Role of HVAC in GMP Compliance
In GMP-regulated facilities, HVAC systems are not support utilities. They are core quality systems. Their primary purpose is to establish and maintain environmental conditions that prevent contamination, control cross-contamination, and ensure consistent manufacturing conditions. Regulators have treated HVAC this way for decades, and that position has not changed.
From a compliance standpoint, HVAC performance is inseparable from product quality.
Impact of HVAC Parameters on GMP Quality System
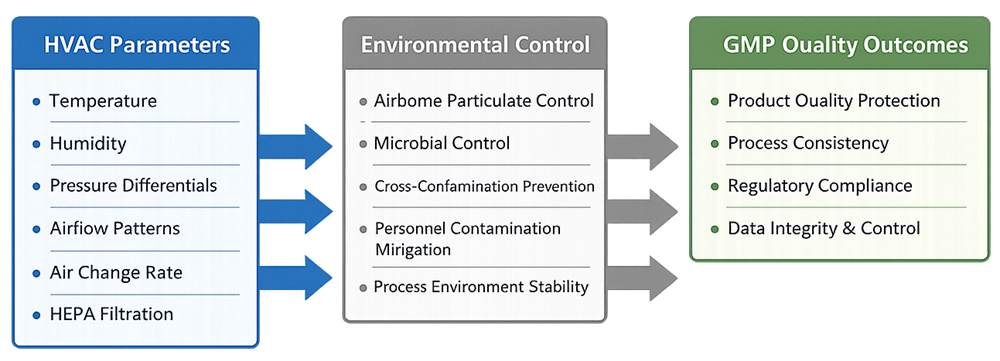
Environmental Control as a GMP Requirement
GMP regulations require facilities to be designed and operated in a manner that prevents contamination and mix-ups. HVAC systems are the primary engineering control used to meet this expectation by:
- Maintaining defined temperature and humidity ranges
- Controlling airborne particulate and microbial levels
- Establishing pressure differentials between rooms of different cleanliness
- Supporting classified environments where required
If these conditions cannot be demonstrated as controlled, the facility is not operating in a compliant state. There is no workaround.
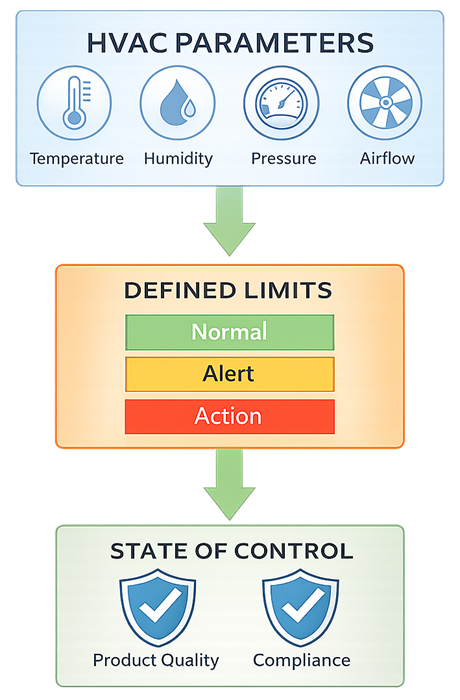
To maintain a sustained state of control, HVAC systems must operate within defined environmental parameter limits that are monitored and managed through the quality system.
Contamination and Cross-Contamination Control
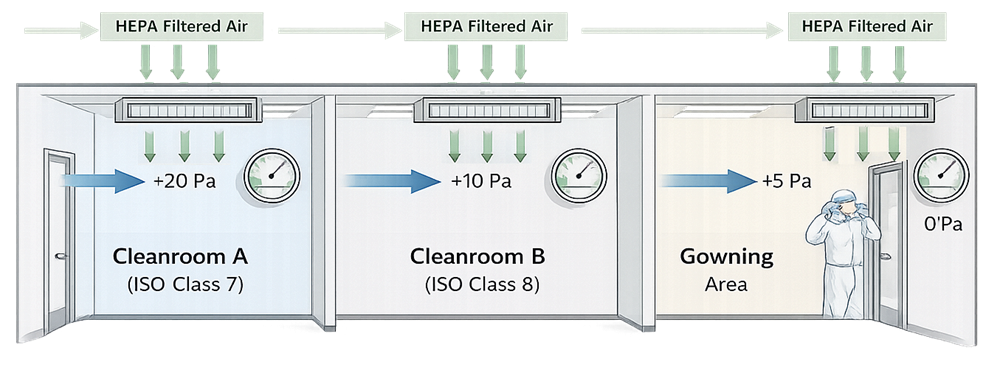
HVAC systems play a direct role in contamination control strategy by managing airflow direction, filtration, and room pressurization.
