Gamma Sterilization Process
Gamma sterilization is a terminal sterilization method that uses ionizing radiation emitted from Cobalt-60 sources to deliver a validated absorbed dose to product. The objective is to achieve a defined Sterility Assurance Level while preserving product integrity.
In the vast majority of cases, gamma sterilization is performed by specialized contract irradiation facilities. Ownership and control of the radioactive source require dedicated licensing, shielding infrastructure, and regulatory oversight, making in-house operation uncommon for pharmaceutical and medical device manufacturers.
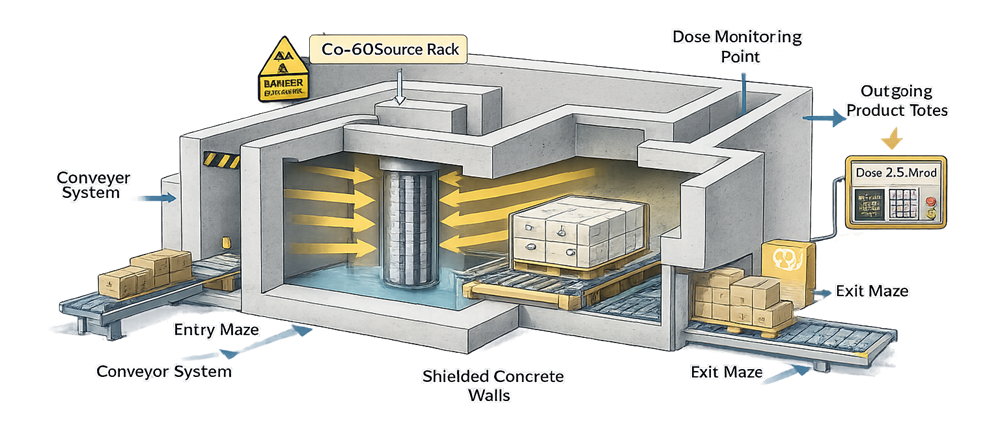
The manufacturer retains responsibility for validation, dose substantiation, and lifecycle control, even when sterilization is outsourced. The process is structured around five core technical phases:
- Product bioburden assessment
- Dose establishment study
- Dose mapping
- Routine production irradiation
- Dose audit and periodic verification
These elements operate under the framework of ISO 11137.
1. Product Bioburden Assessment
Bioburden assessment establishes the baseline microbial population present on or within the product prior to sterilization.
Objectives
- Quantify average bioburden per product unit
- Determine variability between lots
- Identify microbial flora characteristics
- Assess resistance profile relevance
Bioburden data must be representative of routine manufacturing conditions. Sampling plans should reflect:
- Worst-case product configurations
- Maximum hold times prior to sterilization
- Routine packaging configuration
- Normal environmental monitoring performance
The bioburden study supports:
- Selection of sterilization dose method
- Validation of dose substantiation model
- Ongoing dose audit frequency
Uncontrolled or unstable bioburden is a regulatory red flag. A stable, characterized bioburden program is foundational to radiation sterilization.
2. Dose Establishment Study
Dose establishment determines the minimum radiation dose required to achieve the target Sterility Assurance Level, typically 10⁻⁶. The following diagram illustrates the verification dose concept and statistical linkage between bioburden assessment, verification testing, and establishment of the validated sterilization dose.
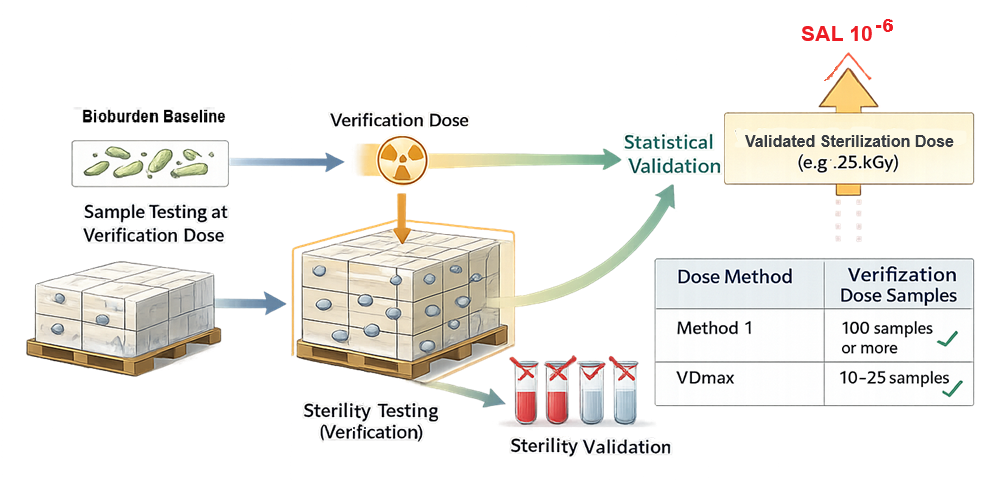
This is performed in accordance with ISO 11137 using one of the approved statistical methodologies:
Method 1 – Bioburden-Based Method
Primarily used for high-volume, commercially mature products with stable and well-characterized bioburden.
