Process Qualification
Process Validation is a lifecycle-based activity performed to demonstrate that a manufacturing process is capable of consistently producing product that meets predefined quality attributes, specifications, and regulatory requirements. In regulated pharmaceutical and medical device environments, process validation provides documented evidence that the process is understood, controlled, and suitable for its intended use.
Process validation is not a single event. It is an ongoing activity that spans process design, qualification, and continued monitoring throughout the commercial lifecycle.
Purpose of Process Validation
The primary purpose of process validation is to establish and maintain a state of control over the manufacturing process. By demonstrating that critical process parameters are identified, understood, and controlled, process validation supports product quality, patient safety, and regulatory compliance.
Effective process validation reduces variability, prevents defects, and enables reliable and reproducible manufacturing outcomes.
Process Validation Lifecycle
Process validation is typically structured into three lifecycle stages.
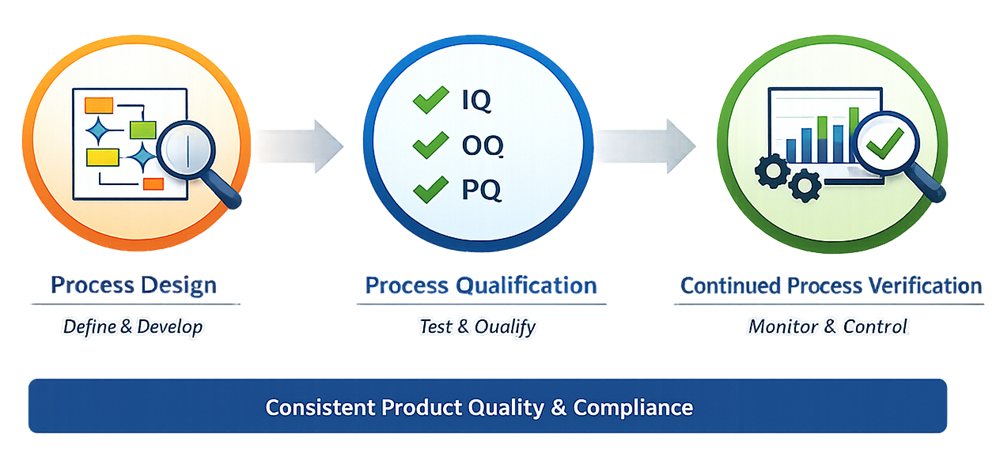
Process Design
Process Design focuses on developing process understanding and defining how the process will operate to consistently deliver acceptable product quality. Activities in this stage include:
- Defining the process flow and sequence of operations
- Identifying critical process parameters and critical quality attributes
- Establishing operating ranges and control strategies
- Assessing risks associated with process variability and failure modes
Process Design establishes the scientific and technical foundation for downstream qualification and monitoring activities.
Process Qualification
Process Qualification confirms that the process design can be successfully implemented in the intended manufacturing environment and that the process performs as expected under defined conditions.
Process Qualification typically includes:
Installation Qualification (IQ)
Verification that equipment, facilities, and utilities used in the process are installed correctly and in accordance with approved design and specifications.
Operational Qualification (OQ)
Verification that equipment and systems operate as intended within defined operating ranges, including testing of controls, alarms, and critical functions.
Performance Qualification (PQ)
Demonstration that the integrated process consistently produces product meeting predefined acceptance criteria when operated under routine or representative conditions.
Successful completion of Process Qualification provides objective evidence that the process is capable of controlled operation.
Continued Process Verification
Continued Process Verification (CPV) ensures that the process remains in a state of control throughout routine manufacturing. This stage involves:
- Ongoing monitoring of critical process parameters
- Review and trending of process performance data
- Evaluation of deviations, shifts, or adverse trends
- Periodic assessment of process capability and stability
CPV supports early detection of process drift and enables timely corrective and preventive actions.
Tools Supporting Process Validation
Process validation activities are supported by a range of analytical and quality tools, selected based on process complexity and risk.
Commonly used tools include:
- Process flow diagrams to visualize process steps and controls
- Risk assessments to identify and prioritize potential failure modes
- Statistical methods to assess variability, capability, and trends
- Defined performance metrics to evaluate process effectiveness
- Monitoring and control systems to maintain process stability
The selection and application of tools should be appropriate to the specific process and aligned with regulatory expectations.
Process Validation Documentation
Process validation is documented through a structured set of protocols, reports, and supporting records that may include:
- Process validation plans
- Qualification protocols and reports
- Risk assessments and control strategies
- Data analyses and performance summaries
- Continued process verification reports
Documentation provides objective evidence of process understanding, execution, and control.
Regulatory Context
Process validation activities are guided by applicable regulations and regulatory guidance, including FDA expectations for lifecycle-based validation. Validation approaches should be risk-based, science-driven, and tailored to the specific process and product.
Summary
Process Validation establishes and maintains confidence that a manufacturing process is capable of consistently delivering product that meets quality and regulatory requirements. When implemented as a lifecycle activity, process validation supports robust process control, continuous improvement, and sustained compliance throughout commercial production.
