Radiation Sterilization Validation and Lifecycle Control
Radiation sterilization validation for both gamma and electron beam modalities is governed by the same statistical and lifecycle framework defined in ISO 11137. Although gamma systems utilize photon emission from radioactive decay and e-beam systems utilize electrically accelerated electrons, the validation philosophy, dose substantiation methodology, and lifecycle control expectations are identical.
This article addresses validation and lifecycle management for:
- Gamma sterilization systems
- Electron beam sterilization systems
The objective is to establish and maintain a validated sterilization dose that reliably achieves the required Sterility Assurance Level, typically 10⁻⁶, for a defined product and configuration.
This discussion focuses on validation architecture, statistical substantiation, qualification rigor, configuration control, change management, and oversight—not radiation physics, which is addressed separately.
1. Regulatory and Standards Framework
Radiation sterilization validation is structured under ISO 11137. The standard defines:
- Dose establishment methodologies
- Verification dose procedures
- Dose audit requirements
- Bioburden considerations
- Requalification expectations
Regulatory authorities evaluate compliance based on:
- Scientific justification of sterilization dose
- Statistical substantiation
- Controlled execution of validation studies
- Ongoing monitoring and lifecycle governance
When sterilization is performed by a contract irradiator, regulatory accountability remains with the product manufacturer.
2. Validation Lifecycle Structure
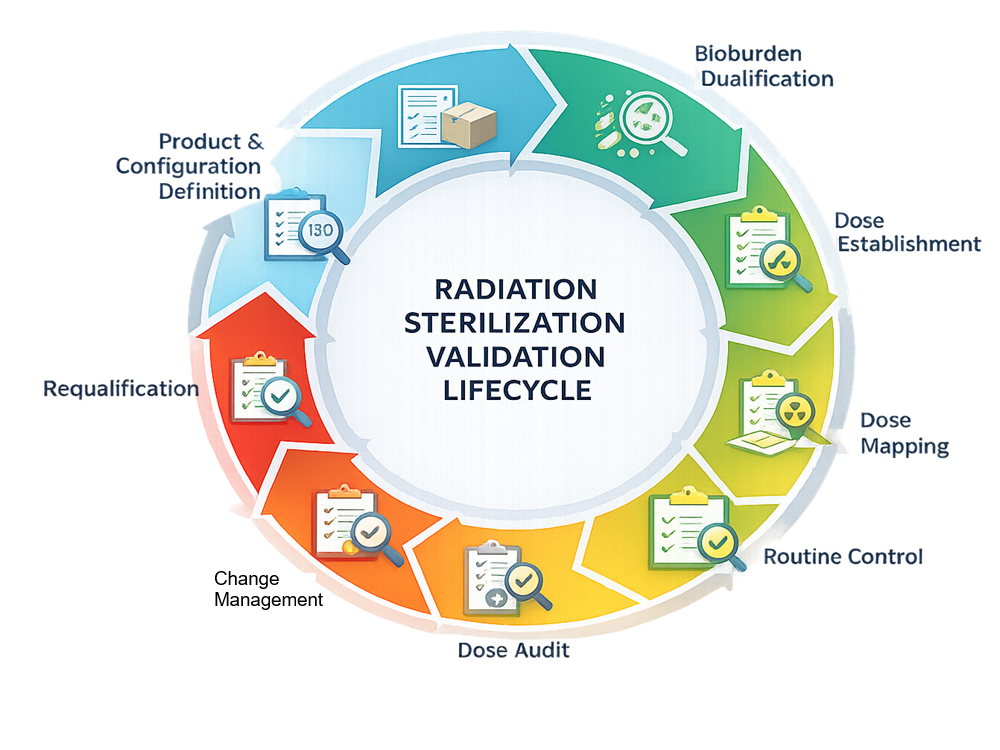
Radiation sterilization validation follows a defined lifecycle:
- Product and configuration definition
- Bioburden program qualification
- Dose establishment study
- Dose mapping
- Routine production control
- Dose audit and periodic verification
- Requalification and change control
Each phase must be protocol-driven, risk-based, and fully documented.
The diagram below illustrates the radiation sterilization validation lifecycle, demonstrating the cyclical relationship between dose establishment, routine control, change management, and requalification activities for both gamma and electron beam systems.
3. Product and Configuration Definition
Validation applies to a clearly defined product family and configuration. This includes:
- Product geometry and thickness
- Material composition
- Packaging system
- Density characteristics
- Orientation during irradiation
- Pallet or carton configuration where applicable
Radiation dose distribution is configuration-dependent. Any change affecting density, packaging, stacking, or orientation must be evaluated under change control. Product family grouping must be scientifically justified based on equivalence of radiation absorption characteristics and bioburden profile.
