In-Process Inspection Equipment
1. Overview
In-process inspection equipment is used during pharmaceutical manufacturing to monitor product quality attributes while production is underway. These systems detect defects, contamination, and process deviations in real time and remove non-conforming units before they proceed to downstream operations.
Unlike end-of-line quality testing, in-process inspection is integrated directly into the manufacturing line. Inspection equipment operates continuously during production and provides immediate feedback on product quality. This allows operators and process engineers to identify abnormal conditions early and prevent the accumulation of defective material.
In-process inspection equipment is used during pharmaceutical manufacturing to monitor product quality attributes while production is underway. These systems detect defects, contamination, and process deviations in real time and remove non-conforming units before they proceed to downstream operations.
Unlike end-of-line quality testing, in-process inspection is integrated directly into the manufacturing line. Inspection equipment operates continuously during production and provides immediate feedback on product quality. This allows operators and process engineers to identify abnormal conditions early and prevent the accumulation of defective material.
In solid dosage manufacturing, in-process inspection is commonly positioned between major processing steps such as compression, dedusting, coating, and packaging. Typical systems inspect tablets, capsules, filled containers, or packaged units.
The illustration below presents a typical arrangement of in-process inspection equipment within a pharmaceutical tablet manufacturing line, showing how devices such as tablet dedusters, metal detectors, vision inspection systems, and checkweighers are positioned between major processing steps to detect defects before products proceed downstream.
Proper use of in-process inspection equipment supports process control, reduces batch rejection risk, and strengthens overall quality assurance within the manufacturing process.
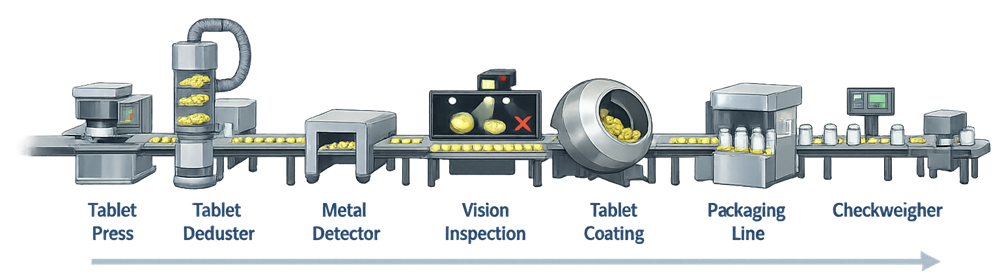
In solid dosage manufacturing, in-process inspection is commonly positioned between major processing steps such as compression, dedusting, coating, and packaging. Typical systems inspect tablets, capsules, filled containers, or packaged units.
Proper use of in-process inspection equipment supports process control, reduces batch rejection risk, and strengthens overall quality assurance within the manufacturing process.
2. Types of In-Process Inspection Equipment
Several types of inspection systems are commonly integrated into pharmaceutical manufacturing lines. Each system is designed to detect specific defects or product deviations.
2.1 Metal Detectors
Metal detectors identify metallic contamination within tablets, capsules, or packaged products. These systems generate electromagnetic fields and monitor disturbances caused by conductive particles.
Contamination sources may include worn tooling, broken machine components, or foreign materials introduced during handling. Modern pharmaceutical metal detectors are capable of detecting ferrous, non-ferrous, and stainless steel fragments at very small sizes.
When contamination is detected, the system automatically activates a reject mechanism to remove the affected unit from the product stream.
2.2 Checkweighers
Checkweighers measure the weight of individual units while they travel along a conveyor system. Units falling outside defined weight limits are automatically rejected.
Weight verification provides indirect confirmation of dosage consistency and helps detect problems such as:
- Missing tablets in bottles
- Overfilled or underfilled containers
- Improper capsule fill weight
- Mechanical feeding problems
Checkweighers also generate statistical data used for process monitoring and adjustment.
2.3 Vision Inspection Systems
Vision inspection systems use cameras and image processing software to evaluate visual characteristics of pharmaceutical products. These systems can detect defects that may not be easily visible during routine manual inspection.
ypical attributes inspected include tablet color and appearance, surface defects, chipped tablets, cracks, embossing verification, container fill levels, label presence, and packaging integrity. Vision systems provide rapid and consistent inspection capability and are widely used in automated production and packaging lines.
The illustration below presents the operating concept of an automated vision inspection system where tablets move along a conveyor while cameras and lighting systems capture images used to detect visual defects.
