EtO Sterilization Process
Ethylene Oxide EtO sterilization is a low-temperature gaseous sterilization method widely used for heat- and moisture-sensitive medical devices and combination products. It remains an established, regulator-accepted technology for complex devices with long lumens, mixed materials, electronics, and polymers that cannot tolerate steam or dry heat.
From a compliance standpoint, EtO sterilization is governed by a science-based validation framework aligned with ISO standards and FDA expectations. Unlike steam sterilization, EtO lethality is not driven by temperature alone. It is a multi-variable chemical process requiring tight control of gas concentration, humidity, temperature, and exposure time. In short, EtO is effective, but only when managed with discipline.
1. Ethylene Oxide Source, Storage, and Sterilizer Architecture
Effective EtO sterilization depends on controlled chemical supply, engineered gas delivery systems, and pressure-rated sterilizer design. The sterilization cycle cannot be separated from the infrastructure that supports it. Gas origin, storage conditions, metered delivery, chamber construction, and exhaust treatment all directly influence process safety, consistency, and regulatory compliance.
A disciplined understanding of this infrastructure is essential before evaluating lethality or validation strategy. The following diagram illustrates the engineered gas supply, metered delivery, and exhaust control architecture supporting an EtO sterilization system.
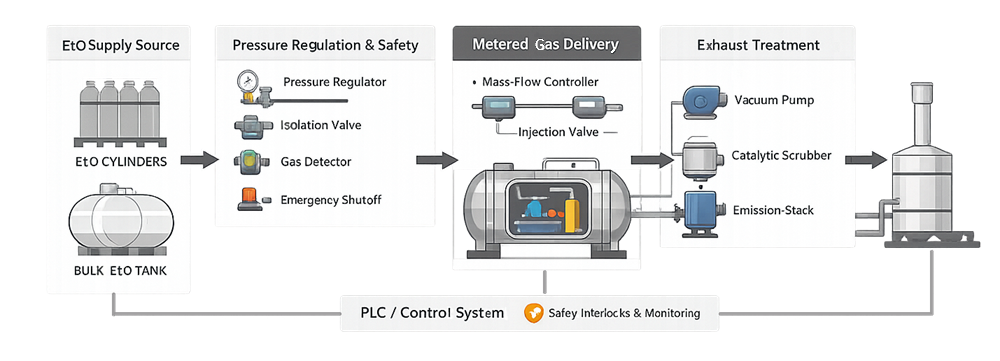
1.1. Ethylene Oxide Source and Supply Infrastructure
Ethylene oxide is produced industrially through controlled oxidation of ethylene. For medical and pharmaceutical sterilization applications, EtO is supplied in medical-grade purity with defined impurity limits. Sterilization facilities receive EtO in one of the following forms:
- 100 percent ethylene oxide
- Ethylene oxide blended with nitrogen
- Ethylene oxide blended with carbon dioxide
Blended mixtures are commonly used to reduce flammability risk and improve handling safety. Supply configurations depend on facility scale:
- High-pressure cylinders for smaller installations
- Ton containers for moderate throughput
- Bulk storage tanks for commercial sterilization operations
The selected configuration influences facility design, ventilation requirements, hazard classification, and emission control strategy.
1.2. Storage and Gas Delivery Systems
Ethylene oxide is stored as a liquefied compressed gas under pressure. The photograph below illustrates a secured EtO cylinder bank connected to a regulated manifold system supplying the sterilizer.

It presents significant hazards:
- Flammable across a broad concentration range
- Explosive under certain air mixtures
- Toxic and classified as a carcinogen
Accordingly, storage systems require:
- Dedicated, ventilated areas
- Explosion-rated electrical classification
- Gas detection and alarm systems
- Temperature control
- Secure cylinder restraint or bulk containment
Gas delivery to the sterilizer is controlled through a regulated manifold or bulk feed system. Typical components include:
- Pressure regulators
- Isolation valves
- Mass flow control devices or metered injection systems
- Safety interlocks
- Emergency shutoff valves
Sterilizers generally do not measure gas concentration directly. Instead, gas dosing is calculated using pressure-volume-temperature relationships and controlled injection volumes. This engineering principle is fundamental to understanding cycle reproducibility. Injection strategies may include:
- Single gas pulse
- Multiple pulsed injections
- Vacuum-assisted gas introduction
The delivery system must provide consistent, repeatable gas quantities under validated conditions.
