Instrument Risk Classification
1. Introduction
Analytical instruments used in regulated laboratories generate data that supports a wide range of activities, including product release testing, stability studies, process validation, and research and development. The regulatory significance of this data varies depending on how the instrument is used within the laboratory environment.
Because of this variability, laboratories must apply a structured method for evaluating the risk associated with each instrument. Instrument risk classification provides a systematic framework for assessing the potential impact of an instrument on product quality, patient safety, and regulatory compliance.
Risk classification ensures that qualification activities, operational controls, and lifecycle management practices are aligned with the significance of the analytical data generated by the instrument. Instruments that support critical GMP decisions require a higher level of qualification and control than instruments used for non-regulated or supporting activities.
This article describes the principles and criteria used to classify analytical instruments according to regulatory and quality risk within pharmaceutical laboratory environments.
2. Purpose of Instrument Risk Classification
Instrument risk classification establishes a structured method for evaluating the potential impact of analytical instruments on regulated laboratory operations. The purpose of this evaluation is to ensure that qualification activities and lifecycle controls are applied proportionally to the regulatory significance of the instrument.
Risk classification supports consistent decision-making across laboratory equipment by defining how the intended use of an instrument influences the level of control required throughout its lifecycle. Key objectives of instrument risk classification include:
- identifying instruments that directly support GMP quality decisions
- determining the level of qualification required for each instrument
- aligning lifecycle controls with regulatory impact
- supporting data integrity and reliability in laboratory operations
- providing a documented basis for qualification strategy decisions
By establishing a clear risk classification framework, laboratories can allocate validation resources efficiently while maintaining regulatory compliance.
3. Risk Factors Considered in Instrument Classification
Risk classification evaluates several factors that determine the potential impact of an analytical instrument on product quality and regulatory compliance. The following diagram presents the primary factors typically evaluated when determining the risk classification of analytical instruments. These factors include the intended use of the instrument, the criticality of the generated analytical data, the complexity of the instrument, and the degree of computerized system involvement. Evaluating these elements together helps laboratories determine the potential regulatory impact of the instrument.
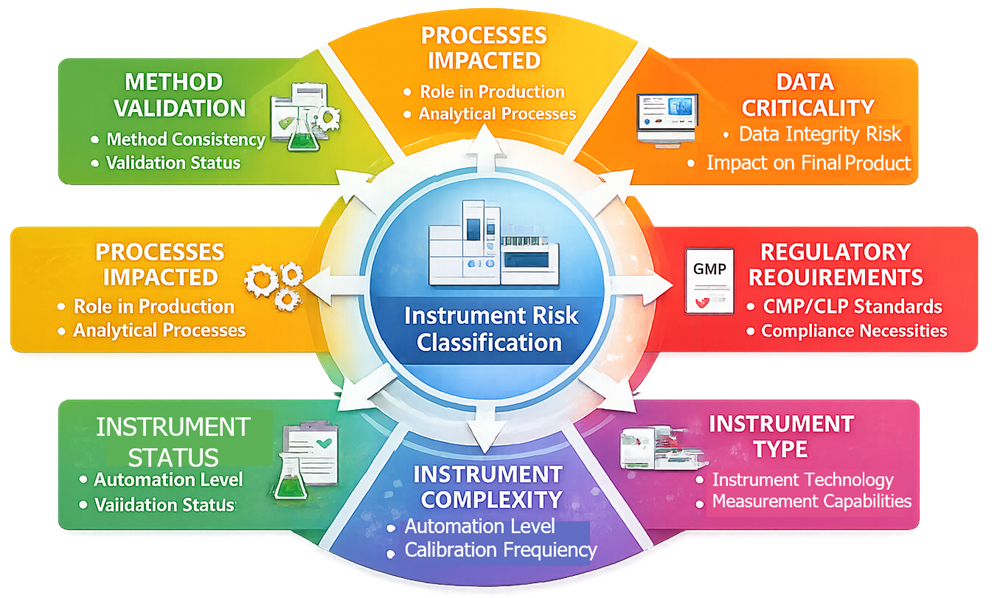
3.1 Intended Use of the Instrument
The most important factor in risk classification is the intended use of the instrument within the laboratory environment.
- Instruments used for product release testing, stability testing, or regulatory submissions generate data that directly supports GMP decisions. These instruments typically require the highest level of qualification and lifecycle control.
- Instruments used for development studies, troubleshooting investigations, or general laboratory support typically present a lower regulatory risk because the generated data does not directly determine product disposition.
