Moist Heat Sterilization Principles
Moist heat sterilization using saturated steam remains the most established and defensible method of terminal sterilization in pharmaceutical and biotechnology manufacturing. When properly engineered and controlled, steam sterilization provides predictable lethality, reliable penetration, and a high degree of sterility assurance.
This article defines the scientific foundation and control strategy that support validated steam sterilization systems.
1. Scientific Basis of Steam Sterilization
1.1 Saturated Steam and Latent Heat Transfer
Sterilization effectiveness depends on saturated steam, not simply elevated temperature.
When saturated steam contacts a cooler surface, it condenses. During condensation, latent heat is released rapidly and uniformly. This phase change delivers significantly more energy than dry heat at the same temperature, enabling efficient microbial destruction. Key principle:
Sterilization occurs through condensation, not through dry heating.
Superheated steam behaves like dry heat and is less effective for sterilization purposes.
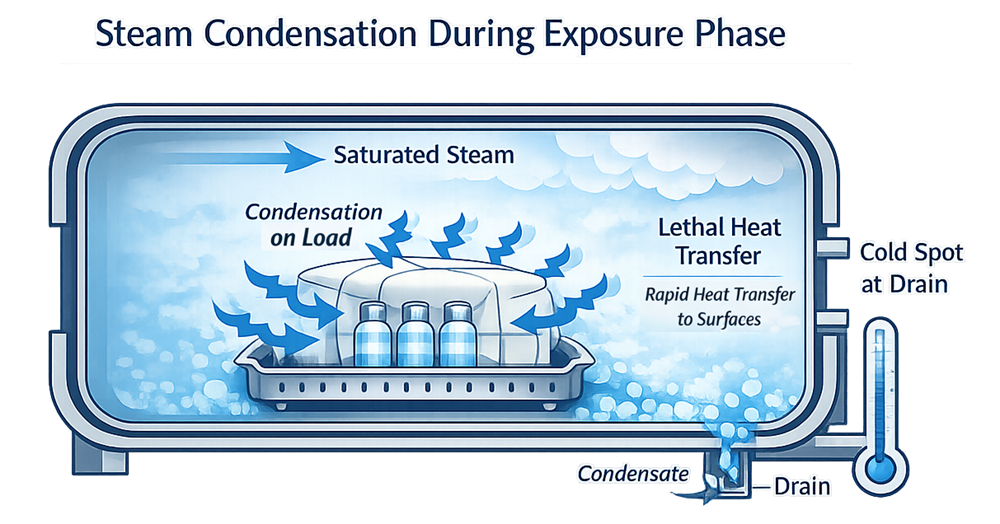
1.2 Air Removal and Steam Penetration
Air is the primary obstacle to effective sterilization.
Air pockets:
- Prevent steam contact
- Reduce heat transfer
- Create cold spots
- Compromise lethality
Common air removal methods include:
- Gravity displacement
- Pre-vacuum cycles
- Steam flush pressure pulse systems
Effective air removal is a prerequisite for uniform lethality across the load.
1.3 Temperature–Time Relationship and Lethality
Microbial destruction follows first-order kinetics. The lethality delivered during a cycle depends on both temperature and exposure time.
The reference temperature for moist heat sterilization is: 121.1 °C
Lethality is expressed as F₀, defined as the equivalent exposure time at 121.1 °C with a z-value of 10 °C.
Where:
- T = measured temperature
- z = temperature change required for tenfold reduction in D-value
F₀ provides a standardized way to compare cycles.
2. Steam Quality Requirements
Steam used for sterilization must meet defined physical characteristics.
2.1 Dryness Fraction
Steam must be sufficiently dry to:
- Prevent excessive condensate accumulation
- Maintain effective heat transfer
- Avoid load wetting
Typical expectation: dryness fraction ≥ 0.95
2.2 Non-Condensable Gases
Non-condensable gases reduce heat transfer efficiency and interfere with sterilization. Excessive NCG levels:
- Create cold spots
- Delay heating
- Reduce lethality
Routine monitoring of NCG levels supports validated performance.