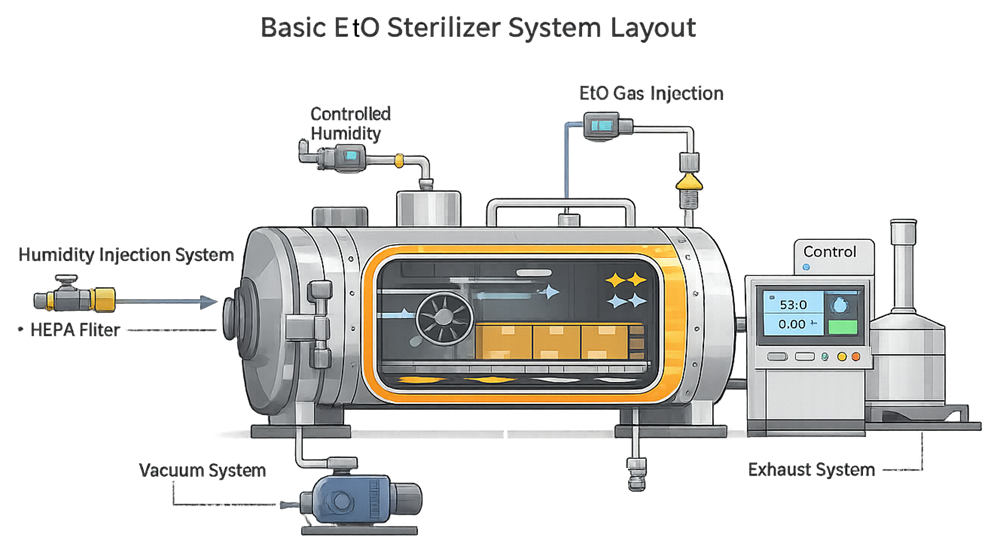
1.3. Basic EtO Sterilizer Design
An EtO sterilizer is a sealed, pressure-rated vessel integrated with environmental control and gas-handling subsystems. It is not simply a heated chamber.
The photograph below illustrates a production-scale double-door ethylene oxide sterilizer chamber with integrated pallet rail transport and industrial control interface.

Core system elements typically include:
Sterilization Chamber
A pressure vessel designed to withstand vacuum and moderate positive pressure. The chamber may be steam-jacketed or electrically heated and includes a sealed, interlocked door system. Internal circulation fans promote uniform temperature and gas distribution.
Vacuum System
Vacuum pumps are used to evacuate air prior to gas injection and to remove EtO following exposure. Vacuum improves gas penetration and uniformity. The system includes valves, condensers, and pressure monitoring instrumentation.
Humidification System
Relative humidity is introduced through controlled steam or vapor injection. Proper hydration of microorganisms significantly enhances lethality. Humidity control is therefore a critical design feature.
Gas Injection System
Metered injection valves deliver defined quantities of EtO into the evacuated chamber. Interlocks prevent unsafe pressure or concentration conditions.
Temperature Control System
Heating jackets or internal heaters maintain the validated process temperature. Multiple temperature sensors provide feedback to the control system to ensure uniform distribution.
Aeration and Exhaust Handling
After exposure, EtO must be removed from both the chamber and the product. Aeration may occur within the sterilizer or in a dedicated aeration chamber. Exhaust gases are directed through emission control systems such as catalytic oxidizers, thermal oxidizers, or scrubbers to meet environmental regulations.
A simplified layout of a typical EtO sterilizer system is shown below.
1.4. Integrated System Perspective
EtO sterilization is an integrated engineered system combining:
- Chemical handling
- Pressure vessel design
- Vacuum engineering
- Environmental emission control
- Industrial safety controls
- Automated process control
Each subsystem influences process performance and validation outcomes. Changes to gas supply pressure, humidity control accuracy, vacuum efficiency, or exhaust performance can affect lethality, residual levels, or safety compliance.
For this reason, qualification and lifecycle control must address the sterilizer as a complete system rather than a standalone chamber.
Understanding this infrastructure provides the foundation for evaluating mechanism of action, process phases, critical parameters, and validation strategy.
2. Fundamental Mechanism of Action
Ethylene oxide is an alkylating agent. It destroys microorganisms by reacting with:
- Proteins
- DNA
- RNA
The result is irreversible disruption of cellular metabolism and replication. Because EtO penetrates packaging and complex device geometries effectively, it is particularly suited for:
- Long or narrow lumens
- Multi-component assemblies
- Porous materials
- Pre-packaged sterile barrier systems
This deep penetration capability is the primary operational advantage of EtO over other modalities.
3. Core Process Phases
EtO sterilization is not a single step. It is a structured, multi-phase process. The overall EtO sterilization cycle follows a structured sequence of controlled phases.
