a terminal sterilization method that uses high-energy electrons generated by an accelerator to deliver a defined absorbed radiation dose to product. The objective is achievement of the required Sterility Assurance Level, typically 10⁻⁶, through controlled ionizing radiation exposure.
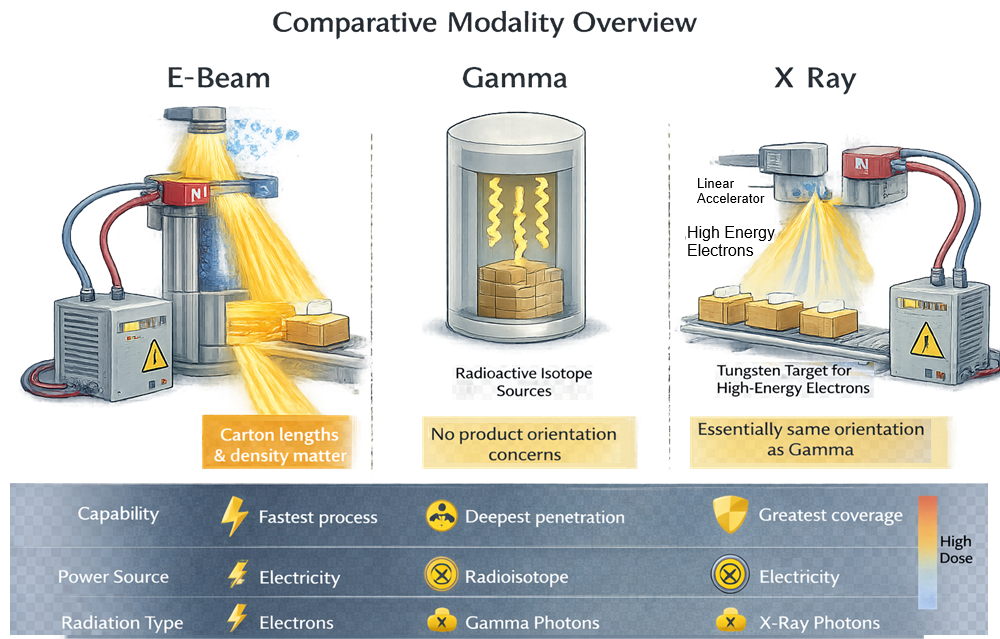
Unlike gamma sterilization, which relies on continuous radioactive decay from Cobalt-60, e-beam systems use electrically powered accelerators. The beam can be switched on and off, and dose delivery is extremely rapid.
E-beam sterilization is widely used for:
- Medical devices
- Single-use components
- Packaging materials
- Certain pharmaceutical systems
- Products sensitive to prolonged irradiation exposure
Dose establishment and validation are governed under ISO 11137.
1. Radiation Source and Physics
E-beam sterilization uses high-energy electrons generated by electrically powered particle accelerators. Unlike gamma systems, which rely on radioactive decay, e-beam systems produce radiation only when energized.
Common accelerator technologies include:
- Linear accelerators
- Dynamitrons
- Rhodotrons
Each system generates electrons and accelerates them through a high-voltage electric field, typically in the range of 5–10 MeV for industrial sterilization applications.
The following diagram illustrates the fundamental architecture of an electron beam sterilization system, showing how electrons are generated, accelerated through a high-voltage column, magnetically scanned across the product path, and delivered as a controlled irradiation beam. The schematic highlights the electrically powered nature of the system, the absence of radioactive material, and the integration of beam scanning with conveyor-based product transport.

Electron Generation and Acceleration
The process begins with an electron gun, which emits electrons from a heated cathode. These electrons are accelerated through a vacuum tube by applying a high potential difference. As they gain kinetic energy, they form a focused, high-velocity electron beam.
The beam is magnetically scanned across the product width to achieve uniform surface coverage while the product moves on a conveyor system beneath or through the beam.
Radiation is therefore delivered as charged particles, not photons.
Interaction with Matter
Because electrons carry charge and mass, they interact strongly with materials:
- They cause direct ionization of molecules
- They deposit energy rapidly
- They lose energy quickly as they penetrate material
- They scatter when encountering density changes
This produces intense, localized energy deposition with very high dose rates. Exposure times are typically measured in seconds.
Microbial inactivation occurs through:
- DNA strand breaks
- Free radical formation
- Disruption of cellular replication
Lethality remains dose dependent and is expressed in absorbed dose units (kGy).
