Steam-in-Place (SIP) Utility Systems
Steam-in-Place (SIP) utility systems are used to achieve controlled thermal sterilization of product-contact equipment, piping, and process pathways without disassembly. In GMP manufacturing, SIP systems are considered direct product-impact utilities, as their performance directly affects microbial control, sterility assurance, and product safety. SIP systems therefore require deliberate design, formal qualification, and ongoing lifecycle control.
Unlike CIP systems, which focus on soil removal, SIP systems are designed to deliver validated moist heat sterilization conditions using clean or pure steam under defined temperature, pressure, and time parameters.
The diagram below illustrates a typical Steam-in-Place utility system and its interfaces with GMP process equipment.
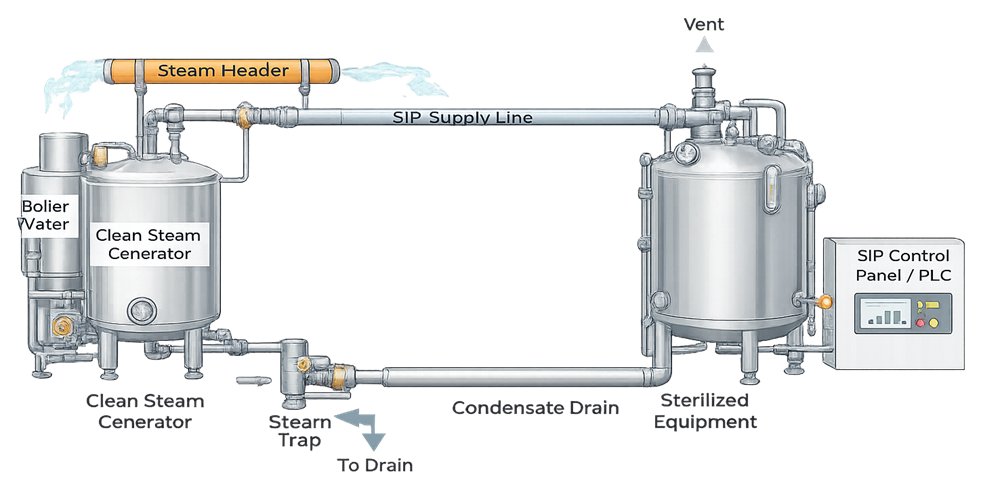
A representative SIP utility system skid is shown below to illustrate a typical physical implementation of the architecture described above.
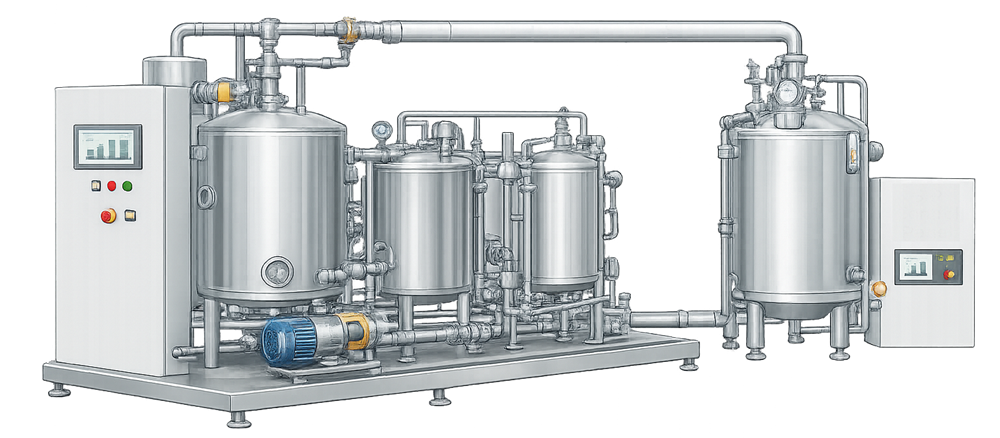
SIP System Design Considerations
Effective SIP performance is driven by system design that ensures complete steam penetration, uniform heat transfer, and reliable condensate removal across all sterilized surfaces.