2.3 Superheat
Steam must not be significantly superheated. Superheated steam:
- Does not condense immediately
- Delivers less effective heat transfer
- Behaves closer to dry heat
Proper steam conditioning ensures saturated conditions at the point of use.
Methods of Verifying Steam Saturation
1. Temperature–Pressure Relationship
For saturated steam, chamber temperature and pressure follow a defined thermodynamic relationship. Deviation between measured pressure and temperature beyond expected saturation tables may indicate:
- Superheat
- Excess non-condensable gases
- Instrumentation error
Routine comparison of recorded temperature and pressure during exposure phase provides indirect confirmation of saturated conditions.
2. Steam Quality Testing
Formal steam quality testing evaluates:
- Dryness fraction
- Non-condensable gases
- Degree of superheat
These tests are typically performed:
- During initial qualification
- After major steam system modification
- During periodic requalification
Standards reference: ISO 17665 and EN 285 testing methods.
3. Steam Conditioning Design Controls
Engineering design elements that minimize superheat include:
- Proper steam traps
- Steam separators
- Correct piping slope
- Avoidance of excessive pressure drop
- Proximity of steam source to sterilizer
Design control is as important as testing.
Practical Validation Considerations: Steam Condition and Load Thermal Response
Saturated steam is a fundamental requirement for effective moist heat sterilization. In well-designed pharmaceutical facilities, supply steam superheat is uncommon but remains a potential risk that must be assessed during qualification.
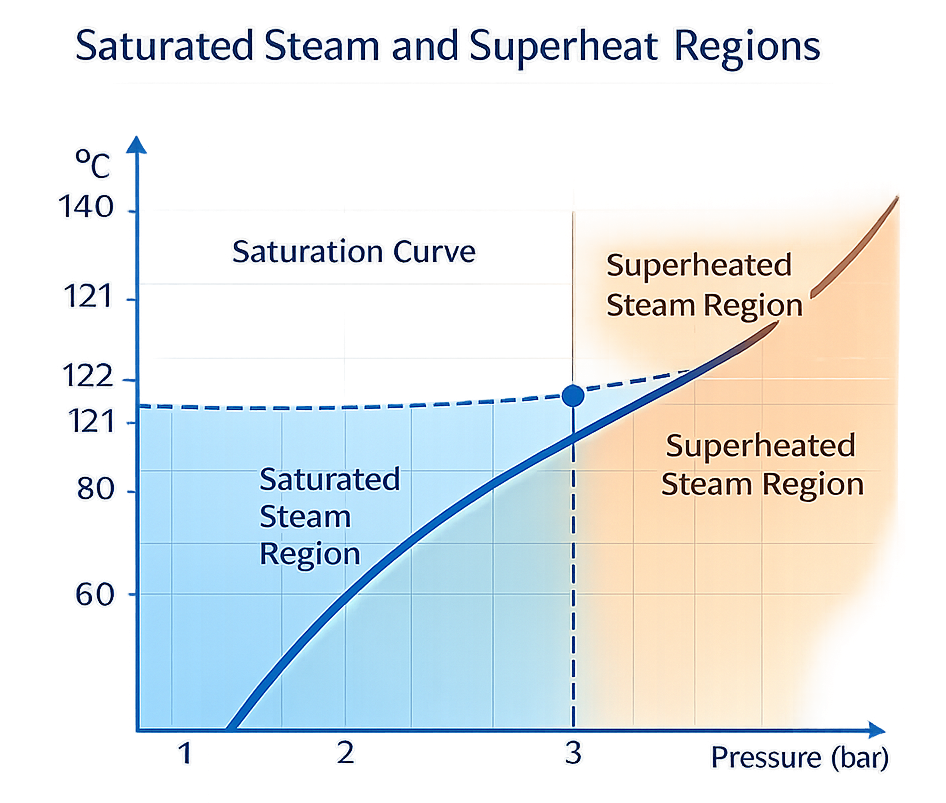
Utility-Level Steam Superheat
True superheated steam originates at the utility level and may result from:
- Excessive pressure drop
- Improper steam conditioning
- Inadequate separator performance
- Piping design deficiencies
Superheat is typically identified during formal steam quality testing performed during installation qualification or major requalification activities. It is not continuously monitored during routine cycles but must be documented and justified as part of system qualification.
