Endotoxin Control Strategy in GMP Manufacturing
Endotoxin control in GMP manufacturing is a preventive, system-based discipline. It is not achieved through finished product testing alone. Effective control requires upstream risk management, engineered system design, validated processes, and routine monitoring.
The objective is straightforward: prevent endotoxin introduction, prevent bacterial proliferation, and remove or inactivate endotoxin where required.
1. Foundational Principle: Control the Source
Endotoxin originates from Gram-negative bacteria. Consequently, endotoxin risk is fundamentally a microbial control risk.
Endotoxin does not appear spontaneously. It is generated when Gram-negative bacteria grow, multiply, and undergo cell lysis. Therefore, any condition that permits bacterial proliferation represents a potential endotoxin amplification point.
From a GMP perspective, endotoxin control is a preventive risk management activity. It requires identification of conditions that allow bacterial survival, growth, or biofilm formation, followed by implementation of engineering and procedural controls.
Regulatory expectations for microbial and water system control are embedded in:
• 21 CFR 211 – requirements for equipment cleaning, sanitation, and water system control
• EU GMP Annex 1 – expectations for contamination control strategy and utility oversight
• ICH Q9 – requirement for systematic risk identification, evaluation, and control
Under these frameworks, endotoxin control must be based on documented risk assessment and justified control measures.
Primary Endotoxin Risk Drivers
The following conditions represent elevated endotoxin risk because they support Gram-negative bacterial growth or persistence:
• Water systems, especially ambient or inadequately sanitized systems
• Moist environments with intermittent flow or stagnation
• Biofilm formation inside piping, tanks, and equipment
• Raw materials of biological or natural origin
• Inadequately cleaned product-contact equipment
• Open processing steps allowing environmental exposure
Each of these conditions can increase:
• Probability of bacterial proliferation
• Magnitude of endotoxin generation
• Difficulty of subsequent removal
In risk terminology:
• Hazard: Presence of Gram-negative bacteria
• Hazardous situation: Conditions allowing bacterial growth or biofilm formation
• Harm: Patient exposure to endotoxin exceeding established limits
Effective control strategy therefore focuses on reducing the likelihood of bacterial proliferation and minimizing the opportunity for endotoxin accumulation.
Risk-Based Interpretation
Endotoxin control should be evaluated using structured risk assessment. Key considerations include:
• Route of administration
• Maximum daily dose
• Process exposure time
• Water system design robustness
• Historical trend data
High-risk products, particularly parenterals and intrathecal products, require more stringent control measures due to lower allowable endotoxin limits defined in USP <85> Bacterial Endotoxins Test and European Pharmacopoeia 2.6.14.
The foundational control logic is therefore: Control microbial growth → Limit endotoxin generation → Maintain compliance with defined endotoxin limits.
Endotoxin control begins upstream. It is not primarily an analytical testing activity. The diagram below presents a system-level view of endotoxin control in GMP manufacturing. It illustrates the relationship between primary endotoxin risk sources, preventive control layers embedded in facility and process design, and verification mechanisms required to demonstrate sustained compliance. The model emphasizes that endotoxin assurance is achieved through layered prevention and continuous oversight, not reliance on finished product testing alone.
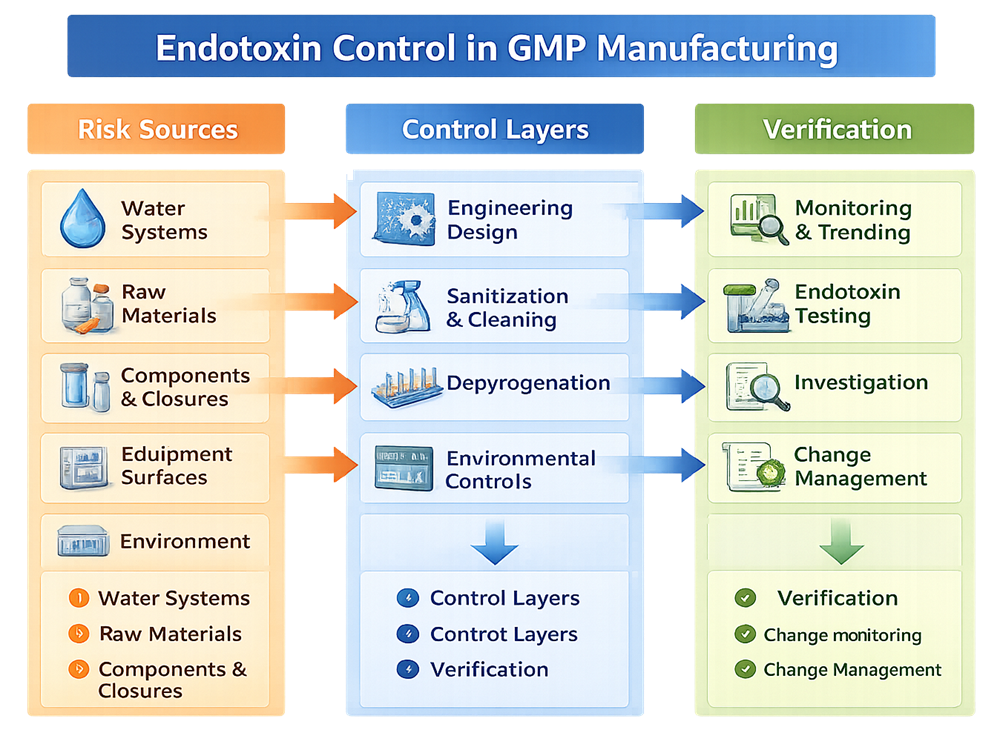
2. Water Systems as the Primary Risk Vector
Purified Water and Water for Injection systems represent the dominant endotoxin risk within pharmaceutical manufacturing. Because endotoxin originates from Gram-negative bacteria, any condition that allows bacterial survival, growth, or biofilm persistence within the water system directly increases endotoxin amplification potential. Water systems are uniquely vulnerable because they provide:
• Continuous moisture
• Nutrient availability at trace levels
• Internal surface area for attachment
• Complex piping geometries
These characteristics make design integrity critical.
