Preventive Maintenance and System Reliability Strategy
Preventive maintenance is a structured control program designed to preserve equipment reliability, maintain validated state, and prevent unplanned failures that may affect product quality or GMP system performance.
Purpose and Scope
In GMP-regulated environments, equipment and utility systems must operate consistently within validated parameters. Mechanical wear, component degradation, environmental exposure, and operational stress introduce performance drift over time. Without structured maintenance controls, validated systems gradually lose reliability and may operate outside intended design conditions.
Preventive maintenance provides a systematic approach to:
- Preserve equipment functionality
- Reduce failure probability
- Maintain control of critical process parameters
- Support sustained qualification status
- Protect product quality and environmental control
This article defines the framework for establishing, executing, and evaluating a preventive maintenance program aligned with lifecycle validation principles.
The scope includes:
- Production equipment
- Utility systems
- HVAC systems
- Environmental monitoring infrastructure
- Laboratory equipment
- Automation and control systems
Regulatory and Quality System Alignment
Preventive maintenance expectations are embedded within GMP and quality system regulations. Applicable U.S. regulatory references include:
- 21 CFR 211.67 – Equipment cleaning and maintenance
- 21 CFR 211.68 – Equipment controls to assure proper performance
- 21 CFR 211.100 – Written procedures and adherence
- 21 CFR 820.70 – Production and process controls
- 21 CFR 820.72 – Inspection and measuring equipment control
These regulations require:
- Written maintenance procedures
- Documented execution
- Equipment suitability for intended use
- Prevention of contamination or performance degradation
- Control of systems influencing product quality
Preventive maintenance is therefore a regulatory expectation, not an optional reliability initiative.
Preventive Maintenance Program Structure
Preventive maintenance must be integrated into the equipment and utility lifecycle rather than treated as a standalone operational task. The following model illustrates how maintenance events interact with qualification, ongoing GMP operation, verification activities, and periodic program review to preserve validated state.
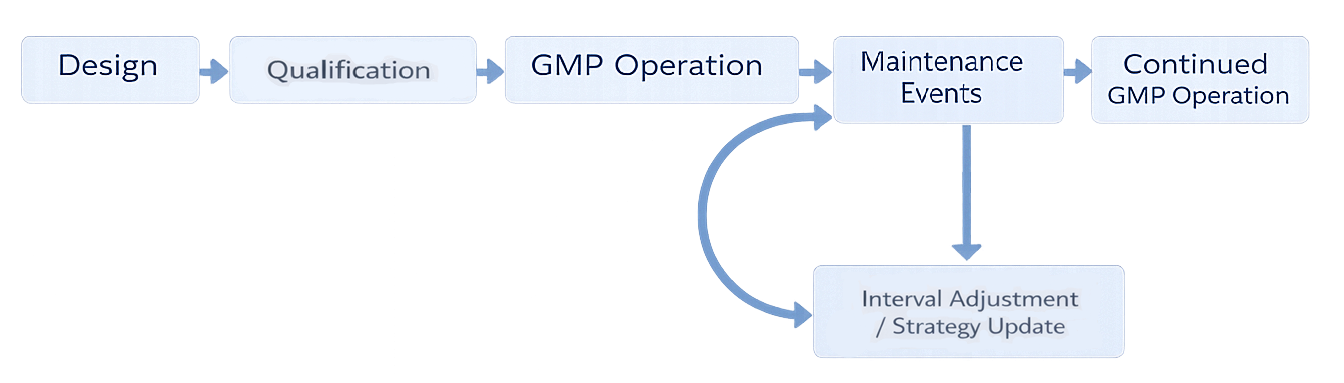
A compliant maintenance program includes:
- Equipment inventory and classification
- Defined maintenance tasks and frequencies
- Risk-based criticality assessment
- Documented procedures and work instructions
- Execution documentation and review
- Verification of system readiness after maintenance
- Periodic program effectiveness review
Each equipment asset should be evaluated for:
- Product impact
- Process criticality
- Failure consequences
- Historical reliability
- Redundancy
- Environmental exposure
Critical systems require more structured maintenance controls and documentation depth.