Key Operating Characteristics
E-beam sterilization has distinct physical properties:
Very high dose rate
Short exposure duration
Electrical on/off capability
No radioactive source handling
Stable beam output not subject to decay
Because the radiation source is electrically controlled, there is no continuous emission and no long-term source strength decline, unlike gamma sterilization.
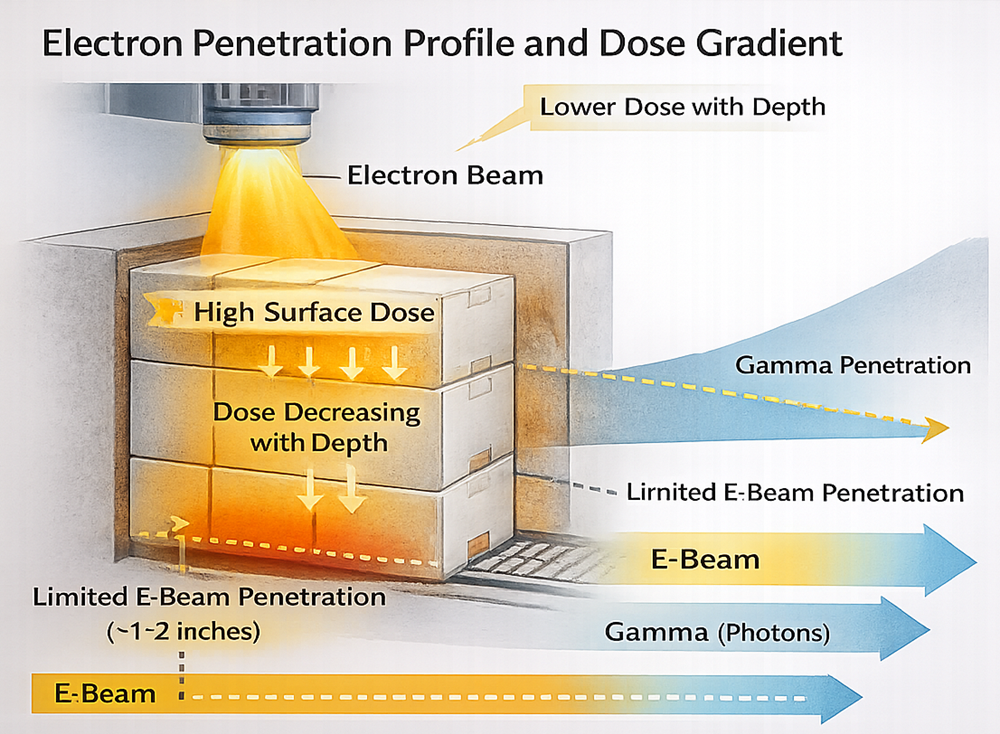
Penetration Behavior
Electron penetration depth is limited compared to gamma photons.
Penetration is influenced by:
- Electron energy (MeV)
- Product density
- Material atomic composition
- Product thickness
As electrons penetrate, they progressively lose energy and eventually stop within the material. This produces:
- Steep dose gradients
- Potential overexposure at surface
- Risk of underdosing in dense core regions
Maximum practical penetration for sterilization energies is generally suitable for low- to moderate-density products and cartons, but not dense palletized loads. The following diagram illustrates the characteristic electron beam dose gradient, demonstrating high surface dose, rapid energy attenuation with depth, and the resulting penetration limitations compared to photon-based radiation systems.
Design Implications
Because of limited penetration and strong interaction with matter:
- Product thickness must be evaluated carefully
- Packaging density must be controlled
- Orientation during irradiation may require optimization
- Double-sided exposure or flipping may be required
- Dose mapping is highly configuration dependent
In e-beam sterilization, product geometry and packaging configuration are not secondary variables. They are primary determinants of dose distribution and must be treated as validated parameters.
The image below shows a vault-based industrial electron beam sterilization installation. The system includes the accelerator assembly mounted adjacent to a heavily shielded concrete irradiation vault, with conveyor-based product transport passing through a shielded penetration tunnel. This configuration is typical of high-energy, high-throughput commercial sterilization facilities.

2. Microbial Inactivation Mechanism
Like gamma radiation, e-beam causes:
- DNA strand breaks
- Free radical formation
- Disruption of microbial replication
Microbial lethality follows logarithmic reduction kinetics and is expressed in absorbed dose (kGy).
Because dose rate is extremely high, exposure times are often measured in seconds rather than hours.
3. Typical E-Beam Facility Configuration
An e-beam facility generally includes:
- Electron accelerator
- Beam scanning system
- Conveyor transport
- Product handling system
- Shielded irradiation vault
- Real-time dosimetry monitoring
Products typically pass under or through the beam in a conveyorized system. In some configurations, products may be flipped or exposed from both sides to improve dose distribution.