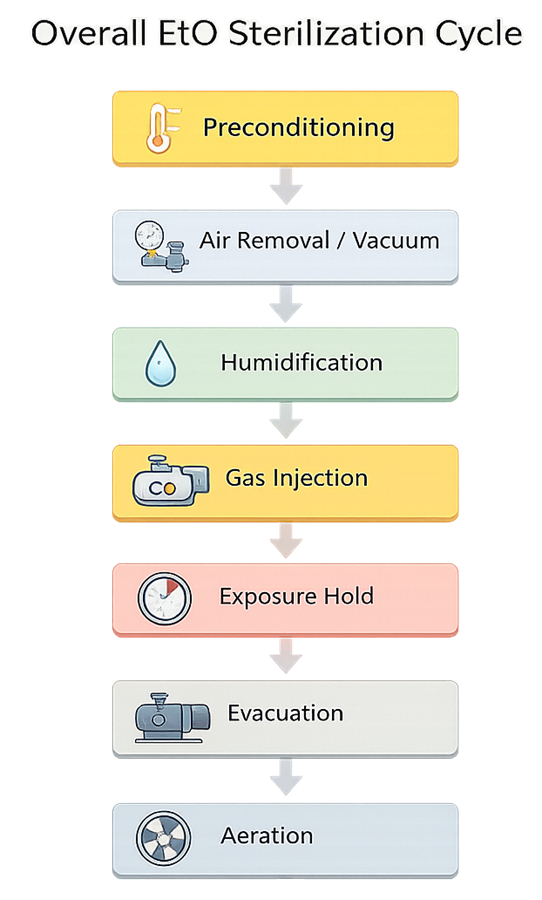
3.1 Preconditioning
Purpose: Prepare product and packaging for effective sterilization. Controlled parameters:
- Temperature
- Relative humidity
- Time
Humidity is critical. Microbial lethality increases significantly when organisms are properly hydrated. Poor humidity control is one of the most common causes of inconsistent performance.
3.2 Gas Exposure Phase
Purpose: Deliver validated lethality. Critical parameters:
- Gas concentration
- Chamber temperature
- Relative humidity
- Exposure time
- Pressure profile
These variables work together. Changing one without evaluating the others is poor practice and a frequent root cause of validation drift. Industrial systems may use:
- 100% EtO
- EtO blended with nitrogen or CO₂
Gas delivery can be performed through:
- Single injection
- Multiple pulsed injections
- Vacuum-assisted cycles
3.3 Post-Exposure Evacuation
Purpose: Remove residual EtO from the chamber and product surface.
Vacuum and air washes reduce chamber concentration prior to unloading.
3.4 Aeration
Purpose: Reduce residual EtO and byproducts within the product. Aeration may occur:
- In the sterilizer
- In a dedicated aeration chamber
- In a controlled room
This phase is not optional. Residual control is a regulatory and patient safety requirement.
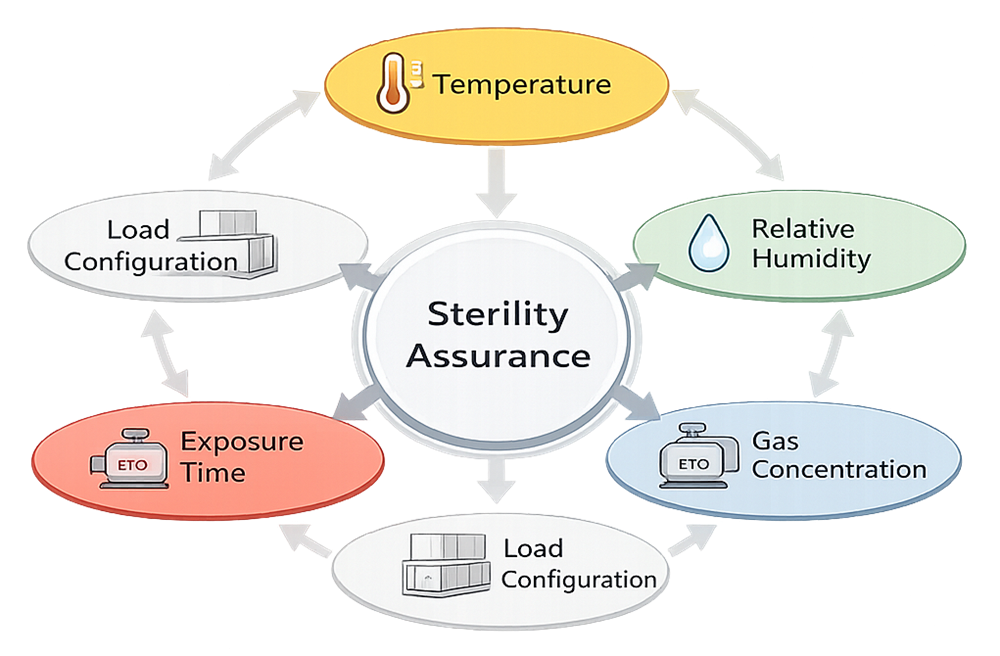
4. Critical Process Parameters
EtO sterilization performance is defined by the interaction of five primary variables:
- Temperature
- Relative humidity
- Gas concentration
- Exposure time
- Load configuration
Each must be defined during development and locked through validation. Traditional industry practice establishes lethality using:
- Overkill approach
- Half-cycle validation
- Bioburden-based method
The chosen approach must align with product risk and regulatory strategy. EtO lethality is driven by the interaction of multiple controlled variables.
