Restricted Access Barrier Systems (RABS)
1. Purpose and Scope
Restricted Access Barrier Systems are engineered contamination control systems used in aseptic processing to reduce operator-derived contamination risk at critical processing locations. RABS combine physical barriers, controlled interventions, and ISO 5 unidirectional airflow within a cleanroom environment to provide enhanced separation between personnel and exposed sterile product.
This article defines RABS design principles, operational models, airflow control expectations, decontamination strategy, qualification approach, and regulatory considerations. Emphasis is placed on understanding RABS as an integrated system dependent on facility HVAC and procedural discipline rather than as an independent containment enclosure.
2. Definition and System Architecture
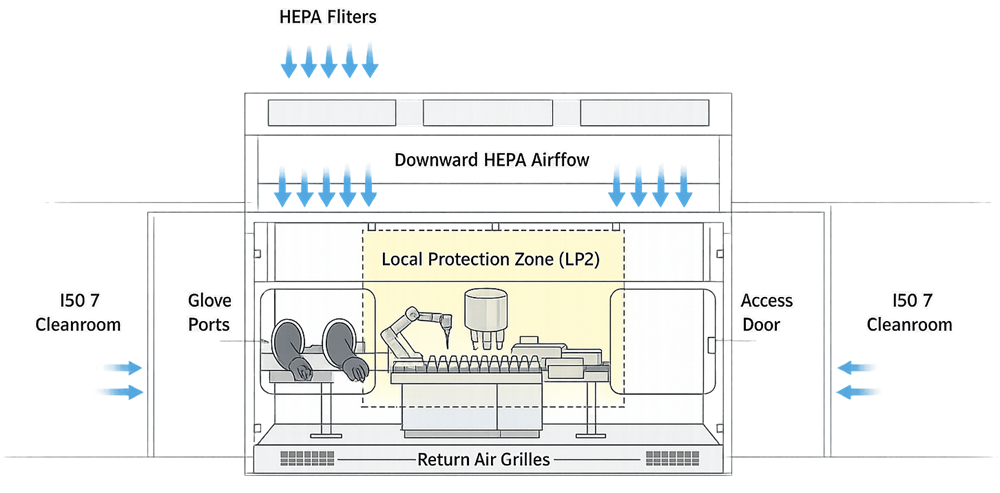
A RABS is a barrier system installed around critical processing equipment, typically filling lines or aseptic assembly operations. It incorporates rigid walls, viewing panels, glove ports, rapid transfer ports, and restricted access doors. The critical zone inside the RABS is supplied with ISO 5 unidirectional airflow from ceiling-mounted HEPA filters.
Example of a Restricted Access Barrier System integrated into an aseptic filling line.

Unlike isolators, RABS operate within the surrounding cleanroom classification, commonly ISO 7. The cleanroom environment supports the RABS through pressure cascade control, background air cleanliness, and environmental monitoring.
RABS reduce contamination risk by limiting direct operator access to the ISO 5 critical zone. Interventions occur through glove ports or controlled door openings under defined procedures.
A typical RABS configuration illustrates the relationship between barrier enclosure, airflow supply, and critical processing zones.
3. Open vs Closed RABS
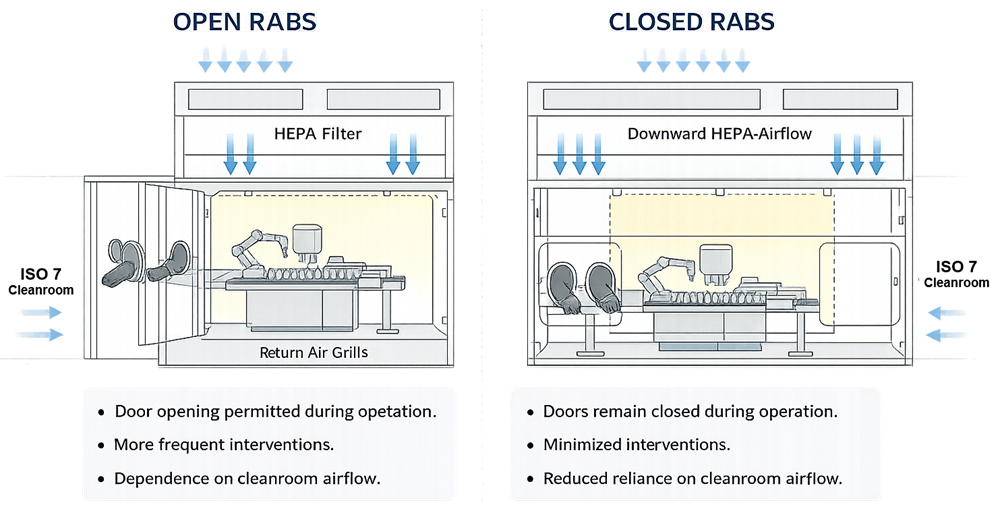
RABS configurations are generally categorized as open or closed.