E-beam facilities may be:
- In-house installations
- Contract sterilization providers
Compared to gamma facilities, infrastructure is less complex because no radioactive material licensing is required.
4. Product Bioburden Assessment
Bioburden assessment requirements are identical in principle to gamma sterilization.
The manufacturer must:
- Establish baseline bioburden
- Demonstrate stability
- Define worst-case product configuration
- Control manufacturing variability
Bioburden data support dose establishment using ISO statistical methods.
Unstable or poorly controlled bioburden is incompatible with reduced-dose strategies.
5. Dose Establishment Study
Dose establishment for e-beam sterilization follows the statistical framework defined in ISO 11137. The objective is to determine the minimum absorbed dose required to achieve the target Sterility Assurance Level, typically 10⁻⁶. Although the radiation modality differs from gamma, the dose substantiation methodology is identical in principle.
Approved ISO Approaches
The standard provides three primary dose-setting methods:
Method 1 – Bioburden-Based Statistical Method
Method 1 is the most established and statistically rigorous approach.
It is typically used for:
- High-volume commercial products
- Stable and well-characterized bioburden
- Mature manufacturing processes
The method involves:
- Determination of average product bioburden
- Selection of a verification dose based on ISO tables
- Irradiation of a statistically defined number of product units
- Sterility testing of those units
- Statistical confirmation that the proposed sterilization dose achieves SAL 10⁻⁶
Method 1 requires a relatively large sample size and provides strong statistical confidence. It is often preferred for routine commercial manufacturing.
Method 2 – Incremental Dose Response Method
Method 2 is a dose-response approach that evaluates sterility outcomes at multiple increasing dose levels.
It involves:
- Irradiating product groups at incremental doses
- Performing sterility testing at each level
- Constructing a dose-response relationship
- Extrapolating the minimum sterilization dose
Method 2 requires more samples and laboratory effort. It is less commonly used in routine commercial practice but may be applied when bioburden variability or development-stage data require a more exploratory approach.
VDmax – Verification Dose Method
VDmax is a reduced-sample statistical approach suitable for products with controlled and relatively low bioburden.
The most common form is VDmax 25, which verifies the acceptability of a 25 kGy sterilization dose.
Execution typically includes:
- Determination of average bioburden
- Irradiation of a limited number of product units at a defined verification dose
- Sterility testing
- Statistical confirmation that 25 kGy achieves SAL 10⁻⁶
VDmax can also be used to justify sterilization doses lower than 25 kGy when supported by stable and sufficiently low bioburden data.
This method reduces sample size and time but requires disciplined bioburden control.
Execution Elements Common to All Methods
Regardless of the selected approach, dose establishment includes:
- Irradiation of defined sample quantities under controlled conditions
- Verification dose exposure
- Sterility testing using validated methods
- Statistical substantiation of the sterilization dose
For e-beam systems specifically, samples must be irradiated using the validated orientation and configuration defined during dose mapping. Because penetration is limited and dose gradients may be steep, configuration control is critical during validation studies.
Dose Selection Considerations
While 25 kGy has historically been applied as a conventional sterilization dose, current regulatory expectation requires dose substantiation based on actual product bioburden and statistical validation, not reliance on historical norms.
Dose selection must consider:
- Bioburden magnitude and variability
- Product density and thickness
- Packaging configuration
- Material radiation tolerance
- Manufacturing process stability
Outcome
The result of the dose establishment study is a validated sterilization dose specific to:
- The defined product
- The validated configuration
- The selected irradiation parameters
It is not a generic industry value. It is a statistically supported and configuration-controlled parameter within the validated lifecycle.
