The Risk-Based Approach to Validation
The risk-based approach to validation is a structured methodology used to define validation scope, depth, and rigor based on the potential impact of systems, equipment, and processes on product quality, patient safety, and regulatory compliance. This approach recognizes that not all systems and activities present the same level of risk and that validation effort should be commensurate with that risk. When properly applied, a risk-based approach supports compliance while avoiding unnecessary or redundant validation activities.
Regulatory Perspective
Regulatory expectations for validation are grounded in established risk-based and lifecycle principles reflected in widely recognized industry frameworks. ICH Q9 establishes a systematic approach to quality risk management, emphasizing the identification, evaluation, control, and review of risks throughout the product and system lifecycle. These principles form the foundation for risk-based decision-making in validation activities.
Consistent with this approach, the U.S. Food and Drug Administration Process Validation Guidance (2011) describes validation as a lifecycle process encompassing process design, qualification, and ongoing process monitoring. This framework reinforces the expectation that validation activities, requalification decisions, and ongoing oversight are driven by risk, process understanding, and performance data rather than fixed schedules.
Similarly, ASTM International E2500 promotes a science- and risk-based approach to the verification of GMP systems and equipment, focusing validation effort on functions and controls that directly impact product quality and patient safety. Together, these frameworks support a validation strategy in which scope, rigor, and requalification frequency are justified through documented risk assessment and ongoing review.
Risk Assessment as the Foundation
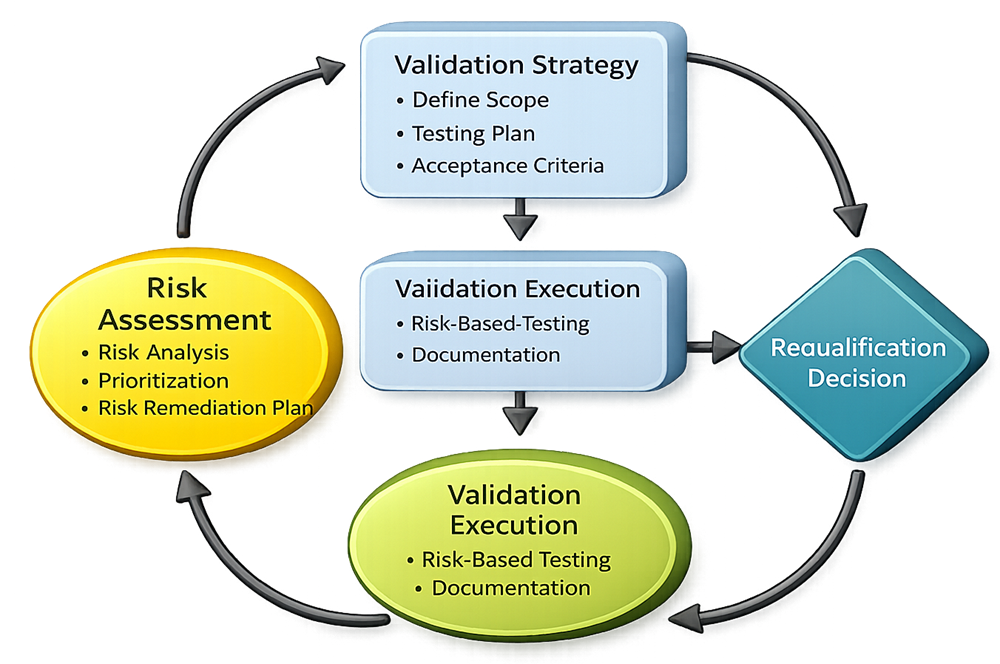
A risk-based validation program begins with a documented risk assessment. The objective is to identify credible failure modes, sources of variability, and hazards that could adversely affect critical quality attributes or compliance. Structured tools such as Failure Mode and Effects Analysis (FMEA) are commonly used to systematically evaluate risks and to establish a transparent, defendable basis for validation decisions. The assessment considers system function, intended use, interfaces, controls, and historical performance where available.
Risk Prioritization
Identified risks are evaluated and prioritized based on severity, probability of occurrence, and detectability. This prioritization ensures that validation resources are focused on high-risk and high-impact areas rather than applied uniformly across all systems. Risks with a direct impact on product quality or patient safety receive the highest level of scrutiny, while lower-risk functions may be addressed through procedural controls or reduced testing.
Risk Control and Mitigation
Risk control measures are defined to reduce identified risks to an acceptable level. These controls may include design features, engineering controls, alarms and interlocks, procedural safeguards, preventive maintenance, calibration programs, training, and quality system controls. Risk mitigation is documented and, where applicable, residual risk is re-evaluated to confirm that controls are effective and adequate.
Validation Strategy Definition
The outputs of the risk assessment are used to define the validation strategy. This strategy establishes validation scope, testing requirements, acceptance criteria, and the level of documentation required for each system or process. The strategy ensures that validation activities are directly linked to risk and focuses testing on critical functions, parameters, and controls that protect product quality and compliance.
Validation Execution
Validation activities are executed in accordance with approved protocols and the defined strategy. Testing is targeted toward critical requirements and risk-driven acceptance criteria. Data are collected, reviewed, and documented to demonstrate that systems perform as intended and remain in a state of control under defined operating conditions.
Lifecycle Risk Management
A risk-based approach to validation extends beyond initial qualification. Ongoing risk management is maintained through performance monitoring, deviation and trend analysis, periodic assessment, and change control. Changes to systems, equipment, or processes are evaluated for risk impact and may trigger additional validation or reassessment activities. This lifecycle approach ensures that validation remains current and appropriate as conditions evolve.
Summary
The risk-based approach to validation enables organizations to align validation effort with actual risk, supporting both regulatory compliance and operational efficiency. By systematically identifying, prioritizing, and controlling risks throughout the system lifecycle, this approach ensures that validation activities remain focused, defendable, and proportionate to their impact on product quality and patient safety.