Risk-Based Maintenance Strategy
Maintenance frequency must reflect risk, not convenience. Factors influencing maintenance intervals include:
- Manufacturer recommendations
- Operational hours or cycle count
- Environmental stress
- Historical failure patterns
- Consequence of failure
- Detection capability
Risk-based maintenance aligns task frequency with:
- Probability of failure
- Severity of impact
- Ability to detect degradation
Over-maintenance introduces unnecessary intervention risk. Under-maintenance increases failure probability. A balanced program relies on documented risk evaluation.
Maintenance intervals should be established through structured risk evaluation rather than fixed default schedules. The following model illustrates how operational, environmental, and failure data are integrated to determine and periodically reassess maintenance frequency.
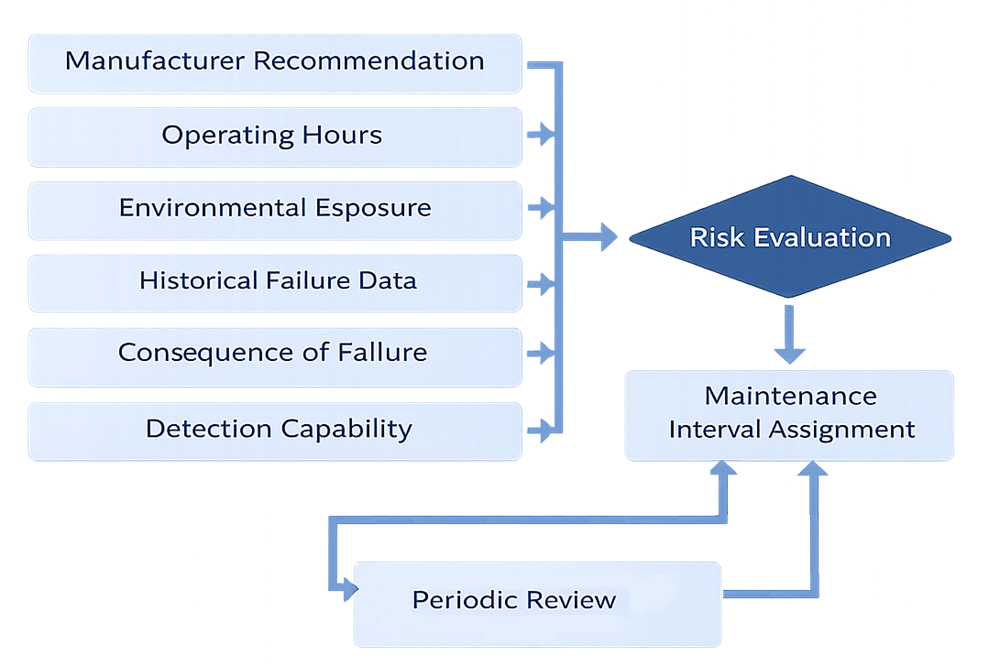
Maintenance Classification and Impact Level
Not all maintenance activities carry equal qualification impact. Maintenance tasks may be categorized as:
Routine / Low Impact
- Lubrication
- Visual inspection
- Filter replacement where design is unchanged
- Minor adjustments not affecting critical parameters
Typically requires:
- Documentation
- Verification of normal operation
Moderate Impact
- Component replacement
- Sensor cleaning
- Control adjustments
- Minor mechanical repairs
May require:
- Functional verification
- Limited re-testing
High Impact
- Critical component replacement
- Control system modification
- Motor or pump replacement
- HVAC component replacement affecting airflow or pressure
Requires:
- Formal impact assessment
- Defined requalification scope
- Documented release decision
Maintenance must always trigger evaluation of qualification status.
Maintenance Impact Assessment
Before returning equipment to GMP use, the organization must determine whether maintenance affects validated state. Impact assessment should evaluate:
- Change to design intent
- Change to operating range
- Change to critical process parameters
- Change to alarm limits
- Change to control logic
- Risk to environmental classification
The outcome may determine:
- Verification only
- Partial requalification
- Full requalification
Failure to formally assess maintenance impact is a common inspection observation.