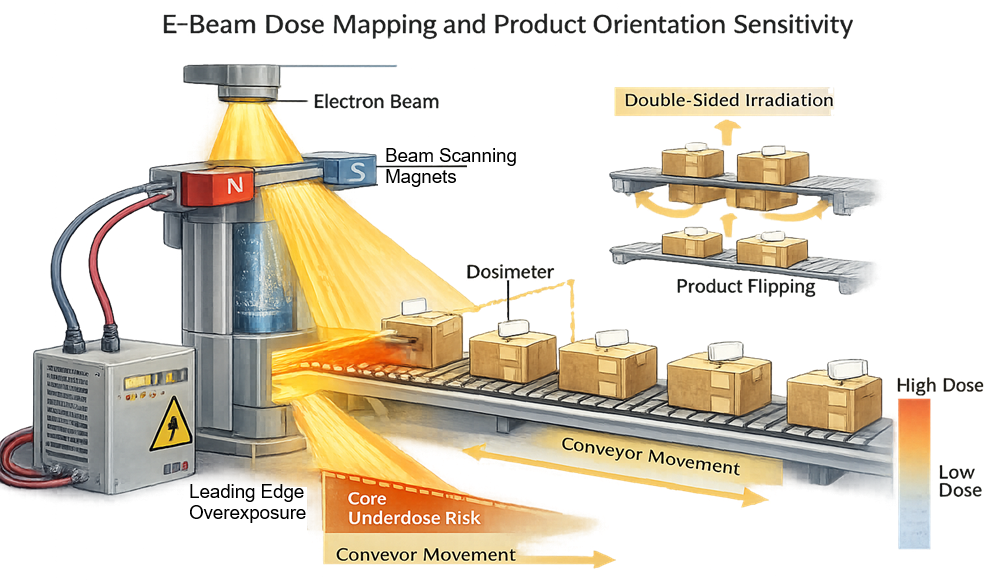
6. Dose Mapping and Distribution Control
Dose mapping is particularly critical in e-beam sterilization because:
- Penetration depth is limited
- Density variation strongly influences dose absorption
- Dose gradients can be steep
Mapping determines:
- Minimum absorbed dose location
- Maximum absorbed dose location
- Dose uniformity ratio
Typical e-beam challenges:
- Shadowing effects
- Uneven product density
- Conveyor orientation sensitivity
- Edge overexposure
Acceptance criteria require that the lowest measured absorbed dose within the defined worst-case product configuration is equal to or greater than the validated sterilization dose established during dose substantiation.
In addition, the highest measured absorbed dose must not exceed the maximum radiation tolerance of the product materials, packaging components, or functional performance limits.
E-beam mapping often requires evaluation from multiple orientations or double-sided exposure.
The following diagram illustrates the sensitivity of electron beam dose distribution to product orientation, conveyor direction, and density variation. It highlights common distribution effects such as leading-edge overexposure, rapid dose attenuation with depth, and potential core underdose, as well as the use of double-sided irradiation or product flipping to achieve acceptable dose uniformity. This configuration-dependent behavior is a defining characteristic of e-beam sterilization and must be controlled during validation and routine processing.
7. Routine Production Irradiation
Routine control parameters include:
- Beam energy
- Beam current
- Conveyor speed
- Scan width
- Product orientation
Real-time dosimetry systems monitor dose delivery. Routine dosimeters are typically placed at defined monitoring positions within each production lot.
Because the beam is electrically controlled, dose output is stable and does not decay over time, unlike gamma sources.
8. Dose Audit and Periodic Verification
Ongoing control includes:
- Periodic verification dose testing
- Bioburden monitoring
- Review of dose records
- Requalification after significant changes
Triggers for reassessment include:
- Packaging modifications
- Density changes
- Process changes affecting bioburden
- Equipment parameter adjustments
Lifecycle control is equivalent in rigor to gamma sterilization.
9. Modality Characteristics and Application Boundaries
Electron beam sterilization offers distinct operational and physical characteristics that differentiate it from gamma and X-ray systems. These characteristics define both its strengths and its constraints.
Operational Characteristics
- Extremely fast processing due to very high dose rate
- Electrical on/off control of radiation generation
- No radioactive isotope handling or decay management
- Stable beam output not subject to source depletion
- Precise control of beam current, energy, and conveyor speed
These attributes make e-beam attractive for high-throughput applications and facilities seeking to avoid radioactive source licensing and long-term isotope management.
Physical Constraints
- Limited penetration depth compared to photon-based modalities
- Strong sensitivity to product density and thickness
- Steep dose gradients within product cross-sections
- Increased complexity in dose mapping
- Potential need for product flipping or double-sided exposure
- Unsuitable for very dense or fully palletized loads
Because electrons interact strongly with matter and lose energy rapidly, product geometry and packaging configuration are primary determinants of dose distribution.
Engineering Implication
E-beam sterilization is highly effective when products are designed with penetration depth and density limitations in mind. It is not universally interchangeable with gamma sterilization, particularly for dense pallet configurations requiring deep photon penetration.
The following diagram provides a high-level comparison of the three primary industrial radiation sterilization modalities, highlighting differences in radiation type, penetration behavior, energy source, and configuration sensitivity. While all modalities deliver absorbed dose measured in kilogray and follow the same statistical validation framework, their physical behavior and operational boundaries differ substantially.cantly.