4. Bioburden Program Qualification
Radiation sterilization dose substantiation is bioburden-based. The manufacturer must establish:
- Average bioburden per product unit
- Bioburden variability
- Representative sampling methodology
- Defined monitoring frequency
Bioburden studies must reflect routine manufacturing conditions, including worst-case holding times and packaging states. An unstable bioburden program invalidates reduced-dose approaches and may require re-establishment of sterilization dose.
5. Dose Establishment – Qualification Perspective
Dose establishment is the central Performance Qualification activity for radiation sterilization. It determines the minimum absorbed dose required to achieve SAL 10⁻⁶ for the defined product and configuration. ISO 11137 permits three statistical dose substantiation approaches:
Method 1 – Bioburden-Based Statistical Qualification
Method 1 uses measured product bioburden and a defined verification dose exposure followed by sterility testing of a statistically defined sample size. If acceptance criteria are met, a sterilization dose achieving SAL 10⁻⁶ is confirmed. From a qualification standpoint, this method requires:
- Validated bioburden test method
- Demonstrated bioburden stability
- Controlled irradiation at prescribed verification dose
- Formal statistical analysis
- Approved validation report
Method 1 is commonly used for established, high-volume products.
Method 2 – Incremental Dose Response Qualification
Method 2 exposes product groups to increasing radiation dose levels and evaluates sterility outcomes at each level. A dose-response relationship is constructed and the sterilization dose is extrapolated. This approach requires:
- Multiple controlled irradiation levels
- Expanded sterility testing
- Statistical modeling of lethality
Method 2 is less commonly used in routine commercial practice and is generally applied when additional dose-response characterization is required.
VDmax – Verification Dose Qualification
VDmax is a reduced-sample statistical method designed to confirm the adequacy of a specified sterilization dose, most commonly 25 kGy. It requires:
- Determination of average bioburden
- Irradiation of a limited sample size at a defined verification dose
- Sterility testing to confirm statistical acceptance
VDmax may also be used to justify sterilization doses lower than 25 kGy when supported by stable, low bioburden data.
Regardless of method selection, dose establishment is a formal GMP qualification activity requiring:
- Approved validation protocol
- Defined sampling plan
- Traceable configuration control
- Controlled irradiation parameters
- Validated sterility testing
- Documented statistical justification
The validated sterilization dose becomes a critical process parameter and must be incorporated into routine control and change management systems.
6. Dose Mapping
Dose mapping characterizes absorbed dose distribution within the defined product configuration. Objectives include:
- Identification of minimum absorbed dose location
- Identification of maximum absorbed dose location
- Determination of dose uniformity
Acceptance criteria require that:
- The lowest measured absorbed dose is equal to or greater than the validated sterilization dose
- The highest measured absorbed dose does not exceed material or functional tolerance limits
Modality-specific considerations:
Gamma systems:
- Pallet-level dose gradients
- Deep penetration distribution
- Source decay compensation
E-beam systems:
- Orientation sensitivity
- Limited penetration depth
- Conveyor speed and beam energy control
- Potential need for double-sided irradiation
Dose mapping defines the validated configuration envelope for routine processing.
7. Routine Production Control
Routine production must ensure that each lot receives absorbed dose within validated limits. Controls include:
- Defined irradiation parameters
- Routine dosimetry
- Verification of configuration conformity
- Review of irradiation records
For gamma systems, source strength decay requires exposure time adjustment.
For e-beam systems, beam energy, beam current, and conveyor speed are validated critical parameters.
Configuration drift during routine production is not acceptable and must be controlled.
8. Dose Audit and Periodic Verification
Dose audits confirm continued validity of the established sterilization dose. Dose audit activities typically include:
- Verification dose irradiation
- Sterility testing
- Statistical confirmation
Audit frequency is determined by ISO requirements and production volume. Triggers for reassessment include:
- Bioburden shifts
- Product or packaging changes
- Irradiation parameter changes
- Configuration modification
- Repeated deviations
Dose audit is a continued Performance Qualification activity, not optional surveillance.
9. Change Management and Oversight
When sterilization is outsourced, operational execution and regulatory responsibility are distinct.
Manufacturer Responsibilities
The manufacturer retains responsibility for:
- Dose establishment and approval
- Bioburden control
- Configuration definition
- Change impact assessment
- Review of irradiation documentation
- Oversight of contract facility performance
The manufacturer remains accountable for sterility assurance. The diagram below distinguishes validation ownership and operational responsibilities between the product manufacturer and the contract irradiation facility when radiation sterilization is outsourced.