- Requires determination of average product bioburden
- Uses a defined verification dose
- Involves sterility testing of a statistically significant sample size
- Provides strong statistical substantiation
Method 1 is the most widely used approach for routine commercial products.
Method 2 – Incremental Dose Method
An experimental dose-response approach in which product units are exposed to increasing radiation dose levels and sterility testing is performed at each increment.
- Larger total sample size
- More laboratory intensive
- Less commonly used in routine commercial practice
Method 2 is typically reserved for special cases or development-stage evaluations.
VDmax Method – Verification Dose Method
A reduced-sample statistical approach suitable for products with controlled, relatively low bioburden.
- Commonly used to substantiate a 25 kGy sterilization dose (VDmax 25)
- Requires irradiation of a limited number of units at a defined verification dose
- Faster and more efficient than Method 1
VDmax may also be used to justify a sterilization dose lower than 25 kGy when supported by stable bioburden data.
Method Selection Considerations
The selected approach depends on:
- Magnitude and variability of product bioburden
- Production volume
- Product type and material compatibility
- Organizational risk tolerance
- Regulatory strategy
Execution Elements
Regardless of method, the dose establishment study includes:
- Controlled irradiation of defined sample quantities
- Sterility testing at the prescribed verification dose
- Statistical substantiation of the sterilization dose
Historically, 25 kGy was often applied as a default industry value. Modern regulatory expectation, however, emphasizes substantiation based on actual product bioburden rather than reliance on historical convention.
The outcome of the study is a validated sterilization dose, supported by statistical evidence and ongoing control requirements — not a nominal or assumed value.
3. Dose Mapping
Dose mapping characterizes radiation distribution throughout the product load configuration. The following diagram illustrates typical dose distribution within a pallet configuration and identification of minimum and maximum absorbed dose locations.
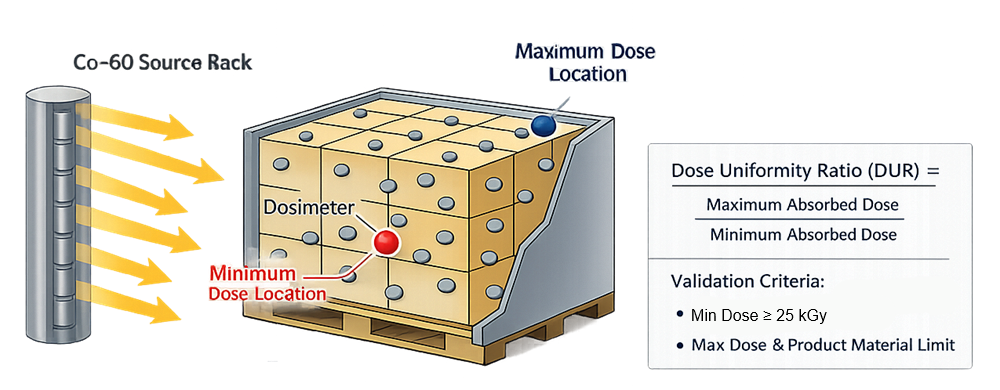
Gamma photons penetrate deeply and irradiators expose product from multiple angles, absorbed dose varies based on:
- Product density
- Pallet geometry
- Packaging material
- Orientation relative to source
- Conveyor path
During dose mapping:
- Dosimeters are placed throughout worst-case product locations
- Minimum and maximum absorbed dose points are identified
- Dose Uniformity Ratio is calculated
Acceptance criteria require:
Minimum dose ≥ validated sterilization dose
Maximum dose ≤ product material tolerance
Dose mapping must be performed:
- During initial validation
- After configuration changes
- When product density or packaging changes
The contract irradiation facility often executes physical irradiation, but the manufacturer defines the load configuration and worst-case model.
4. Routine Production Irradiation
Once validated, routine production proceeds under controlled parameters.