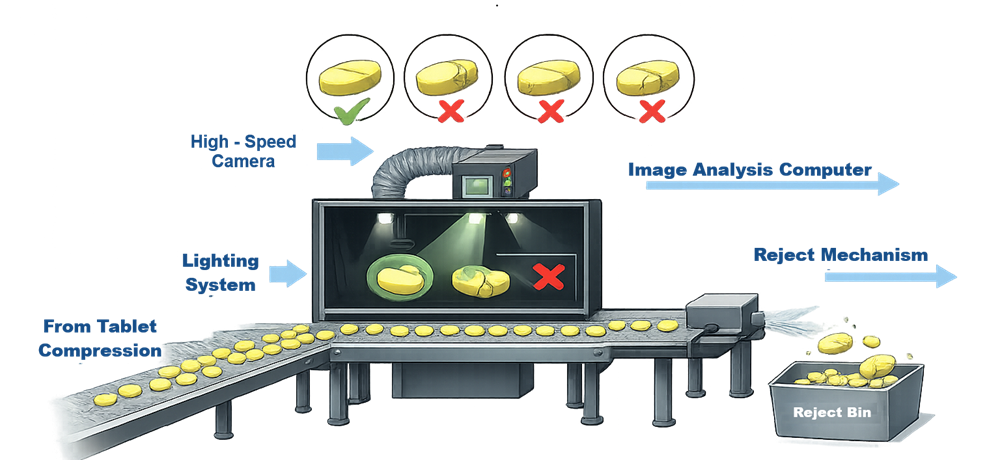
2.4 Tablet Dedusters
Tablet dedusters remove loose powder and small particles generated during compression. While their primary function is product cleaning, dedusters indirectly support inspection processes by improving product visibility and preventing dust accumulation that could interfere with downstream inspection systems.
During dedusting, tablets typically pass through vibrating spiral channels or perforated surfaces where loose powder is removed through vacuum extraction. Broken tablets or fragments may also be separated during this process.
Although dedusters are not strictly inspection devices, they play an important role in preparing tablets for downstream inspection such as metal detection or vision inspection.
The illustration below presents the principle of tablet dedusting performed immediately after compression, where loose powder and small fragments are removed before tablets proceed to downstream inspection equipment.
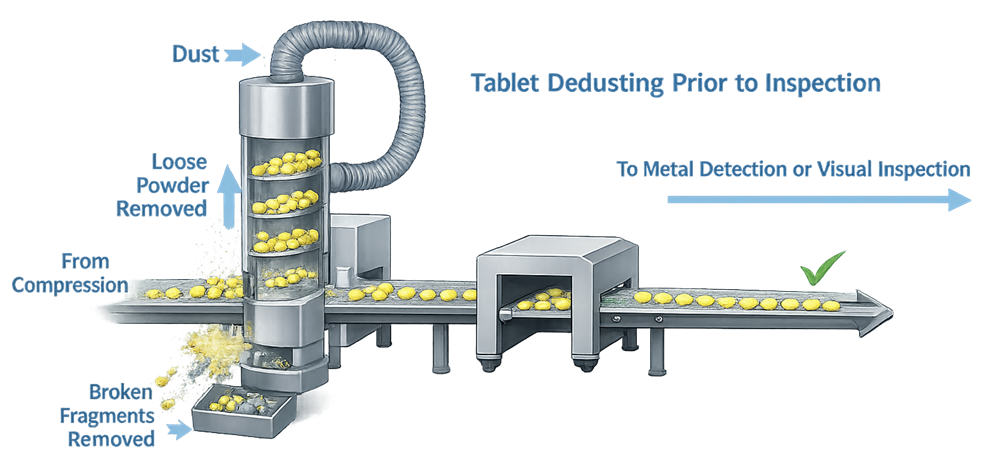
3. Visual Inspection and Sampling
Visual inspection remains an important component of pharmaceutical manufacturing control strategies. While automated inspection systems are widely used, manual inspection may still be required for certain products, process stages, or batch release activities.
The illustration below presents a typical manual inspection station used for visual examination of pharmaceutical tablets or capsules. At this workstation, trained operators inspect sampled units under controlled lighting conditions to detect visible defects such as chips, cracks, discoloration, contamination, or surface irregularities. Manual inspection stations are designed to provide consistent illumination, ergonomic positioning, and a neutral inspection background to support reliable defect detection during sample evaluation.

Manual visual inspection typically focuses on detecting visible defects including:
- Broken or chipped tablets
- Cracked capsules
- Color variation
- Surface contamination
- Improper fill levels
- Container defects or closure problems
Because manual inspection cannot practically examine every individual unit in large production batches, sampling strategies are used to evaluate product quality.
Sampling plans are typically based on statistical quality control principles. Acceptance sampling approaches such as AQL (Acceptable Quality Level) are often used to determine appropriate sample sizes and acceptance criteria. AQL-based sampling defines:
- The number of units to inspect from a batch
- The maximum allowable number of defective units within the sample
- Acceptance or rejection criteria for the batch
Sampling plans must be justified based on product risk, batch size, and defect severity. Critical defects that may impact patient safety typically require stricter acceptance limits than cosmetic defects.