Deviation between measured chamber temperature and pressure from expected saturation tables may indicate superheat or excessive non-condensable gases and requires investigation.
Load-Dependent Thermal Behavior
In addition to utility-level considerations, certain load configurations may exhibit localized thermal behavior that differs from simple saturated steam expectations. Porous or highly adsorptive materials such as:
- Textile packs
- Gauze
- Dense porous assemblies
may demonstrate localized temperature elevations due to:
- Steam condensation within the matrix
- Heat retention characteristics
- Limited evaporative cooling
This phenomenon is not equivalent to supply steam superheat. It reflects load heat transfer dynamics. Such behavior is typically detected during:
- Load development studies
- Thermocouple mapping
- Performance qualification runs
Temperature elevations within the load do not necessarily compromise sterilization and may increase lethality margin. However, they must be:
- Scientifically evaluated
- Documented
- Confirmed not to adversely affect product integrity or packaging performance
Validation Position
Steam condition and load thermal response are both evaluated during qualification. Steam quality testing confirms appropriate utility performance, while load development and PQ studies characterize load-specific thermal behavior.
Sterilization validation must demonstrate:
- Effective air removal
- Saturated steam exposure
- Uniform heat distribution
- Adequate lethality at the coldest location
Understanding the distinction between steam supply condition and load heat dynamics is essential for defensible validation.
3. Critical Engineering Controls
Steam sterilization systems rely on defined engineering controls to maintain repeatable performance.
3.1 Vacuum Integrity
Pre-vacuum systems require:
- Vacuum leak rate testing
- Confirmed chamber tightness
- Documented acceptance criteria
Poor vacuum integrity compromises air removal.
3.2 Drain Temperature Monitoring
The chamber drain is typically the coldest location during sterilization. Monitoring drain temperature:
- Confirms minimum exposure conditions
- Provides worst-case reference
- Supports lethality calculation
- 3.X Verification of Saturated Steam Conditions
- Saturated steam is a prerequisite for effective moist heat sterilization. The control strategy must therefore include mechanisms to ensure that steam entering the chamber is not significantly superheated.
3.3 Verification of Saturated Steam Conditions
Saturated steam is a prerequisite for effective moist heat sterilization. The control strategy must therefore include mechanisms to ensure that steam entering the chamber is not significantly superheated.
3.4 Control System Logic
Validated sterilizers must include:
- Defined cycle phases
- Interlocks preventing premature exposure
- Alarm functionality
- Time and temperature recording
- Data retention controls
Control systems must prevent release of incomplete or failed cycles.
4. Microbiological Considerations
4.1 Biological Indicators
Biological indicators demonstrate sterilization margin beyond measured temperature data. Common organism: Geobacillus stearothermophilus
BI placement targets:
- Cold spots
- Worst-case penetration points
- Densely packed load areas
Engineering data establishes capability. Biological indicators confirm lethality margin.
5. Regulatory and Standards Framework
Steam sterilization principles align with established regulatory expectations:
- 21 CFR 211.113
- EU GMP Annex 1
- ISO 17665
- PDA Technical Report No. 1
These standards require:
- Scientifically justified cycle development
- Documented qualification
- Ongoing monitoring
- Change impact assessment
6. Relationship to Qualification
Understanding these principles is essential before:
- Developing sterilization loads
- Performing thermocouple mapping
- Executing qualification protocols
- Establishing requalification scope
Qualification verifies performance. Principles explain why performance occurs.
Without engineering understanding, validation becomes procedural rather than scientific.
