Qualification vs Testing
Qualification and testing are related but fundamentally different activities in pharmaceutical and medical device environments. Treating them as interchangeable is a common and avoidable mistake that leads to poor validation strategy, weak documentation, and unnecessary regulatory exposure.
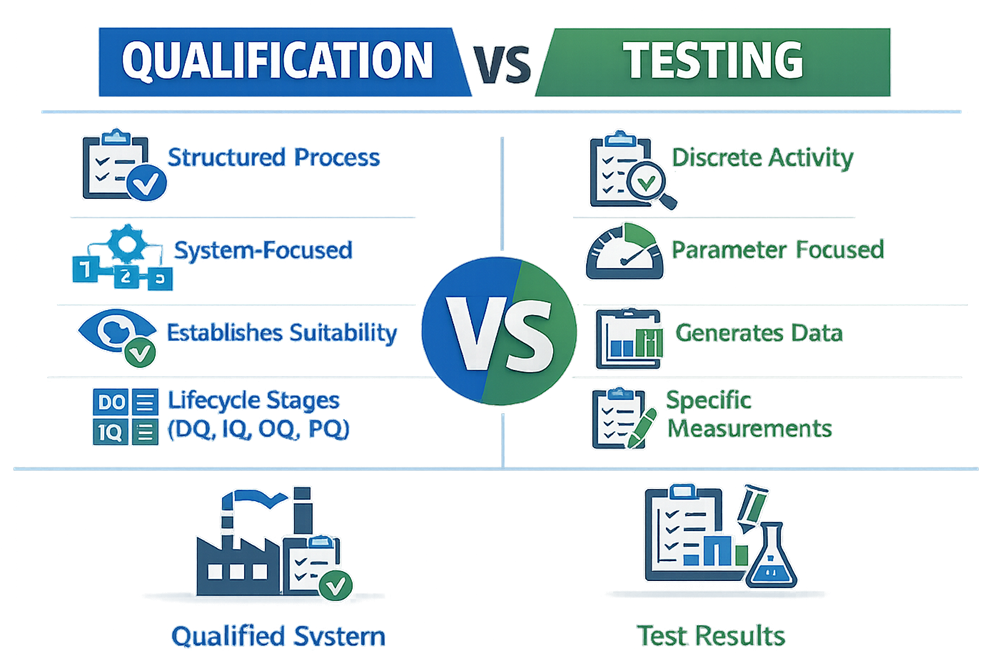
At a high level, qualification establishes suitability and control, while testing generates data. One is a structured lifecycle activity. The other is a discrete technical action.
What Qualification Is
Qualification is the structured process of establishing and documenting that equipment, systems, utilities, or facilities are:
- Properly designed
- Correctly installed
- Operating as intended
- Capable of performing reliably and repeatedly within defined limits
Qualification is always intent-driven. It answers one core question: Is this system fit for its intended GMP use?
Qualification applies to:
- Equipment
- Utilities
- Facilities
- Computerized systems
- Automated processes
Core Qualification Stages
- Design Qualification (DQ)
Confirms that the proposed design meets user requirements and regulatory expectations before installation. This is where many organizations fail by skipping formal design verification. - Installation Qualification (IQ)
Verifies that the system is installed per approved design, drawings, specifications, and manufacturer recommendations. - Operational Qualification (OQ)
Demonstrates that the system operates within defined ranges and performs required functions accurately and consistently. - Performance Qualification (PQ)
Confirms that the system performs as intended under routine operating conditions, typically using representative loads, materials, or product.
Qualification is document-centric, risk-based, and traceable. Testing is embedded within qualification, but qualification is not reducible to testing alone.
What Testing Is
Testing is the act of executing specific measurements, checks, or evaluations to generate objective data. Testing answers a different question: Does this specific parameter meet its acceptance criterion right now?
Testing may be performed:
- Inside qualification protocols
- Outside qualification as part of QC, engineering, or development
- On products, materials, components, or systems
Common Types of Testing
- Analytical Testing
Chemical, physical, or microbiological analysis to assess quality attributes such as identity, potency, purity, or sterility. - Performance Testing
Functional testing of equipment or devices to confirm mechanical, electrical, or software behavior. - Stability Testing
Long-term or accelerated studies to establish shelf life and storage conditions. - Compliance Testing
Verification against regulatory or consensus standards such as safety, electrical, or environmental requirements.
Testing produces results. By itself, it does not establish suitability, control, or validated state.
The Critical Distinction
| Aspect | Qualification | Testing |
|---|---|---|
| Purpose | Establish fitness for intended use | Generate measurement data |
| Scope | System lifecycle | Single activity |
| Structure | Planned, documented, traceable | Task-level execution |
| Regulatory Role | Foundational compliance evidence | Supporting evidence |
| Outcome | Qualified system | Test result |
Key point:
You can test endlessly and still fail to qualify a system. Conversely, you cannot qualify a system without testing.
Why This Distinction Matters
Regulators do not ask, “Did you test it?”
They ask, “How do you know this system is suitable and under control?”
Organizations that blur qualification and testing typically show the following symptoms:
- Over-testing with no justification
- Weak or missing DQ
- OQ scripts masquerading as validation strategy
- PQ treated as optional or symbolic
- Poor change-control impact assessments
These patterns are well known to inspectors.
Bottom Line
- Qualification is a structured, lifecycle-based demonstration of system suitability.
- Testing is a technical activity used to generate evidence.
- Testing supports qualification, but it does not replace it.
- Treating testing as validation is a shortcut that fails under inspection.
This distinction is not academic. It is foundational to defensible validation practice.
