Analytical Instrument Qualification Strategy (Risk-Based)
1. Introduction
Analytical instruments must be qualified to demonstrate that they are fit for their intended use. The extent of qualification required varies depending on the technical complexity of the instrument and the regulatory impact of the data it generates.
A structured, risk-based approach is required to translate instrument classification into a defined qualification strategy. This ensures that qualification activities are appropriate, consistent, and aligned with regulatory expectations without introducing unnecessary effort.
This article defines how qualification strategies are determined for analytical instruments using a risk-based framework.
2. Objective of Qualification Strategy
The objective of qualification strategy determination is to define the level, scope, and depth of qualification activities required for each analytical instrument. The strategy establishes:
- which qualification phases are required
- the extent of testing within each phase
- the level of documentation and control
- the need for software validation and data integrity controls
The outcome is a clear and justified approach that ensures the instrument operates reliably within its intended use.
3. Basis for Strategy Determination
Qualification strategy is determined using two primary inputs:
- instrument complexity
- regulatory impact of the generated data
Instrument complexity defines the technical characteristics of the system, including hardware configuration, automation, and software involvement. Regulatory impact reflects how the instrument is used, particularly whether the generated data supports GMP decisions such as product release, stability, or validation.
These two inputs are evaluated together to determine the appropriate level of qualification. The diagram below presents the decision model used to determine the appropriate qualification strategy for analytical instruments. Instrument complexity and regulatory impact are evaluated together to define the level of qualification required. This model provides a structured approach for selecting a qualification strategy that is proportional to both technical characteristics and intended use.
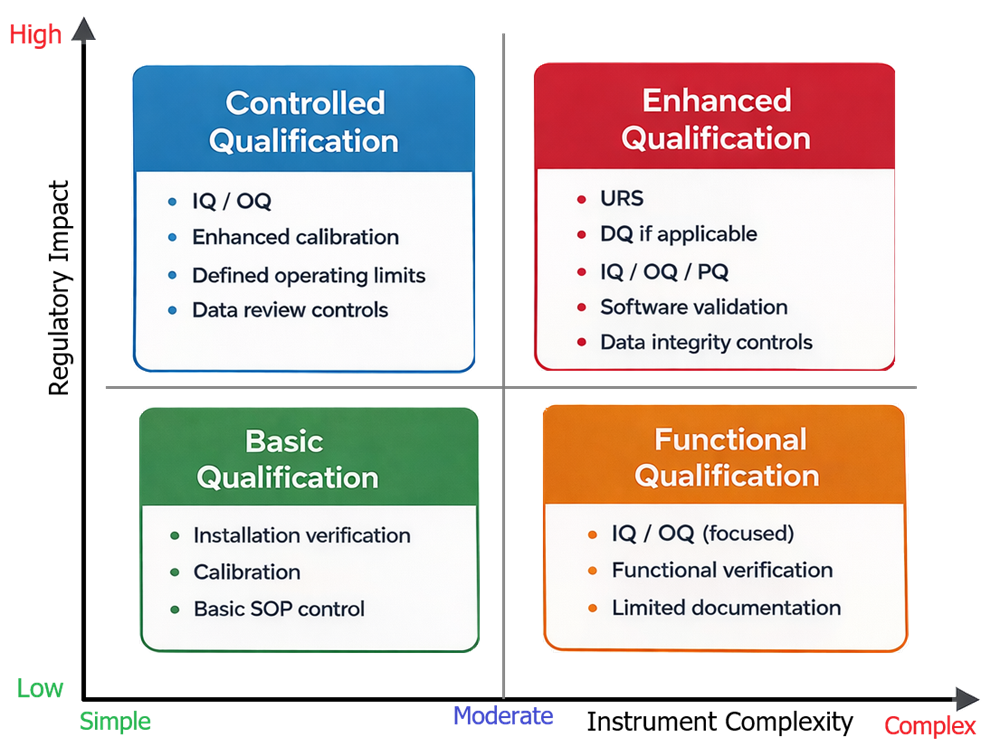
4. Qualification Strategy Models
Qualification strategies are typically defined using tiered models that align qualification effort with instrument complexity and regulatory impact. The following diagram illustrates the three qualification strategy tiers commonly applied to analytical instruments. Each tier represents an increasing level of qualification effort, testing depth, and lifecycle control based on instrument complexity and regulatory impact.
