Pyrogens and Endotoxins
1. What Is a Pyrogen?
A pyrogen is any substance that can cause fever when introduced into the human body.
In pharmaceutical manufacturing, pyrogens are a concern primarily for injectable and implantable products. If present, they can trigger fever, inflammation, hypotension, or in severe cases, shock. Not all pyrogens are the same. The most important type in pharmaceutical manufacturing is bacterial endotoxin.
2. What Is an Endotoxin?
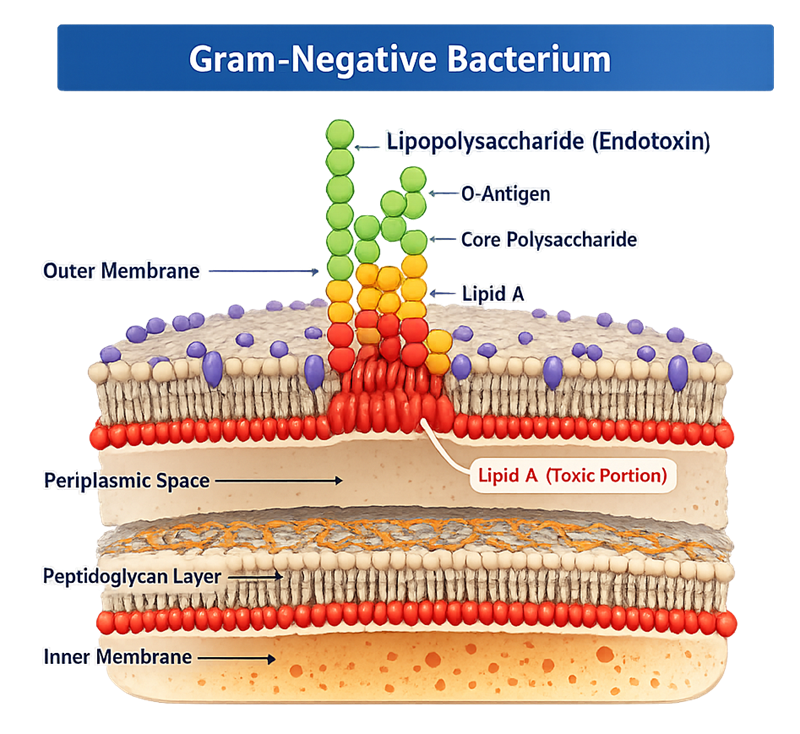
Endotoxins are components of the outer membrane of Gram-negative bacteria. They are not living organisms. They are structural fragments of bacteria.
Chemically, endotoxins are lipopolysaccharides. The biologically active portion is called lipid A. This portion interacts with the human immune system and triggers the fever response.
Key point: Endotoxin = a bacterial structural fragment that causes fever.
The diagram below illustrates the cell envelope structure of a Gram-negative bacterium. Endotoxin, also referred to as lipopolysaccharide, is an integral structural component of the outer membrane. It consists of three regions: the O-antigen, the core polysaccharide, and lipid A. Lipid A anchors the molecule into the outer membrane and represents the biologically active portion responsible for the pyrogenic response. The endotoxin molecule is not secreted under normal conditions; it is released primarily during bacterial growth and cell lysis.
3. Why Endotoxins Are Different from Bacteria
Bacteria are living cells.
Endotoxins are chemical residues from bacteria.
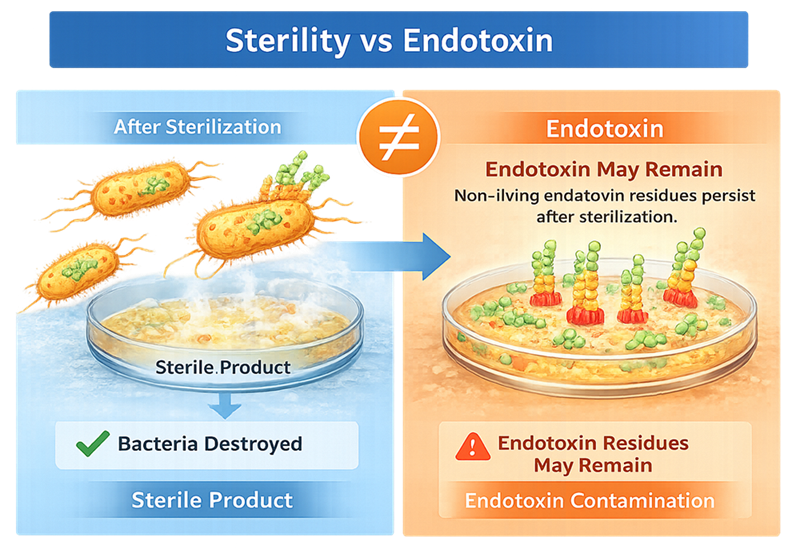
Sterilization processes such as steam sterilization kill bacteria. However, killing bacteria does not automatically destroy endotoxin. Endotoxin molecules can remain even after the microorganism is no longer viable.
This is why sterility and endotoxin control are separate concepts. Sterile product does not necessarily mean endotoxin-free product.
Sterilization eliminates viable microorganisms; however, endotoxin residues derived from Gram-negative bacteria may persist after bacterial destruction.
4. Where Endotoxins Come From
Endotoxins originate from Gram-negative bacteria. These bacteria are commonly found in:
• Water systems
• Moist environments
• Biofilms inside piping or equipment
When bacteria grow and multiply, endotoxin levels increase. When bacteria die and break apart, endotoxin is released into the surrounding environment. For this reason, water systems are the most common source of endotoxin risk in pharmaceutical manufacturing.
5. Measurement Units
Endotoxins are measured in Endotoxin Units, abbreviated EU. One EU represents a defined biological activity relative to a reference endotoxin standard. Limits for pharmaceutical products are calculated based on patient exposure and route of administration.
Testing requirements are defined in USP <85> Bacterial Endotoxins Test and in European Pharmacopoeia 2.6.14.
6. Core Distinction to Remember
• Pyrogen: any fever-causing substance
• Endotoxin: the most significant bacterial pyrogen in pharmaceutical manufacturing
In regulated environments, when professionals discuss “pyrogen control,” they are usually referring to endotoxin control.