2.1 Core Endotoxin Risk Mechanisms in Water Systems
Endotoxin risk in water systems arises primarily from design and operational vulnerabilities that permit microbial proliferation.
| Risk Mechanism | System Condition | Microbial Consequence | Endotoxin Risk Impact |
|---|---|---|---|
| Dead legs | Stagnant branch piping | Localized bacterial growth | Concentrated endotoxin accumulation |
| Low-flow or oversized piping | Reduced velocity and shear | Biofilm development | Persistent endotoxin reservoir |
| Inadequate temperature control | Suboptimal thermal profile | Enhanced mesophilic growth | Increased endotoxin generation |
| Insufficient sanitization | Incomplete microbial reduction | Surviving Gram-negative populations | Recurrent endotoxin release |
| Poor slope and drainability | Residual water retention | Microbial persistence after shutdown | Local endotoxin amplification |
| Infrequently used points of use | Extended stagnation | Bacterial regrowth between uses | Intermittent endotoxin spikes |
| Surface roughness or poor weld quality | Microbial attachment sites | Biofilm establishment | Chronic endotoxin source |
Biofilm formation represents the highest structural risk. Once established, biofilm protects microorganisms from sanitization and enables continuous endotoxin release during cell turnover.
2.2 Structural Risk Interpretation
From a risk perspective:
• Hazard: Presence of Gram-negative bacteria within the water system
• Enabling condition: Stagnation, low shear, nutrient access, or ineffective sanitization
• Risk outcome: Amplification of endotoxin concentration within the circulating loop
Water systems therefore act not merely as transport utilities, but as potential endotoxin generation environments if design integrity is compromised. The critical insight is structural: Endotoxin risk increases when hydrodynamics, temperature control, or sanitization effectiveness are inadequate.
3. Equipment and Component Preparation
Endotoxin may be introduced through:
• Glassware and stainless steel components
• Rubber stoppers and container closures
• Single-use components
• Filling assemblies
Control mechanisms include:
• Validated cleaning procedures
• Controlled drying
• Verified storage conditions
• Depyrogenation processes where required
Dry heat depyrogenation is typically used for glass components when high endotoxin reduction is necessary.
4. Process Design Controls
Open processing steps increase endotoxin risk. Environmental controls must minimize contamination potential. Critical elements:
• Controlled cleanroom classification
• Environmental monitoring
• Minimization of exposed product time
• Proper gowning discipline
While airborne endotoxin is less common than waterborne sources, environmental control reduces indirect contamination risk.
5. Depyrogenation as an Engineered Endotoxin Reduction Step
In specific manufacturing steps, upstream microbial control does not alone provide sufficient assurance that endotoxin levels will remain within defined limits. In such cases, a validated depyrogenation process is intentionally incorporated into the manufacturing flow. Depyrogenation is a designed control measure applied to heat-resistant materials where endotoxin burden must be demonstrably reduced before product contact. Typical applications include:
• Glass vials and ampoules
• Stainless steel product-contact components
• Filling assemblies
• Transfer vessels and tools
The objective is quantifiable endotoxin reduction under defined, worst-case conditions. Depyrogenation validation must demonstrate:
• Defined endotoxin challenge level
• Worst-case load configuration
• Scientifically justified acceptance criteria
• Reproducible performance across qualification runs
The process is qualified independently, with acceptance criteria based on endotoxin reduction capability rather than microbial lethality. Within the broader endotoxin control strategy, depyrogenation functions as a deliberate, validated risk reduction step applied where material design and product use demand additional assurance.
6. Routine Monitoring and Trending
Sustained control requires routine data evaluation. Monitoring typically includes:
• Water system endotoxin testing
• Microbial counts
• Trend analysis
• Investigation of excursions
Isolated passing results do not demonstrate control. Trend stability is the indicator of a robust system. Testing requirements for finished products are defined in USP <85> Bacterial Endotoxins Test and aligned with European Pharmacopoeia 2.6.14.
7. Change Management and Lifecycle Oversight
Endotoxin control must be reassessed when changes occur, including:
• Utility system modifications
• Equipment replacement
• Sanitization frequency adjustments
• Process scale changes
• Raw material supplier changes
Change evaluation must determine whether endotoxin risk is altered and whether requalification is required.