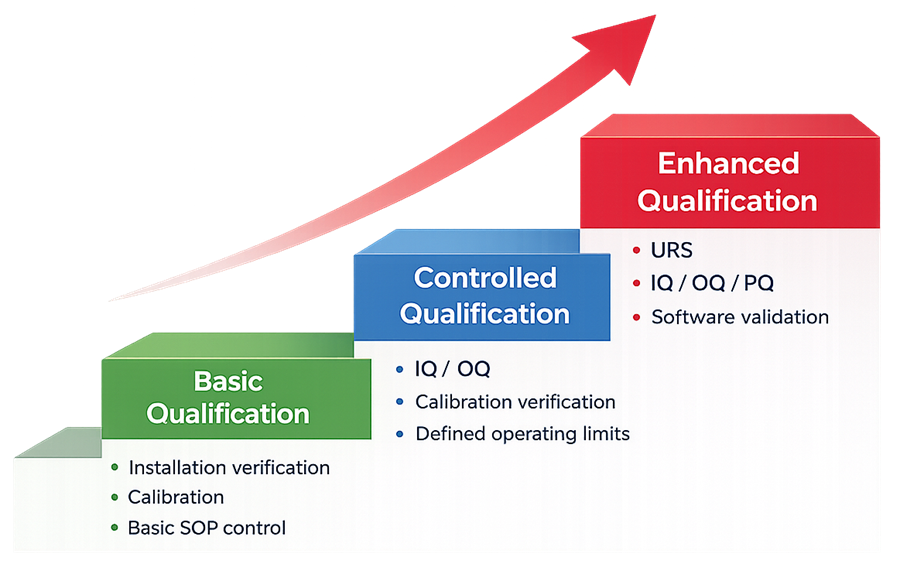
4.1 Basic Qualification Approach
Applicable to instruments with low complexity and low regulatory impact. Typical activities include:
- installation verification
- calibration
- basic functional checks
- minimal documentation
This approach is appropriate for instruments used for general laboratory support where data does not directly support regulated decisions.
4.2 Standard Qualification Approach
Applicable to instruments with moderate complexity or moderate regulatory impact. Typical activities include:
- Installation Qualification
- Operational Qualification
- defined functional testing
- documented acceptance criteria
This approach confirms that the instrument operates according to its intended function and manufacturer specifications.
4.3 Enhanced Qualification Approach
Applicable to instruments with high complexity and high regulatory impact. Typical activities include:
- User Requirements Specification
- Design Qualification where applicable
- Installation Qualification
- Operational Qualification
- Performance Qualification
- integration with analytical methods
- software validation and data integrity controls
This approach ensures that the instrument consistently performs under routine operating conditions and supports critical GMP decisions.
5. Determination of Qualification Scope
Qualification scope defines what is tested, how it is tested, and what documentation is required to demonstrate that an analytical instrument is fit for intended use. It translates the selected qualification strategy into specific, executable qualification activities. Critical functions typically include:
- measurement accuracy and precision
- control of operating parameters
- data acquisition and processing
- system suitability and performance checks
- interfaces with software or external systems
5.1 Critical Functions and Parameters
Scope begins with identification of functions and parameters that impact data quality and intended use. Only those affecting measurement reliability or regulatory decisions are considered critical and require qualification testing.