5. Load Configuration and Packaging Considerations
EtO sterilization is load-sensitive. The following significantly affect performance:
- Product density
- Pallet configuration
- Carton arrangement
- Packaging material permeability
- Lumen length and diameter
Worst-case load identification is not theoretical. It must be justified and documented. Failure to define worst-case configuration upfront often results in costly revalidation.
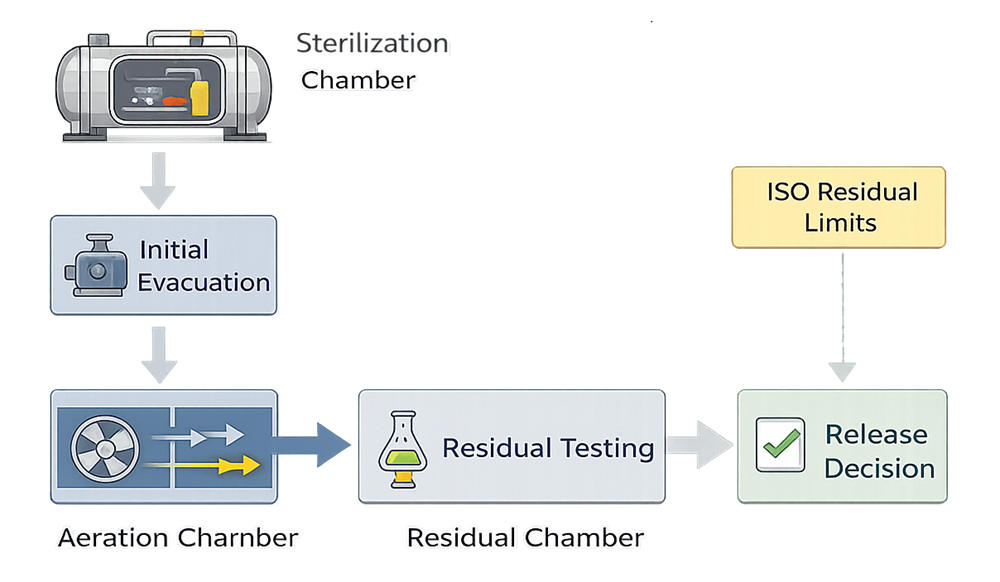
6. Residuals and Byproducts
Ethylene oxide can form:
- Ethylene chlorohydrin
- Ethylene glycol
Residual limits are controlled through ISO standards and toxicological risk assessment. Aeration time must be validated to demonstrate compliance. This is where many organizations underestimate complexity. Sterility alone is not sufficient. Residual safety is equally critical.
Residual control requires controlled evacuation, aeration, and analytical verification.
7. Safety and Facility Considerations
EtO is:
- Flammable
- Explosive under certain conditions
- Carcinogenic
Therefore, systems require:
- Explosion-proof equipment
- Gas monitoring systems
- Environmental controls
- Operator safety protocols
- Emission abatement systems
Regulators pay close attention to occupational and environmental controls.
8. Process Control and Validation Context
The EtO sterilization process must operate within a defined validation and lifecycle control framework. Process parameters, load configuration, humidity conditioning, gas delivery strategy, and aeration controls must be scientifically developed and formally qualified before routine use. Effective implementation requires:
- Defined development strategy
- Structured IQ, OQ, and PQ execution
- Biological indicator methodology
- Cycle lethality confirmation including half-cycle justification where applicable
- Residual evaluation and toxicological assessment
- Routine process monitoring
- Defined requalification criteria
EtO sterilization is not simply a chamber cycle. It is an integrated system combining microbiological control, chemical safety, packaging science, and facility engineering. Validation is the mechanism that converts this complex chemical process into a controlled, reproducible, and regulator-accepted sterilization system.