Preventive maintenance activities do not carry equal qualification impact. The following model illustrates how maintenance events are classified by impact level and how that classification determines the required scope of verification or requalification prior to return to GMP operation.
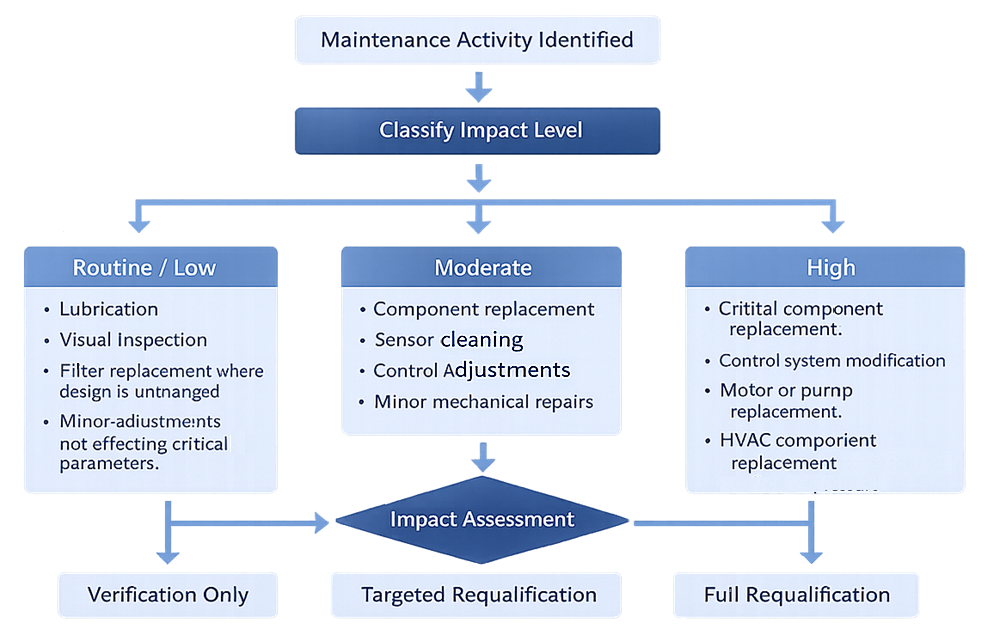
Post-Maintenance Verification and Requalification
Following maintenance, systems must not automatically return to service. Post-maintenance verification may include:
- Calibration confirmation
- Functional testing
- Alarm verification
- Environmental parameter checks
- Utility performance confirmation
Where risk dictates, requalification may include:
- OQ repeat testing
- Targeted PQ testing
- Airflow or pressure cascade verification
- Utility sampling
The depth of verification must be proportional to risk and documented in a structured decision process.
Interface with Change Control
Maintenance that alters configuration, design, or performance parameters must enter formal change control. Triggers for change control include:
- Replacement with different model or specification
- Software or firmware updates
- Control logic modifications
- Design improvement implementation
Maintenance and change control must operate in coordination.
Uncontrolled modification undermines validated state.
Periodic Review and Program Effectiveness
Preventive maintenance programs require periodic evaluation to confirm effectiveness. Periodic review should evaluate:
- Failure frequency
- Repeat maintenance events
- Unplanned downtime
- Deviation trends
- Equipment replacement patterns
- CAPA linkage
Program review supports:
- Interval adjustment
- Strategy refinement
- Asset retirement decisions
- Continuous improvement
Maintenance effectiveness is demonstrated by stability and absence of repeat failures.
Lifecycle Control Summary
Effective system reliability governance requires:
- Structured maintenance planning
- Risk-based task frequency
- Defined impact assessment
- Post-maintenance verification
- Change control integration
- Periodic performance review
Preventive maintenance preserves validated state and protects GMP system integrity. When managed systematically, it supports sustained operational reliability and regulatory compliance.
