Pharmaceutical Water System Design and Distribution
Pharmaceutical water systems must be intentionally engineered to consistently deliver water that meets its defined quality attributes at the point of use. Design and distribution are not aesthetic or convenience decisions. They are primary control mechanisms that determine whether the system can maintain a sustained state of control.
Regulatory expectations are clear: water system design must be appropriate for its intended use and capable of preventing chemical, microbiological, and endotoxin contamination over the full operating lifecycle.
Design Driven by Intended Use
The selected water quality category establishes the minimum design requirements for the system. Systems intended to produce Purified Water and Water for Injection differ in complexity, operating conditions, and risk profile, but the governing principle is the same. Design must:
- Support the required water quality at all points of use
- Minimize the potential for contamination and biofilm formation
- Allow effective sanitization
- Enable monitoring and control of critical parameters
Designing beyond intended use adds unnecessary complexity. Designing below it creates compliance risk.
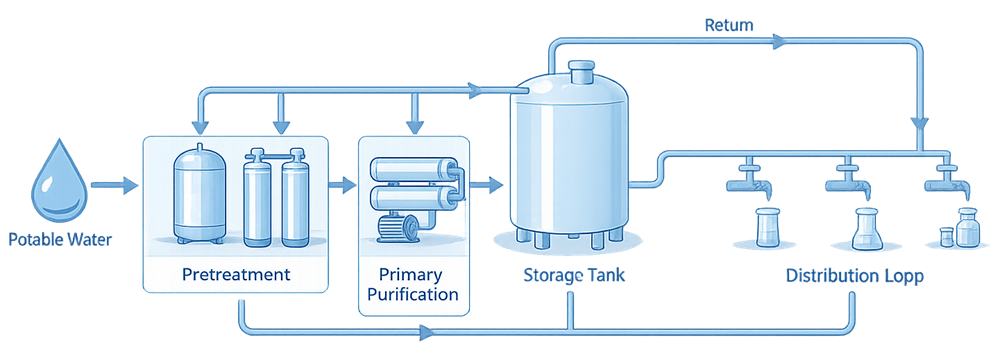
Source Water and Pretreatment
All pharmaceutical water systems start with potable water. Pretreatment is required to ensure consistent feed quality and protect downstream purification technologies. Typical pretreatment elements include:
- Particulate removal
- Hardness control
- Chlorine or chloramine reduction
- Organic load reduction
Pretreatment design is often underestimated. Poor pretreatment is a common root cause of membrane fouling, microbial instability, and shortened system life.
Primary Purification Technologies
Primary purification removes dissolved salts, organic compounds, and microorganisms to meet the applicable pharmacopeial requirements. Common technologies include:
- Reverse osmosis
- Electrodeionization or deionization
- Distillation for WFI systems where applicable
The following image provides a representative example of a skid-mounted pharmaceutical water purification and storage assembly.

The image below provides a representative example of a skid-mounted Water for Injection generation system used in GMP manufacturing environments.

Technology selection must be justified based on:
- Required water quality
- Feed water characteristics
- Operational reliability
- Sanitization compatibility
The objective is not maximum purity. The objective is reliable compliance.
Storage System Design
Storage tanks are integral parts of the system, not passive vessels. Key design expectations include:
- Materials compatible with the water quality
- Smooth, sanitary internal surfaces
- Vent filtration
- Sloped bottoms for complete drainage
- Integration with circulation and sanitization strategies
Poorly designed storage tanks are frequent contributors to microbial excursions.
Distribution Loop Design
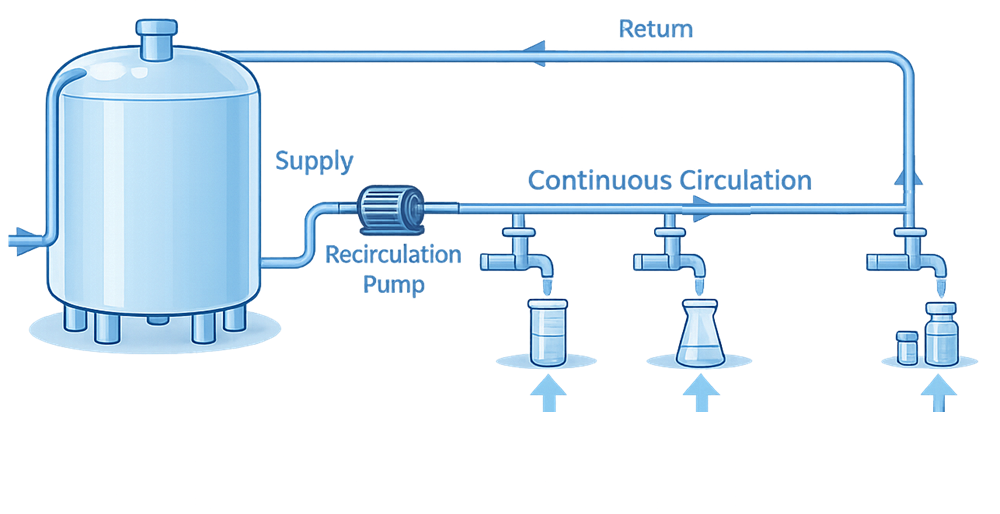
Distribution is where many systems fail, even when purification is sound. A compliant distribution system:
- Operates in continuous circulation
- Maintains adequate velocity to limit stagnation
- Minimizes dead legs
- Uses sanitary components and fittings
- Supports effective sanitization
Distribution design must ensure that water quality at the most remote point of use is equivalent to that at the generation point.
Materials of Construction
Material selection is driven by water quality, sanitization method, and long-term system stability. Common expectations include:
- Sanitary stainless steel or qualified polymeric materials
- Orbital welding where applicable
- Documented surface finish
- Compatibility with chemical or thermal sanitization
Material choices must be defendable, documented, and consistent throughout the system.
Sanitization Strategy as a Design Input
Sanitization is not an afterthought. It must be embedded into system design. Acceptable strategies include:
- Thermal sanitization
- Chemical sanitization
- Combination approaches
Design must allow complete and effective sanitization of:
- Generation equipment
- Storage tanks
- Distribution piping
- Points of use
Systems that cannot be effectively sanitized cannot be reliably controlled.
Control and Monitoring Integration
Design must support monitoring of critical quality and operating parameters, including:
- Conductivity or resistivity
- TOC
- Temperature
- Flow and circulation status
Instrumentation placement and accessibility are part of design justification, not later optimization.
Common Design Deficiencies
Recurring industry issues include:
- Dead legs justified on paper rather than eliminated
- Inadequate circulation velocity
- Sanitization methods incompatible with materials
- Storage tanks treated as non-critical components
- Distribution complexity exceeding operational discipline
These deficiencies are routinely cited during inspections because they are avoidable.
Summary
Pharmaceutical water system design and distribution are foundational to compliance. A well-designed system makes monitoring straightforward, qualification efficient, and long-term control achievable. A poorly designed system cannot be rescued through testing alone.
Design must be fit for intended use, operationally simple, and inherently controllable. That principle has not changed, and it continues to define successful pharmaceutical water systems.