Open RABS allow doors to be opened during production under defined conditions. They rely heavily on background cleanroom performance and procedural control. Because doors may be opened, contamination control is more dependent on environmental stability and operator behavior.
Closed RABS are designed to remain closed during operation. Doors are opened only under defined, controlled conditions such as maintenance or setup. Closed designs provide improved separation from personnel and better contamination control, although they remain dependent on the surrounding cleanroom for environmental support.
Neither configuration operates as a fully sealed containment system. Both depend on facility HVAC for pressure control and background air quality. Open and closed RABS configurations differ primarily in intervention control and reliance on background cleanroom performance.
4. RABS vs Isolators
RABS and isolators are not equivalent technologies.
Isolators are sealed enclosures that are decontaminated, typically using automated vaporized hydrogen peroxide cycles, prior to operation. They are largely independent of the surrounding cleanroom classification and provide a higher degree of separation from operators.
RABS do not undergo automated internal biodecontamination cycles prior to each production run. Surface disinfection is manual, and contamination control depends on cleanroom background conditions, airflow integrity, and operator discipline.
Isolators establish a defined internal pressure regime independent of the room. RABS operate within the room pressure cascade and do not create a fully independent environment.
The level of separation and contamination control provided by RABS is intermediate between open cleanroom processing and isolator technology.
5. Airflow and Pressure Control
RABS rely on ISO 5 unidirectional airflow delivered over defined critical processing areas. Within the enclosure, the region directly protecting exposed sterile product is referred to as the Local Protection Zone (LPZ). The LPZ represents the area where first air must continuously reach critical sites without obstruction. It is not equivalent to the entire internal volume of the RABS; it is the defined protection footprint aligned with critical manipulations.
The source of ISO 5 airflow may vary by design. In HVAC-integrated configurations, ceiling-mounted cleanroom HEPA filters supply the RABS directly. In these systems, filter coverage over the LPZ must be continuous. Gaps between ceiling filters or misalignment with the critical zone can create lateral entrainment, turbulence, or non-unidirectional regions that compromise first air protection. Airflow performance inside the RABS is therefore directly dependent on facility HVAC stability, diffuser alignment, supply volume, and return air configuration.
In other designs, RABS incorporate dedicated, self-contained fan-filter units mounted above the enclosure. These units provide localized HEPA-filtered downflow independent of general room diffusers and are typically equipped with their own blower controls and monitoring systems. While this configuration reduces reliance on ceiling filter layout, airflow distribution must still be aligned with the LPZ geometry to ensure uniform coverage across the critical processing footprint.
Regardless of configuration, first air must reach exposed sterile components within the LPZ without obstruction from equipment, staging, or glove manipulation. Return air paths within the barrier must remain unobstructed to prevent turbulence or recirculation zones that could disturb the LPZ. Airflow visualization studies are essential to demonstrate directional stability, particularly at glove ports, door seams, and material transfer interfaces where disruption risk is highest.
Pressure relationships between the RABS interior and surrounding cleanroom must be defined and controlled. However, the system is not hermetically sealed; pressure control serves as contamination mitigation rather than absolute containment. Because most RABS remain dependent on cleanroom HVAC performance, instability in supply airflow, pressure cascade, or room classification directly impacts contamination control within the LPZ.
6. Decontamination and Surface Control Strategy
RABS do not undergo automated sterilization cycles prior to use. Internal surfaces are manually cleaned and disinfected according to validated procedures. Sporicide use, disinfectant rotation, and defined contact times are critical components of the contamination control strategy.
Glove surfaces represent a high-risk contamination interface and require defined sanitization frequency and technique. Transfer ports and access doors must be cleaned and disinfected according to validated methods before and after interventions.
RABS should not be described as sterile enclosures. They are aseptic barrier systems supported by procedural control and environmental management.
7. Qualification Strategy
Qualification of a RABS must address both engineered barrier performance and its integration into the aseptic process. Unlike standalone airflow devices, a RABS functions as a hybrid system combining physical barriers, HVAC dependency, procedural controls, and operator interaction. Qualification must therefore evaluate structural integrity, airflow behavior, intervention control, and process simulation.
The depth of qualification should be risk-based and aligned with process criticality, intervention frequency, and product sterility assurance requirements.
7.1 User Requirements Specification
The URS establishes the contamination control intent of the RABS. It should clearly define:
- Intended aseptic process application
- Type of product and sterility assurance level target
- Frequency and type of planned interventions
- Maximum allowable open-door events
- Required ISO classification inside the RABS
- Background cleanroom classification and pressure cascade expectations
- Integration with filling equipment or processing machinery
- Glove port configuration and quantity
- Environmental monitoring requirements
The URS must treat RABS as a system, not as a passive enclosure.