3.2 Criticality of Generated Data
Risk classification must also consider the role of the analytical data produced by the instrument. Some instruments generate critical quality data used to confirm that products meet defined specifications. Examples include measurements supporting:
- assay and potency determination
- impurity analysis
- sterility or microbial testing
- stability study evaluation
Instruments producing critical analytical data require stronger controls to ensure measurement accuracy, data integrity, and traceability.
3.3 Instrument Complexity and Automation
Instrument complexity can influence the likelihood of operational errors or system failures that could affect analytical results.
Complex instruments often include multiple modules, automated sampling systems, and advanced analytical software. These systems may require additional controls such as software validation, user access management, and audit trail review. Higher system complexity can increase both operational risk and validation requirements.
3.4 Degree of Computerized System Integration
Many analytical instruments rely on computerized systems for data acquisition, processing, and storage. Instruments that generate electronic records may require compliance with regulatory expectations related to electronic data management and data integrity. Risk classification should therefore evaluate whether the instrument:
- contains embedded software or firmware
- uses external data acquisition software
- stores analytical data electronically
- integrates with laboratory data management systems
Instruments with extensive computerized functionality typically require additional validation and data integrity controls.
4. Typical Risk Classification Levels
Laboratories commonly group analytical instruments into risk categories that reflect their regulatory significance. Although specific classification models may vary between organizations, a three-level risk structure is widely used. The diagram below illustrates the general risk classification model used to group analytical instruments according to their regulatory impact. Instruments are typically classified into three levels based on the significance of the analytical data they generate and the role of that data in product quality decisions. This classification framework supports consistent determination of qualification scope and lifecycle control expectations.
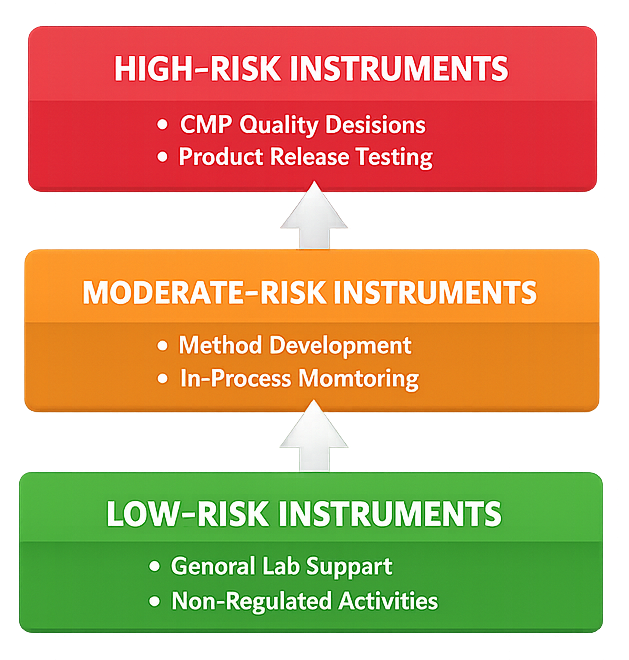
4.1 High-Risk Instruments
High-risk instruments generate data that directly supports critical GMP decisions affecting product quality and patient safety. Examples may include:
- chromatography systems used for release testing
- stability testing instruments used for regulatory submissions
- instruments supporting impurity or potency determination
These instruments require comprehensive qualification, strict lifecycle control, and strong data integrity protections.
4.2 Moderate-Risk Instruments
Moderate-risk instruments support GMP laboratory operations but do not directly determine final product disposition. Examples may include:
- instruments used for in-process monitoring
- equipment used during method development prior to validation
- instruments supporting process characterization studies
These instruments typically require formal qualification but may operate under less stringent lifecycle controls compared with high-risk systems.
4.3 Low-Risk Instruments
Low-risk instruments support general laboratory activities and do not generate data used to support regulated quality decisions. Examples may include:
- instruments used for training activities
- equipment used for preliminary research
- general laboratory support devices
These instruments typically require basic installation verification and calibration but may not require full qualification activities.
5. Application of Risk Classification in Laboratory Governance
Once instruments are assigned a risk classification, the classification guides several aspects of laboratory governance and validation planning. Risk classification influences:
- qualification scope and testing depth
- documentation requirements for IQ, OQ, and PQ
- level of software validation required
- data integrity control expectations
- calibration and maintenance practices
- requalification requirements following changes or repairs
By applying a consistent risk classification model, laboratories can ensure that qualification activities are aligned with the regulatory significance of each instrument.
