Failure Tree Analysis
Failure Tree Analysis (commonly referred to as Fault Tree Analysis, FTA) is a structured risk analysis technique used to evaluate how combinations of failures can lead to a defined undesired event. In regulated pharmaceutical and medical device environments, FTA is typically applied to complex systems, high-risk processes, or scenarios where a single failure mode does not adequately explain potential impact to product quality, patient safety, or compliance.
FTA is a top-down, deductive approach. It begins with a specific undesired event and systematically traces backward to identify the underlying causes, failure mechanisms, and contributing factors that could lead to that event. These relationships are represented in a tree structure using logical dependencies that show how individual failures may occur independently or in combination.
Failure Tree Analysis and Event Tree Analysis – Conceptual Comparison
Failure Tree Analysis (FTA) and Event Tree Analysis (ETA) are related risk analysis techniques, but they serve different purposes and are applied at different stages of risk evaluation.
Failure Tree Analysis focuses on identifying the causes of an undesired event by working backward from the outcome. It is a deductive, top-down approach used to understand how combinations of failures, weaknesses, or control gaps could lead to a specific failure condition. In GMP environments, FTA is commonly used to support failure prevention, control strategy evaluation, and risk-based validation decisions.
Event Tree Analysis, in contrast, evaluates the possible outcomes following an initiating event by working forward through potential event sequences. It is an inductive approach used to explore how different responses, safeguards, or failures may influence downstream outcomes once an event has occurred.
In regulated pharmaceutical and medical device environments, FTA is applied far more frequently than ETA. FTA aligns naturally with risk-based validation, periodic assessment, and requalification planning. ETA is used more selectively, typically for conceptual evaluation of outcome pathways or safety-oriented analyses, rather than as a primary tool for defining validation scope or lifecycle control.
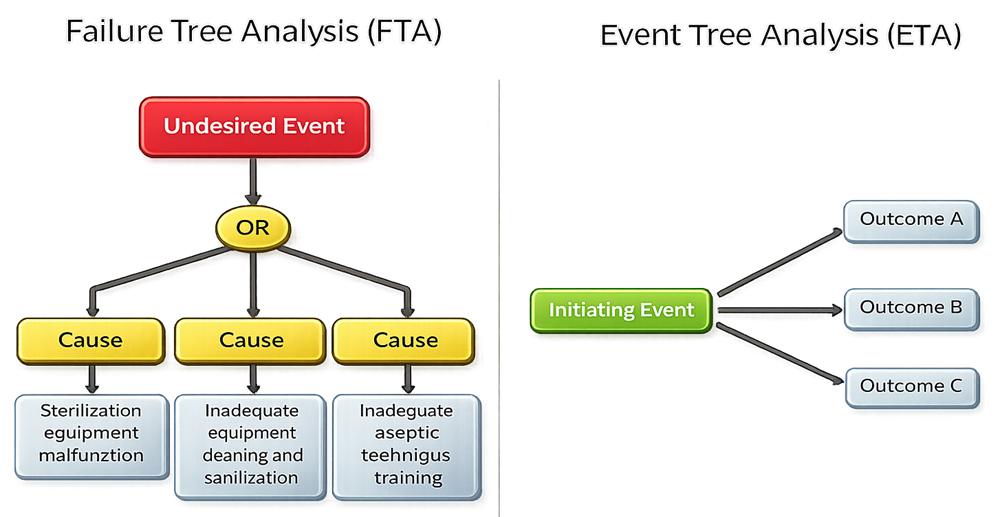
FTA asks “How could this failure occur?”, while ETA asks “What could happen next?”.
Purpose and Application of Failure Tree Analysis
Failure Tree Analysis is used to:
- Understand complex cause-and-effect relationships
- Identify combinations of failures that could bypass existing controls
- Support risk-based decisions where traditional FMEA may be insufficient
- Provide deeper justification for preventive controls, monitoring, or requalification scope
FTA is particularly valuable when assessing systems with multiple interdependencies such as sterile processing, utilities, environmental control systems, and automated equipment.
Methodology of Failure Tree Analysis
Identification of the Undesired Event
FTA begins with the clear definition of an undesired event. In a pharmaceutical manufacturing context, this may include events such as product contamination, loss of sterility assurance, critical equipment failure, or deviation from validated process conditions.