7.2 Installation Qualification
Installation Qualification verifies that the RABS is installed as designed and properly integrated into the facility. IQ should confirm:
- Structural assembly and panel integrity
- Proper sealing at joints and door interfaces
- Correct HEPA filter installation and gasket compression
- Glove port installation and mechanical stability
- Material transfer port installation
- Integration with HVAC supply and return systems
- Pressure monitoring instrumentation
- Alarm and interlock installation
- Utility connections to associated equipment
IQ also confirms that the RABS is positioned within the correct cleanroom classification and that airflow supply diffusers are aligned with critical processing zones.
7.3 Operational Qualification
Operational Qualification verifies that the RABS performs within defined engineering limits under controlled test conditions. OQ typically includes:
- HEPA filter integrity testing
- Downflow velocity profile measurement across the critical zone
- Airflow visualization studies to confirm unidirectional airflow and absence of turbulence
- Smoke studies at glove ports and door interfaces
- Pressure differential verification between RABS interior and surrounding cleanroom
- Alarm and interlock functional testing
- Glove integrity testing where applicable
OQ confirms engineering control of airflow, barrier integrity, and monitoring systems. Testing is generally performed under static or controlled conditions. Acceptance criteria must be predefined and linked to contamination control risk assessment.
7.4 Performance Qualification
he objective of PQ is to confirm that engineering controls and human interaction together maintain sterility assurance under realistic production conditions. This requires demonstration that the defined Local Protection Zone, along with adjacent internal RABS areas influencing airflow stability, maintains validated unidirectional airflow and effective contamination control under both static and dynamic conditions. Accordingly, PQ integrates RABS operation into aseptic process simulation and must include:
- Media fills performed under full RABS configuration
- Representative and worst-case interventions
- Simulated glove manipulations
- Door access events where permitted
- Material transfer operations
- Dynamic airflow visualization during active manipulation
- Environmental monitoring under dynamic conditions
Intervention qualification is part of PQ. Operators must demonstrate that interventions can be performed without disrupting first air or compromising contamination control. This includes glove movement discipline, transfer technique, door opening protocol, and recovery verification. Where intervention frequency is high, PQ should challenge airflow stability during repeated manipulations.
The objective of PQ is to confirm that engineering controls and human interaction together maintain ISO 5 conditions and sterility assurance under realistic production conditions.
RABS qualification must therefore extend beyond airflow measurement. It must demonstrate that barrier integrity, airflow stability, and controlled intervention practices collectively support aseptic processing throughout the intended lifecycle of the system.
8. Regulatory Expectations
Regulatory agencies require that aseptic processing environments be designed, operated, and controlled to prevent microbiological contamination. Under 21 CFR 211.42, aseptic processing areas must be constructed and controlled to maintain appropriate environmental conditions. Section 211.63 requires that equipment be of suitable design and adequately maintained for its intended use. Section 211.113(b) requires written procedures and validated controls to prevent microbiological contamination of sterile drug products.
Restricted Access Barrier Systems fall within these expectations because they function as critical engineering controls within aseptic operations. Their design, installation, and operation must therefore demonstrate suitability, controlled environmental performance, and validated contamination prevention.
EU Annex 1 recognizes RABS as an advanced contamination control technology when properly designed and validated. It emphasizes restricted and justified interventions, validated unidirectional airflow, documented airflow visualization studies, defined door opening policies, and comprehensive environmental monitoring within and surrounding the barrier. Implementation must be supported by a documented contamination control strategy demonstrating maintenance of sterility assurance.
ISO 14644 defines cleanroom classification criteria and particle limits for ISO 5 environments but does not establish aseptic validation strategy. Classification alone is insufficient. Facilities must demonstrate through qualification, intervention control, and lifecycle management that the RABS maintains sterility assurance under routine and worst-case conditions.
9. Ongoing Control and Requalification
RABS performance must be maintained through structured lifecycle control because contamination risk is influenced by airflow stability, glove integrity, HVAC performance, and intervention practices. Degradation can occur gradually through filter loading, glove wear, mechanical adjustment, or procedural drift. Ongoing controls preserve the qualified state.
Routine oversight should include scheduled HEPA filter integrity testing, airflow verification to confirm continued ISO 5 performance, periodic glove integrity testing, preventive maintenance of doors, seals, and interlocks, and trending of environmental monitoring data to detect shifts in non-viable or viable counts. Intervention practices should be periodically reviewed to ensure continued adherence to validated procedures.
Requalification is required when changes may affect airflow, barrier integrity, or contamination risk. Triggers include glove or HEPA filter replacement, structural modification, HVAC adjustments, or significant process changes that alter intervention frequency or staging. The scope of requalification should be defined through risk assessment and may involve targeted OQ testing or broader re-execution of PQ elements.
All modifications must undergo formal change control to evaluate impact on sterility assurance before implementation. RABS performance remains acceptable only when engineering controls, environmental stability, and operational discipline are continuously maintained.