For high-volume tablet or capsule production, sampling may be performed at defined intervals during manufacturing or packaging operations. These periodic inspections provide ongoing confirmation that product appearance and physical integrity remain within acceptable limits.
The illustration below presents a simplified representation of sampling-based visual inspection in which a defined number of units are selected from a production batch and evaluated according to statistically justified acceptance criteria.
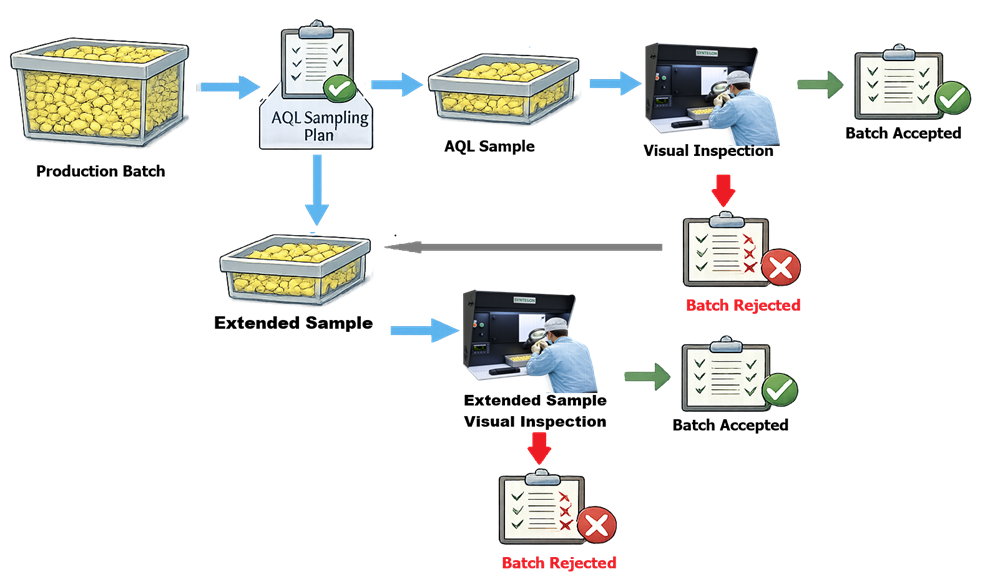
Proper visual inspection requires controlled lighting conditions, standardized inspection procedures, and trained personnel to ensure consistent defect detection.
4. Integration with Manufacturing Lines
In-process inspection equipment is typically integrated directly into manufacturing or packaging lines. These systems operate in coordination with upstream and downstream equipment to ensure smooth product flow and proper rejection of defective units.
Integration considerations include mechanical alignment with conveyors and product transfer systems, synchronization with upstream equipment such as tablet presses, and proper coordination of reject mechanisms.
Inspection systems must ensure that rejected units are physically separated from acceptable product and cannot re-enter the product stream. Reject bins, diverter gates, or pneumatic reject arms are commonly used for this purpose.
The illustration below presents a typical reject mechanism used in pharmaceutical inspection systems where defective units detected by inspection equipment are automatically diverted from the conveyor into a reject container.

Modern inspection equipment may also interface with manufacturing control systems, enabling recording of inspection results, alarm events, and production statistics.
5. Critical Performance Considerations
Several performance characteristics are critical for reliable operation of in-process inspection equipment.
Detection Capability: Inspection systems must reliably detect defects or contaminants at predefined limits. Detection thresholds must be defined based on product risk and regulatory expectations.
Reject System Performance: Reject mechanisms must remove defective units without disturbing normal product flow. Verification of reject function is typically included in routine equipment checks.
False Reject Rate: Inspection systems must balance sensitivity with operational stability. Excessive false rejects can generate unnecessary product loss and disrupt production.
Inspection Speed: Inspection capability must remain effective at the maximum production speeds expected during routine manufacturing.
Data Integrity: Many inspection systems generate electronic records including alarms, reject counts, and inspection statistics. These data may be used for process monitoring and must comply with applicable data integrity requirements where electronic systems are used.
6. Qualification and Validation
In-process inspection equipment used in pharmaceutical manufacturing must be qualified to demonstrate that it operates reliably and consistently detects defined defects or deviations.
The illustration below presents the qualification lifecycle applied to pharmaceutical inspection equipment, demonstrating the progression from installation verification through operational testing and confirmation of performance under production conditions.