6. Integration of Risk Classification with Instrument Categorization
Instrument categorization and instrument risk classification address two different characteristics of analytical equipment.
Instrument categorization describes the technical complexity of the instrument, while risk classification evaluates the regulatory importance of the data generated by the instrument. These two elements are evaluated together when determining the appropriate qualification strategy.
An instrument with high technical complexity does not automatically require the highest level of qualification. The required qualification effort depends on how the instrument is used within the laboratory environment.
The diagram below presents a decision model showing how instrument complexity and regulatory risk are evaluated together to determine the appropriate level of qualification. Instrument complexity reflects the technical characteristics of the system, while regulatory risk reflects the impact of the generated data on product quality and compliance. The intersection of these two dimensions defines the required qualification depth and lifecycle control.
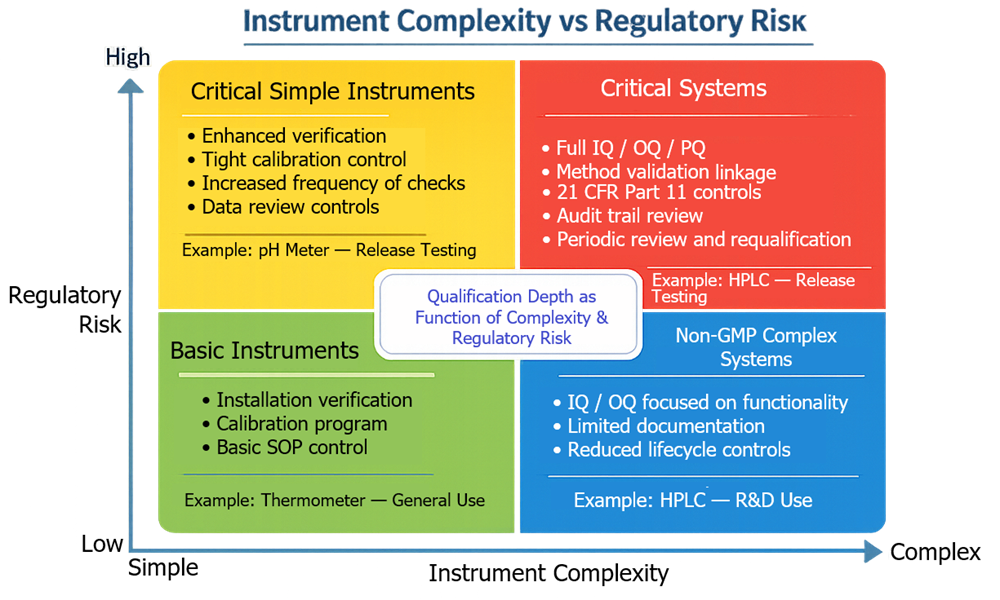
For example, a chromatography system used for GMP release testing represents both high system complexity and high regulatory risk. In this case, extensive qualification and strict lifecycle control are required.
The same type of system used exclusively for non-GMP research may require a reduced qualification scope because the generated data does not directly support regulated quality decisions.
Evaluating both instrument complexity and regulatory impact allows laboratories to apply qualification activities that are proportional to the role of the instrument within the overall quality system.
7. Structured Risk Assessment Methods
Instrument risk classification should be supported by a documented risk assessment process. Structured risk assessment methods provide a systematic and repeatable approach for evaluating how instrument failures could affect analytical data, product quality decisions, and regulatory compliance.
Risk assessment approaches commonly used in pharmaceutical laboratories are based on the principles described in ICH Q9 Quality Risk Management. These methods allow laboratories to evaluate potential failure modes and determine whether additional qualification controls are required. One commonly used method is Failure Modes and Effects Analysis. In this approach, potential instrument failures are evaluated according to several factors, typically including:
- severity of impact on product quality or analytical data integrity
- probability of occurrence of the failure
- ability to detect the failure before data is reported
These factors are evaluated using defined scoring criteria. The combined risk score helps determine the overall risk classification of the instrument and supports justification of the qualification strategy.
Structured risk assessment ensures that instrument risk classification is performed consistently and that qualification activities remain proportional to the potential impact of instrument failure on regulated laboratory operations.
8. Summary
Instrument risk classification provides a structured framework for evaluating the regulatory significance of analytical instruments used in laboratory environments. By assessing intended use, data criticality, instrument complexity, and computerized system involvement, laboratories can determine the level of qualification and lifecycle control required for each instrument.
Risk classification ensures that validation resources are focused on instruments that generate data critical to product quality and regulatory compliance while maintaining efficient governance of laboratory equipment across the organization.