The contract irradiation facility controls:
- Exposure time
- Conveyor speed
- Source configuration
- Facility environmental conditions
Each production batch includes:
- Routine dosimeters
- Load documentation
- Traceability to dose mapping configuration
- Certificate of irradiation
Critical control parameters:
- Delivered minimum dose
- Maximum dose
- Product orientation
- Pallet density limits
Routine process control ensures that every production lot receives a dose within the validated range. Gamma sources undergo radioactive decay over time. Exposure duration must be periodically adjusted to compensate for source strength reduction.
5. Dose Audit and Periodic Verification
Sterilization validation is not a one-time exercise. Dose audits confirm continued validity of the established sterilization dose. Typically performed quarterly or at defined production intervals, dose audits involve:
- Re-verification dose irradiation
- Sterility testing of samples
- Statistical review of results
Triggers for re-evaluation include:
- Bioburden shifts
- Manufacturing process changes
- Packaging changes
- Load configuration modification
- Significant environmental trend deviations
Periodic verification ensures that the validated state is maintained throughout the product lifecycle. Failure to conduct meaningful dose audits is a common compliance deficiency.
6. Governance and Responsibility When Using Contract Irradiators
The following diagram illustrates the governance structure and lifecycle oversight model when gamma sterilization is performed by a contract irradiation facility, clarifying responsibility boundaries between the product manufacturer, the contract irradiator, and regulatory authorities.
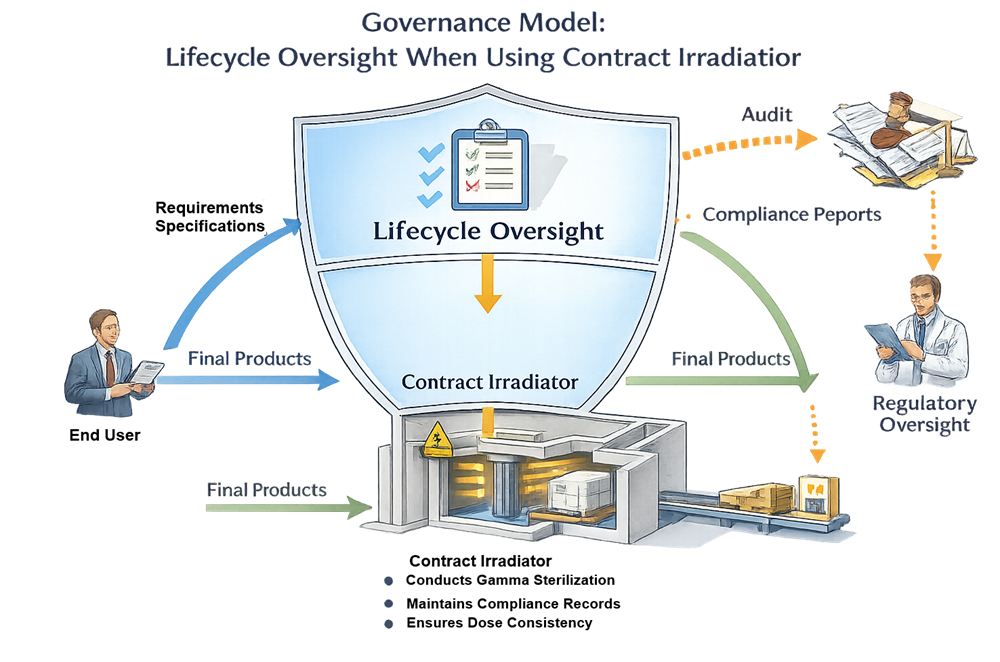
Even when sterilization is outsourced:
- The manufacturer owns the validation
- The manufacturer defines the product family
- The manufacturer approves dose mapping configuration
- The manufacturer reviews irradiation certificates
- The manufacturer controls change management
The contract facility operates under radiation safety and operational controls, but regulatory accountability remains with the product manufacturer. Clear quality agreements must define:
- Responsibilities for dose mapping
- Reporting requirements
- Change notification obligations
- Audit rights
Inspectors routinely review oversight of contract sterilization facilities.
Conclusion
Gamma sterilization is a scientifically robust and regulator-accepted terminal sterilization method. Its reliability depends not merely on exposure to radiation, but on disciplined control of:
- Bioburden
- Dose substantiation
- Load configuration
- Routine monitoring
- Periodic verification
When executed under a structured lifecycle framework consistent with ISO 11137, gamma sterilization provides predictable sterility assurance with broad material compatibility and deep penetration capability.