5.2 Acceptance Criteria
Acceptance criteria define objective pass or fail conditions for qualification testing. They are based on manufacturer specifications, method requirements, or regulatory expectations and must be measurable and relevant to intended use. Criteria must be:
- measurable and objective
- based on manufacturer specifications, method requirements, or regulatory expectations
- aligned with intended use of the instrument
Examples include:
- accuracy within defined tolerance
- repeatability within specified limits
- response linearity across defined range
- system suitability performance
5.3 Qualification Phases
Scope defines which qualification phases are required:
- Installation Qualification to verify correct installation and configuration
- Operational Qualification to verify functional performance across operating ranges
- Performance Qualification where applicable to verify performance under routine conditions
Selected phases must align with instrument complexity and regulatory impact.
5.4 Test Coverage
Test coverage defines the extent and depth of testing performed within each qualification phase. This includes:
- identification of test conditions and operating ranges
- definition of worst-case or boundary conditions where applicable
- selection of representative test scenarios
- inclusion of alarm and failure condition testing where relevant
Test coverage must be sufficient to demonstrate that the instrument performs reliably across its intended operating range.
5.5 Documentation Requirements
Scope defines required documentation, including protocols, test records, traceability to requirements, and final reports. Documentation must provide clear evidence of qualification.
5.6 Linkage to Calibration and Maintenance Programs
Qualification scope must align with ongoing instrument control programs to ensure sustained performance. This includes:
- calibration requirements for critical measurement components
- preventive maintenance activities
- defined intervals for routine verification
- criteria for requalification following maintenance or repair
This linkage ensures that the validated state established during qualification is maintained throughout the instrument lifecycle.
6. Qualification Deliverables
Qualification strategy defines the required documentation to demonstrate fitness for use. The diagram below illustrates the structured hierarchy of qualification deliverables generated during analytical instrument qualification. It shows how planning documents, qualification protocols, execution records, and supporting documentation are organized and linked to provide complete and traceable evidence that the instrument has been qualified for its intended use.
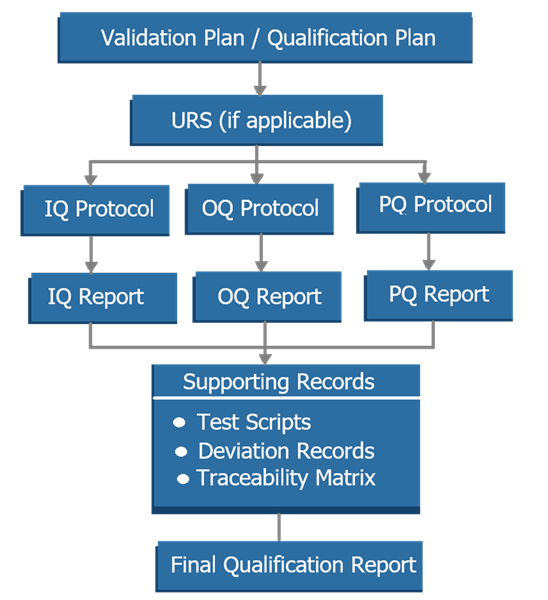
Typical deliverables include:
- qualification plan or validation plan
- User Requirements Specification where applicable
- IQ, OQ, and PQ protocols and reports
- test records and supporting data
- traceability between requirements and testing
- summary report or validation report
Documentation must provide clear evidence that the instrument meets its intended use.
7. Strategy Governance and Control
Once defined, the qualification strategy must be maintained throughout the instrument lifecycle.
Changes to the instrument, its configuration, or its intended use must be evaluated to determine whether requalification is required. Governance includes:
- change control for hardware and software modifications
- assessment of repair impact
- periodic review of instrument performance
- alignment with calibration and maintenance programs
This ensures that the instrument remains in a validated state over time.
8. Summary
Qualification strategy determination defines how analytical instruments are qualified based on their complexity and regulatory impact. By applying a structured, risk-based approach, laboratories can ensure that qualification activities are proportional, justified, and compliant.
A well-defined strategy provides clarity, consistency, and traceability, ensuring that analytical instruments generate reliable data that supports product quality and regulatory requirements.