Construction of the Failure Tree
A hierarchical tree is constructed with the undesired event at the top. Branches are developed to represent contributing failure modes and conditions. Logical relationships are established to show whether failures occur independently or in combination.
Analysis of Failure Causes
Each branch of the tree is systematically evaluated to identify credible root causes. This step relies on subject matter expertise, historical performance data, deviation history, maintenance records, and process knowledge.
Assessment of Consequences
For each failure path, potential consequences are evaluated, including impact to product quality, patient safety, regulatory compliance, and business continuity. This step helps distinguish theoretical failures from credible risk scenarios.
Risk Prioritization and Mitigation
Failure scenarios are prioritized based on severity and likelihood. Risk mitigation measures are then defined, which may include engineering controls, procedural changes, enhanced monitoring, training, preventive maintenance, or additional validation activities.
Failure Tree Analysis Example
Undesired Event: Microbial contamination of a sterile injectable product
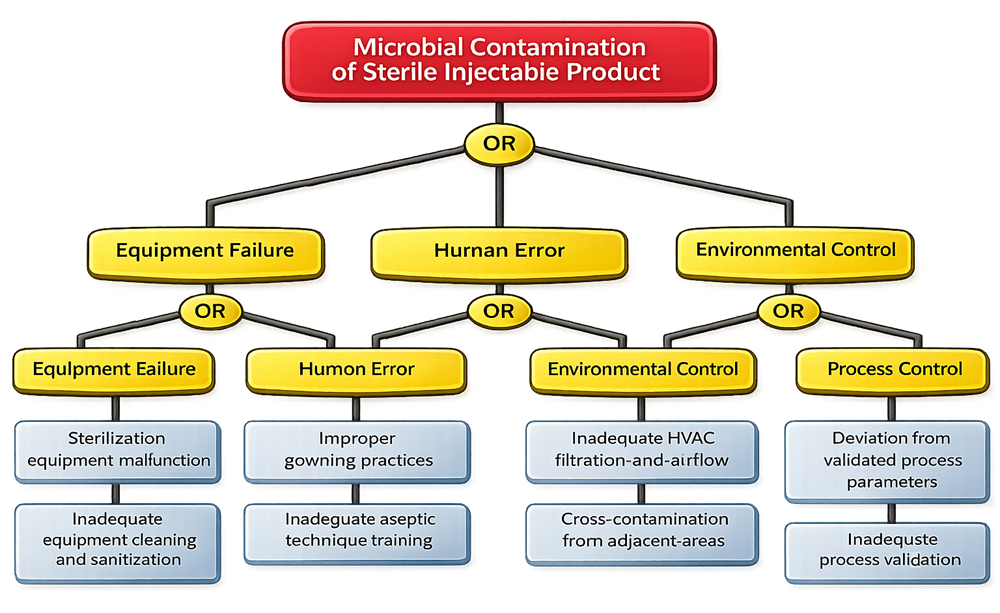
Potential Failure Paths:
Equipment-Related Failures
- Failure of sterilization equipment to achieve validated temperature or pressure
- Inadequate calibration or maintenance of sterilization systems
- Insufficient cleaning and sanitization of production equipment
Human Factors
- Improper gowning practices
- Inadequate training in aseptic technique
- Improper handling of sterile components
Environmental Controls
- Inadequate HVAC filtration or airflow control
- Airborne contamination from adjacent areas
- Insufficient segregation of clean and non-clean spaces
Process Control Failures
- Deviation from validated process parameters
- Inadequate monitoring of critical process parameters
- Lack of validated sterilization or aseptic filling processes
Each branch is evaluated to determine credible failure combinations and to assess whether existing controls are adequate. Where gaps are identified, targeted mitigation measures are implemented to reduce risk to an acceptable level.
Role of Failure Tree Analysis in a Risk-Based Lifecycle
Failure Tree Analysis complements other risk assessment tools by providing deeper insight into complex or high-risk scenarios. It supports risk-based validation strategies, informs periodic assessment focus areas, and may influence requalification scope where system complexity or failure history warrants additional scrutiny.
Summary
Failure Tree Analysis is a powerful analytical tool when applied judiciously. In regulated environments, it is best used to supplement structured risk assessments, strengthen preventive controls, and support defensible validation and lifecycle management decisions for critical systems and processes.
