Bacterial Endotoxin Testing Methods
Bacterial endotoxin testing verifies that pharmaceutical products comply with established endotoxin limits prior to release. Testing does not replace preventive control; it confirms that validated systems and processes have maintained endotoxin levels within acceptable limits.
Endotoxin testing in the United States is governed by USP <85> Bacterial Endotoxins Test. Equivalent requirements are defined in European Pharmacopoeia 2.6.14. These chapters define methodology, calculation of limits, and suitability requirements.
1. Principle of the LAL Reaction
Most bacterial endotoxin testing is based on Limulus Amebocyte Lysate, derived from the blood cells of the horseshoe crab. The lysate contains a highly sensitive enzymatic clotting cascade that is specifically activated by endotoxin, also known as lipopolysaccharide from Gram-negative bacteria. Limulus Amebocyte Lysate is derived from the blood cells of the horseshoe crab (Limulus polyphemus), whose innate immune clotting system forms the basis of endotoxin detection.

In the horseshoe crab, this cascade functions as part of the innate immune defense system. When Gram-negative bacteria enter the crab’s bloodstream, endotoxin activates a sequence of proteolytic enzymes that rapidly form a clot to isolate the invading organism. In laboratory testing, this same biological mechanism is harnessed to detect and quantify endotoxin in pharmaceutical samples.
The reaction proceeds as a stepwise enzymatic cascade:
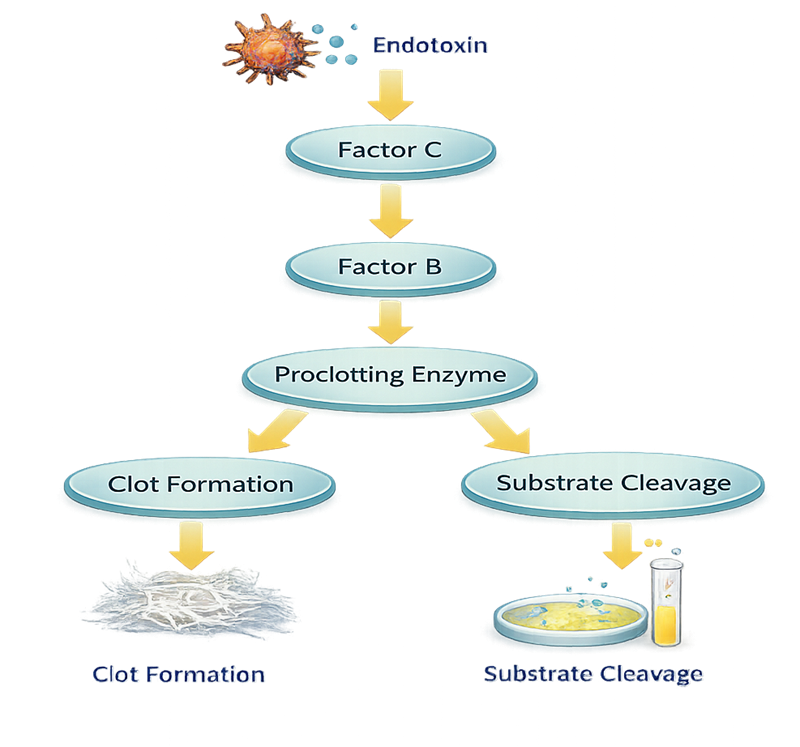
Endotoxin binds to and activates Factor C, an inactive enzyme precursor that serves as the specific endotoxin sensor. Activated Factor C then activates Factor B, which amplifies the signal by converting a proclotting enzyme into its active form. The active clotting enzyme produces the final measurable reaction.
Depending on the assay format, this final step results in:
• Formation of a visible gel clot
• An increase in turbidity
• Cleavage of a synthetic chromogenic substrate with measurable color development
In all cases, the magnitude or rate of the reaction correlates with the concentration of endotoxin present in the sample.
Because the system is enzymatic and amplifying, even very small quantities of endotoxin can trigger a detectable response. This amplification cascade is the basis for the high sensitivity of LAL-based endotoxin testing.
2. LAL Test Methods
Three primary LAL methodologies are compendial.
2.1 Gel-Clot Method
The gel-clot method is the original and simplest form.
Principle:
Endotoxin presence causes formation of a firm gel after incubation. Characteristics:
• Qualitative or semi-quantitative
• Requires visual interpretation
• Endpoint-based detection
• Less instrumentation required
Advantages:
• Simple
• Robust
• Low instrumentation complexity
Limitations:
• Lower precision
• Operator-dependent interpretation
2.2 Turbidimetric Method
The turbidimetric method measures increase in turbidity during clot formation.
Principle:
Endotoxin-induced clotting increases optical density over time.
Two formats:
• Endpoint turbidimetric
• Kinetic turbidimetric
Characteristics:
• Quantitative
• Requires spectrophotometric reader
• Higher sensitivity than gel-clot
Advantages:
• Objective measurement
• Automated data capture
• Suitable for routine QC
Limitations:
• Requires instrument qualification
• Susceptible to product interference
2.3 Chromogenic Method
The chromogenic LAL method quantifies endotoxin by measuring enzymatic cleavage of a synthetic chromogenic substrate. The substrate contains a peptide sequence recognized by the activated clotting enzyme and a bound chromophore. When the clotting enzyme cleaves the peptide bond, the chromophore is released, producing measurable color.