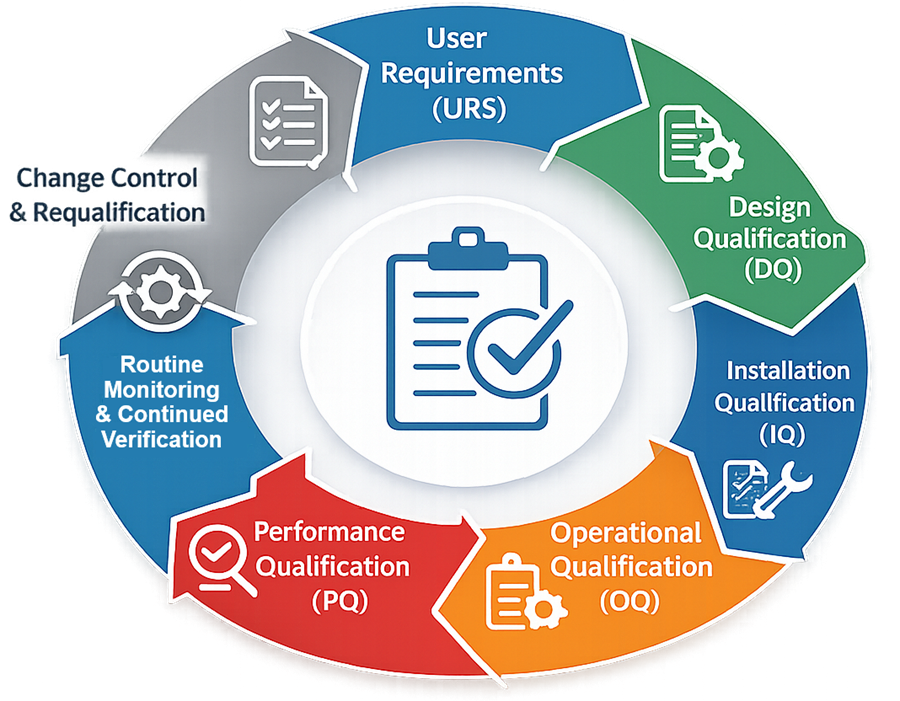
6.1 Installation Qualification
Installation Qualification verifies that the inspection equipment is installed according to manufacturer specifications and engineering design requirements. This typically includes verification of:
- Equipment identification and model configuration
- Mechanical installation and alignment with conveyors or product transfer systems
- Utility connections such as electrical power, compressed air, and vacuum
- Safety interlocks and guarding
- Control system configuration and communication interfaces
Documentation such as manuals, wiring diagrams, and calibration certificates must also be verified during installation qualification.
6.2 Operational Qualification
Operational Qualification demonstrates that the inspection equipment operates correctly across its defined operating range. Testing typically includes verification of:
- System start-up and shutdown functions
- Inspection sensor performance
- Control system operation and alarm functionality
- Reject mechanism performance
- Operator interface functions
Challenge testing is commonly performed during OQ to verify defect detection capability. Examples include:
- Metal detectors challenged with certified metal test standards
- Checkweighers challenged with calibrated weight samples
- Vision systems challenged with known defect samples such as chipped tablets or color variations
The illustration below presents challenge testing of a pharmaceutical metal detector using certified metal test samples to verify that the system reliably detects contamination and activates the reject mechanism.

The objective is to demonstrate reliable detection and proper rejection under controlled test conditions.
6.3 Performance Qualification
Performance Qualification verifies that the inspection system performs effectively during routine manufacturing conditions. PQ testing typically occurs during production runs using representative products and operating speeds. During PQ, the following aspects are evaluated:
- Consistent defect detection during normal production flow
- Correct removal of rejected units
- Absence of unacceptable false reject rates
- Stable operation at routine production speeds
Where appropriate, challenge samples may be periodically introduced during PQ runs to confirm continued detection capability.
6.4 Visual Inspection Validation
For manual visual inspection processes, validation focuses on ensuring that the inspection procedure and sampling plan are capable of detecting relevant product defects. This typically includes:
- Definition of inspection criteria and defect classifications
- Development of statistically justified sampling plans
- Qualification and training of inspectors
- Verification of inspection lighting and workstation conditions
Inspector qualification may include visual acuity testing and periodic performance evaluation using known defect samples. Sampling plans based on AQL principles must be justified to ensure that the inspection process provides adequate confidence in batch quality.
7. Summary
In-process inspection equipment provides an important control mechanism within pharmaceutical manufacturing by identifying defects and contamination during production.
Systems such as metal detectors, checkweighers, vision inspection systems, tablet dedusters, and visual inspection procedures work together to ensure that non-conforming products are detected and removed before further processing occurs.
Proper qualification and validation of inspection equipment demonstrate reliable detection capability, effective rejection of defective units, and stable operation under production conditions. When integrated into the manufacturing process and supported by appropriate sampling strategies, in-process inspection equipment plays a critical role in maintaining consistent product quality.