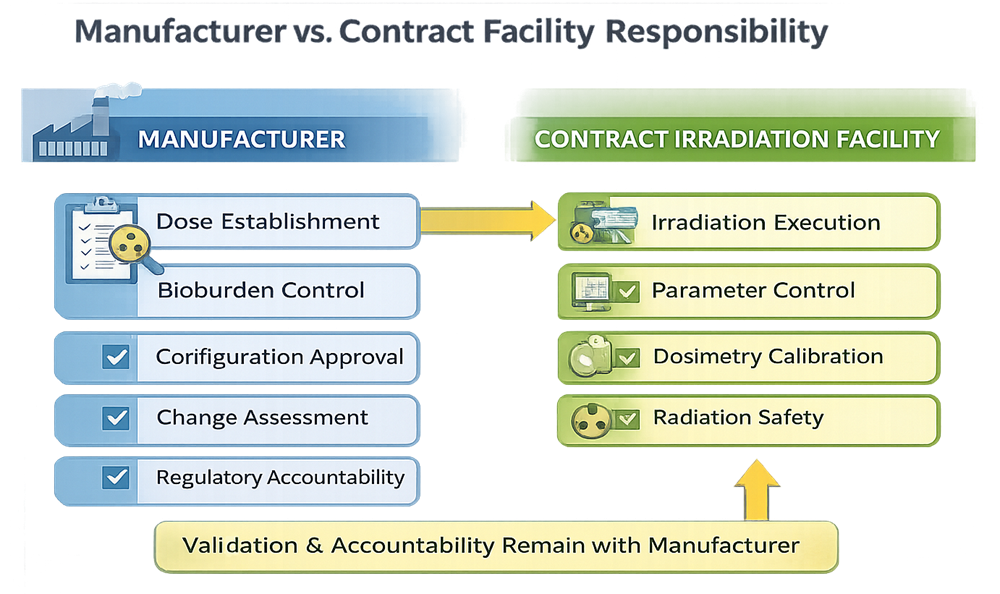
Contract Irradiation Facility Responsibilities
The contract facility is responsible for:
- Execution of irradiation per approved parameters
- Control of accelerator or source systems
- Dosimetry calibration and traceability
- Monitoring of beam energy, beam current, conveyor speed, or source strength
- Radiation safety compliance
The contract facility performs the irradiation process but does not own dose validation for the product.
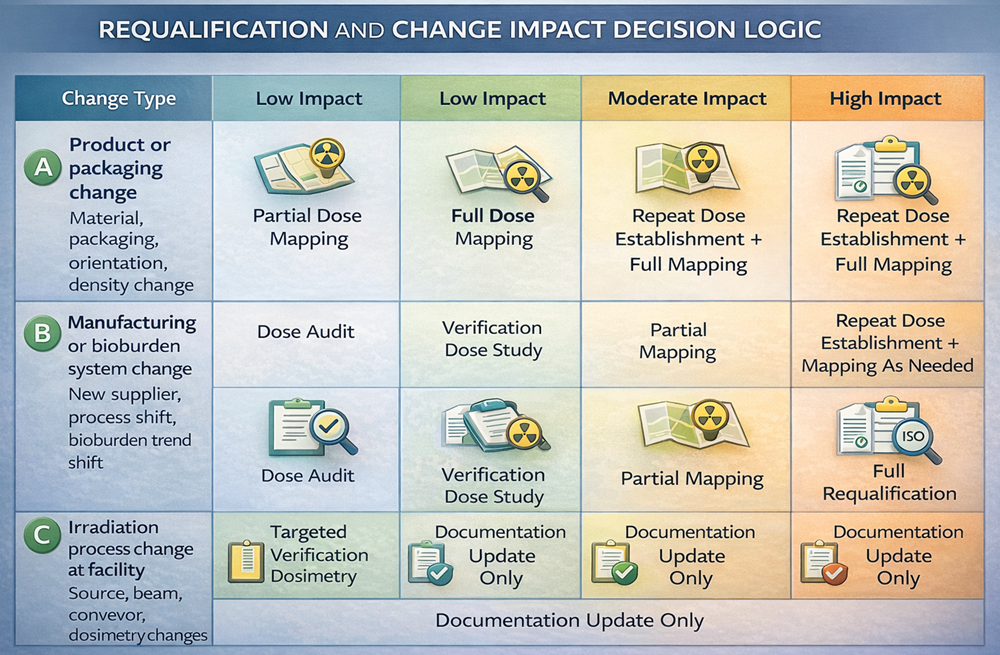
Joint Change Triggers
The following require coordinated evaluation:
- Product material change
- Packaging density modification
- Orientation changes
- Beam energy or conveyor speed adjustment
- Gamma source replenishment or configuration changes
- Dosimetry system modification
Risk assessment determines whether action requires:
- Documentation update
- Targeted verification
- Partial mapping
- Full requalification
The following decision framework illustrates how change classification and impact assessment determine the appropriate level of requalification required to maintain the validated state of the radiation sterilization process.
10. Lifecycle Governance
Radiation sterilization validation is not a one-time event. It is a controlled lifecycle process requiring:
- Bioburden trending
- Dose audit monitoring
- Deviation investigation
- Periodic validation review
- Formal change control
Outsourcing irradiation does not transfer sterility assurance responsibility. A validated radiation sterilization process is maintained through disciplined statistical control, configuration management, and documented oversight throughout the product lifecycle.