This converts the biological clotting cascade into a quantitative optical signal. The chromogenic method is available in two formats: endpoint and kinetic.
Endpoint Chromogenic Format
In the endpoint format, the reaction proceeds for a fixed incubation period. After incubation, absorbance is measured once. The color intensity is proportional to endotoxin concentration. Characteristics:
• Single time-point measurement
• Quantitative result
• Requires calibration curve
• Suitable for moderate-throughput testing
Because measurement occurs at a defined endpoint, timing precision is critical. Over- or under-incubation affects result accuracy.
Kinetic Chromogenic Format (KQCL)
The kinetic chromogenic method continuously monitors absorbance during the reaction. A predefined absorbance threshold is established. The instrument records the time required for the reaction to reach that threshold. Higher endotoxin concentration results in:
• Faster activation of the clotting cascade
• Faster substrate cleavage
• Shorter time to threshold
Lower endotoxin concentration results in:
• Slower cascade progression
• Longer reaction time
The measured parameter is reaction time, not final absorbance.
Calibration standards are plotted as log endotoxin concentration versus reaction time.
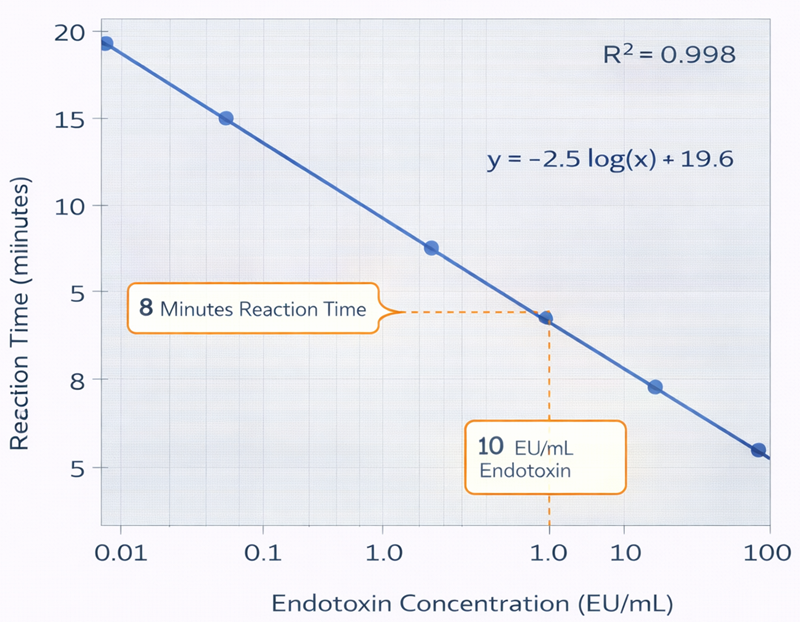
Within the validated range, the relationship is linear and inverse. In the kinetic chromogenic method, endotoxin concentration is determined from the time required for the reaction to reach a predefined absorbance threshold. As endotoxin concentration increases, the enzymatic cascade proceeds more rapidly, resulting in a shorter reaction time. When reaction time is plotted against the logarithm of endotoxin concentration, the relationship is linear within the validated calibration range and exhibits a negative slope. This reverse linear relationship forms the basis of the KQCL standard curve used for quantitative interpolation. Unknown sample concentrations are determined by interpolation against this kinetic standard curve.
Quantitative kinetic chromogenic assays are performed using calibrated microplate readers that measure absorbance at defined wavelengths over time to determine reaction onset.

Characteristics of Chromogenic Methods
• Fully quantitative
• High sensitivity
• Broad dynamic range
• Suitable for automation and microplate readers
• Objective, instrument-based detection
Advantages
• Reduced operator subjectivity compared to gel-clot
• High reproducibility
• Compatible with automated data capture
• Supports statistical validation and trend analysis
Limitations
• Requires calibrated and qualified instrumentation
• Susceptible to product matrix interference
• Requires validated standard curve performance
• Dependent on software and data integrity controls
In modern pharmaceutical quality control laboratories, kinetic chromogenic methods such as KQCL are widely used because they provide precise, reproducible, and automation-compatible endotoxin quantification.
2.4 Comparison of LAL Methods
The three compendial LAL methodologies differ in detection principle, instrumentation requirements, precision, and suitability for automation. The table below summarizes the technical distinctions relevant to method selection, validation, and routine quality control application.
| Attribute | Gel-Clot | Turbidimetric | Chromogenic |
|---|---|---|---|
| Detection Principle | Clot formation | Increase in turbidity | Color development from substrate cleavage |
| Result Type | Qualitative / limit test | Quantitative | Quantitative |
| Measurement Mode | Visual inversion | Optical density (kinetic or endpoint) | Optical density (kinetic or endpoint) |
| Instrument Required | No | Yes (spectrophotometer / plate reader) | Yes (spectrophotometer / plate reader) |
| Sensitivity | Moderate | High | High |
| Precision | Lower | Good | High |
| Automation Capability | Limited | Moderate to High | High |
| Data Integrity Considerations | Manual record | Electronic capture | Electronic capture |
| Typical Use | Limit testing | Routine QC quantitative testing | Routine QC and high-sensitivity testing |
Method selection should be based on product characteristics, required sensitivity, laboratory infrastructure, and data integrity expectations.