Effective HVAC design and operation ensure that:
- Air flows from cleaner areas to less clean areas
- Potential contaminants are diluted and removed
- Adjacent operations are protected from cross-contamination
- Personnel movement does not compromise room conditions
Regulators routinely link HVAC deficiencies to contamination risks, especially in sterile, aseptic, and compounding environments.
To prevent contamination and cross-contamination between adjacent areas, GMP facilities use pressure differentials and controlled airflow to ensure that air consistently moves from cleaner spaces to less clean spaces.
Pressure Cascade and Controlled Airflow
HVAC as a Quality-Critical System
In GMP facilities, HVAC systems should be formally classified as quality-impacting systems when they serve production, compounding, or controlled support areas.
This classification drives expectations for:
- Documented design intent
- Installation and operational qualification
- Defined operating limits and alarms
- Deviation management and investigation
- Change control and requalification
Treating HVAC as a maintenance-only system is a common failure mode and one that inspectors quickly identify.
Regulatory Expectations and Inspection Focus
During inspections, HVAC systems are evaluated not just on design, but on evidence of sustained control.
Inspectors typically focus on:
- Clear linkage between room classification and HVAC design
- Trending of temperature, humidity, and pressure data
- Investigation of environmental excursions
- Justification for alert and action limits
- Requalification following changes or recurring deviations
Documentation gaps or poorly defended limits often draw more scrutiny than minor technical issues.
Lifecycle Perspective
HVAC compliance is not achieved at startup. It is maintained over time.
A compliant HVAC program includes:
- Defined critical parameters and monitoring strategy
- Routine review of performance trends
- Risk-based response to deviations
- Periodic assessment of system suitability
- Requalification driven by risk and performance history
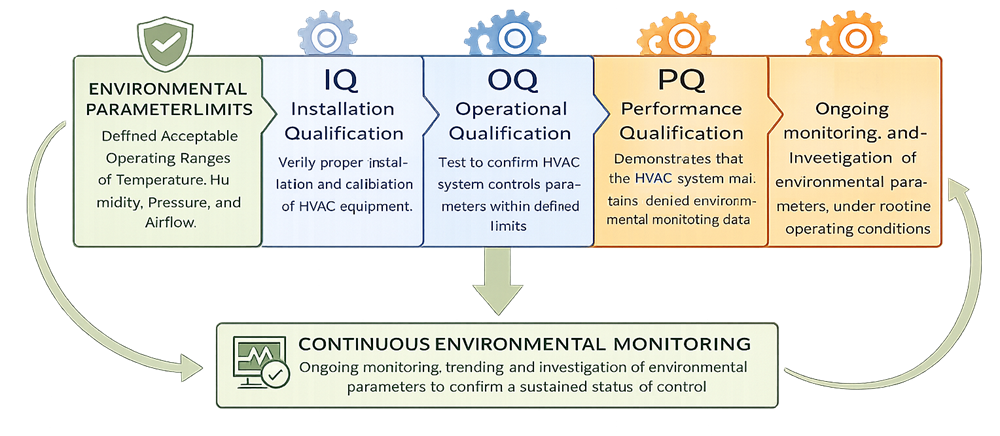
Facilities that adopt this lifecycle approach tend to experience fewer regulatory findings and more stable operations. HVAC systems are qualified to demonstrate their ability to maintain defined environmental parameters, and are then monitored continuously to ensure a sustained state of control.
HVAC Qualification and Monitoring Process
Practical Reality
The role of HVAC in GMP compliance is well established and not subject to interpretation. It is expected to work, to be documented, and to stay in control. Facilities that respect this reality operate smoothly. Facilities that do not eventually face enforcement attention.
