Facility Qualification Lifecycle Overview
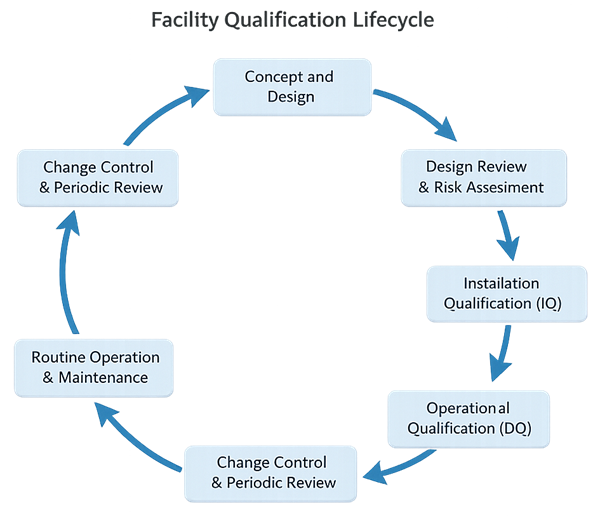
The facility qualification lifecycle defines the structured approach used to ensure that a GMP facility is designed correctly, constructed as intended, and operated in a controlled manner suitable for its intended use. For facilities, qualification is not a single event or a fixed sequence of documents. It is a lifecycle framework that begins with design decisions and continues through operation, change, and periodic review.
Unlike equipment or processes, facilities do not produce product directly. Their role is to provide controlled environments and utilities that enable compliant manufacturing. As a result, the emphasis of facility qualification is placed on design suitability and operational control, rather than on performance qualification in the traditional sense.
Design-Centered Lifecycle Approach
For GMP facilities, Design Qualification principles are foundational. Critical compliance attributes such as material and personnel flow, segregation, HVAC zoning, pressure cascades, environmental control strategies, and utilities capacity are established during the design phase. If these elements are not properly defined and reviewed upfront, downstream qualification activities cannot compensate for poor design choices.
Design review and risk assessment serve as the mechanism by which design intent is evaluated against regulatory expectations and intended use. This phase ensures that compliance is built into the facility rather than tested in after construction.
Installation and Operational Qualification
Following construction and installation, Installation Qualification (IQ) verifies that facility systems, components, and utilities are installed in accordance with approved drawings and specifications. IQ confirms alignment between design intent and the as-built condition. It is not a discovery exercise and should not be used to resolve undocumented field changes or design deficiencies.
Operational Qualification (OQ) then demonstrates that facility systems operate within defined limits and respond appropriately to controls, alarms, and interlocks. For facilities, OQ establishes operational control of systems such as HVAC, environmental controls, and critical utilities. OQ confirms that the facility can be operated in a compliant state under defined conditions.
Lifecycle Control After Qualification
Once IQ and OQ are complete, the facility transitions into routine operation. Maintaining the qualified state relies on ongoing lifecycle controls, including preventive maintenance, calibration, environmental monitoring, deviation management, and formal change control.
Change control and periodic review provide the feedback mechanism that links operational experience back to design assumptions and qualification decisions. When changes occur or performance trends indicate potential drift, the facility is reassessed using a risk-based approach. This may result in targeted requalification or design re-evaluation where necessary.
Position on Performance Qualification for Facilities
Performance Qualification is not considered a default or universal requirement for facilities. In many cases, facility performance is demonstrated through ongoing monitoring programs, equipment qualification, and process validation activities rather than through a standalone facility PQ protocol. Where performance verification is required, it is typically embedded within routine monitoring and lifecycle management rather than treated as a discrete qualification phase.
Lifecycle Perspective
The facility qualification lifecycle is therefore best understood as a continuous loop, driven by design intent, verified through IQ and OQ, and sustained through disciplined operational control. This approach aligns with regulatory expectations and reflects how facilities are practically evaluated during inspections.
Compliance is not achieved by completing a set of qualification documents. It is achieved by maintaining a facility in a demonstrable state of control throughout its operational life.