Quantitative turbidimetric and chromogenic endotoxin assays rely on calibration against a series of known endotoxin standards. These standards are prepared at defined concentrations and analyzed to generate a log-scale standard curve. The instrument response, typically optical density or reaction time, is plotted against the logarithm of endotoxin concentration. Within the validated range, the relationship is linear. Unknown sample concentrations are calculated by interpolating their measured response against this calibration curve. The correlation coefficient and linearity of the curve confirm assay performance and validity.
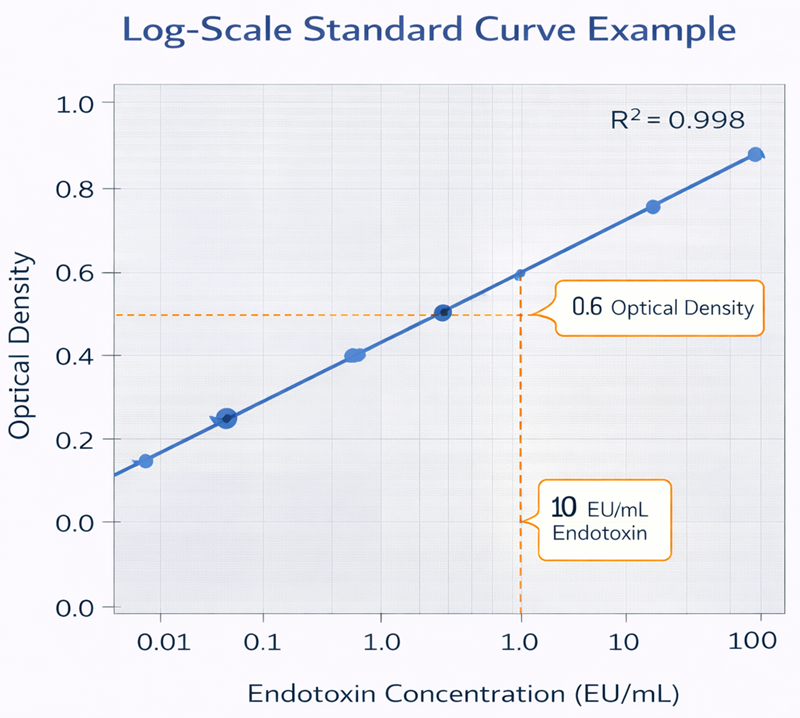
The use of a logarithmic concentration scale reflects the exponential nature of the enzymatic cascade and ensures accurate quantification across a wide dynamic range.
3. Method Suitability Testing
Compendial requirements mandate method suitability testing for each product.
Purpose:
Demonstrate that the product does not interfere with endotoxin detection.
Potential interferences:
• pH extremes
• High protein concentration
• Chelating agents
• Surfactants
• Preservatives
Suitability testing confirms:
• Absence of inhibition
• Absence of enhancement
• Accurate recovery of known endotoxin spike
Without demonstrated suitability, test results are not valid.
4. Calculation of Endotoxin Limits
Endotoxin limits are calculated based on:
Maximum allowable endotoxin exposure per kilogram of body weight
Route of administration
Maximum dose per hour
The compendial formula defines the allowable endotoxin concentration for each product. Testing must demonstrate compliance with calculated limit.
5. Emerging Alternatives
Recombinant Factor C (rFC) methods replace horseshoe crab lysate with recombinant technology.
Principle:
Selective activation of recombinant Factor C by endotoxin. Advantages:
• Animal-free
• Reduced biological variability
• Increasing regulatory acceptance
Compendial acceptance varies by region and requires validation against established methods.
6. Instrument and Data Integrity Considerations
Quantitative LAL methods rely on:
• Calibrated microplate readers
• Controlled incubation temperature
• Validated software
Systems used for electronic data capture must comply with:
• 21 CFR Part 11
• EU GMP Annex 11
Modern endotoxin testing laboratories frequently employ automated kinetic chromogenic systems that integrate incubation, optical measurement, calibration curve generation, and electronic data handling within a single platform. These systems improve precision, reduce manual variability, and support controlled data acquisition in regulated environments.

Instrument qualification is addressed separately in the Endotoxin Analytical System Qualification article.
Core Testing Principle
Bacterial endotoxin testing confirms compliance with established limits using validated, interference-free analytical methods.
The method must be:
• Suitable for the product matrix
• Calibrated and controlled
• Supported by documented recovery studies
• Performed within validated instrument parameters
Testing verifies control. It does not substitute for system design and process validation.