Key design aspects include:
- Steam source and quality
SIP systems typically utilize clean steam generated from qualified pharmaceutical water. Steam quality must be appropriate for sterilization and free of contaminants that could compromise equipment or product contact surfaces. - Distribution and condensate management
Steam distribution piping must be sloped and drainable to prevent condensate pooling. Low points, dead legs, and poorly drained branches represent sterilization risk and must be addressed by design. - Air removal capability
Effective sterilization requires removal of non-condensable gases. System design must support air displacement or evacuation through steam purging, venting, or vacuum-assisted strategies as applicable. - Temperature uniformity
All product-contact surfaces must reliably reach and maintain the target sterilization temperature. Cold spots driven by geometry, insulation gaps, or condensate accumulation must be eliminated or justified. - Instrumentation and monitoring
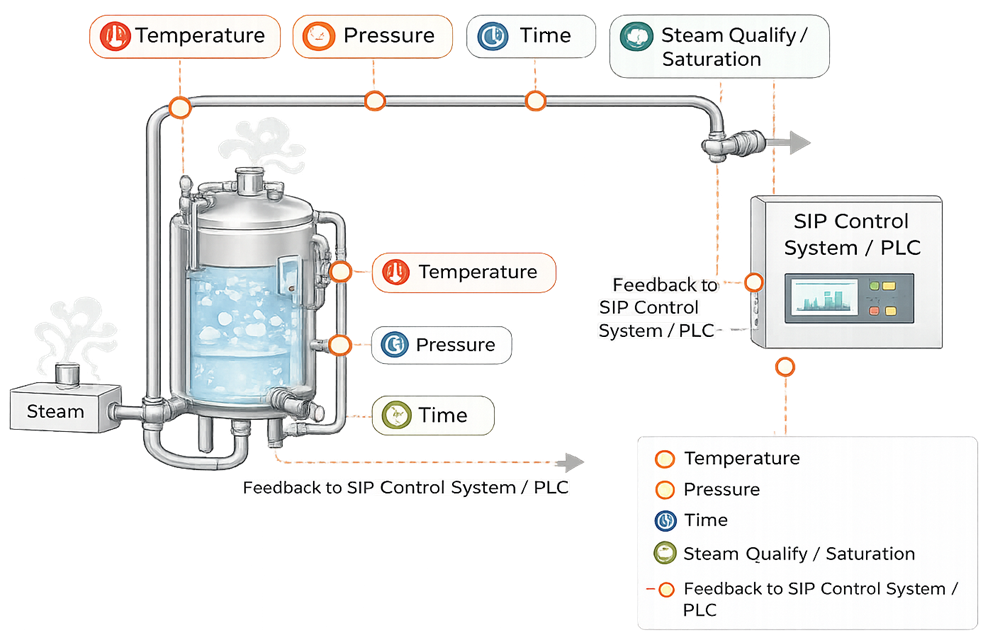
Temperature, pressure, and time must be monitored at representative and worst-case locations. Instrument placement is a design decision, not an afterthought. - Automation and control
SIP systems are typically automated. Control sequences, interlocks, and alarms must enforce correct sterilization execution and prevent premature cycle completion.
Qualification Strategy for SIP Utility Systems
SIP utility systems are qualified to demonstrate that they can consistently deliver sterilization conditions capable of achieving the required sterility assurance level. Qualification focuses on system capability and control rather than routine production monitoring.
Effective SIP performance is achieved through control of defined sterilization parameters rather than steam delivery alone.
Installation Qualification (IQ)
IQ verifies that the SIP system is installed in accordance with approved design documentation and engineering specifications. Typical IQ activities include:
- Verification of steam supply, piping configuration, and insulation
- Confirmation of slopes, drains, traps, and condensate return paths
- Installation verification of temperature and pressure instrumentation
- Review of P&IDs, control hardware, and software configuration
- Calibration status of critical instruments
Operational Qualification (OQ)
OQ demonstrates that the SIP system operates as intended across defined operating ranges and control conditions. OQ activities typically include:
- Verification of temperature and pressure control capability
- Confirmation of air removal effectiveness
- Alarm and interlock challenge testing
- Control system sequencing and data recording verification
- Repeatability of cycle execution
OQ does not establish sterility assurance but confirms control of sterilization parameters.
Performance Verification
Performance verification demonstrates that the SIP system can reproducibly deliver effective sterilization under worst-case conditions. This typically includes:
- Heat distribution studies
- Identification and confirmation of cold spot locations
- Time-at-temperature verification
- Use of biological indicators or equivalent methods where appropriate
Performance verification establishes confidence that the SIP system is capable of supporting validated sterile processing.
SIP utility systems follow a defined qualification lifecycle to ensure continued effectiveness of sterilization performance.
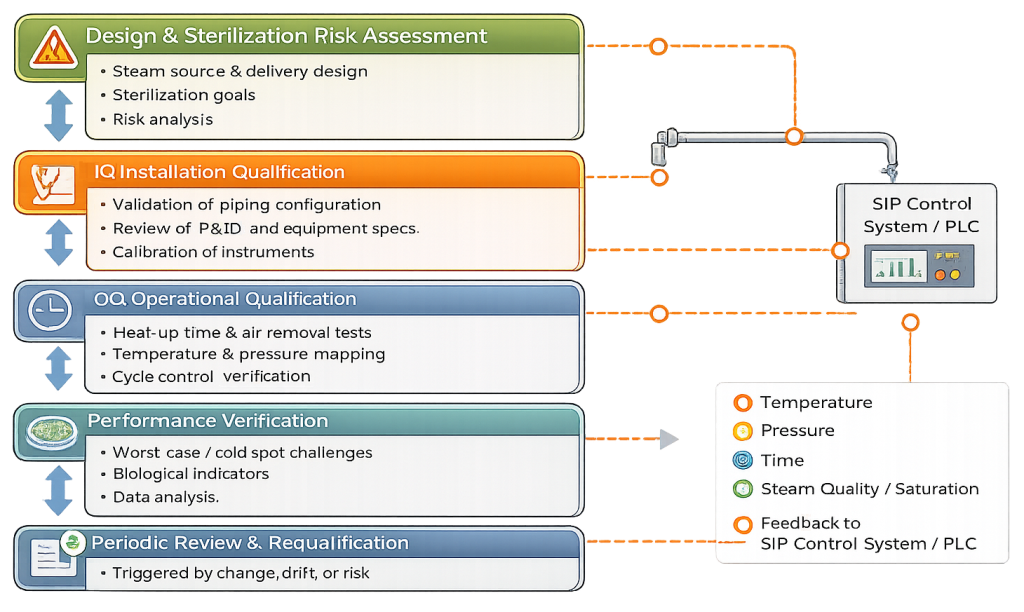
Ongoing Control and Requalification
Once qualified, SIP systems must remain under continuous lifecycle control to ensure sustained effectiveness. Key elements include:
- Routine monitoring of sterilization cycle data
- Review of alarms, deviations, and cycle failures
- Change control for system modifications, insulation changes, instrumentation, or control logic
- Periodic review of performance trends and sterilization effectiveness
Requalification is required when changes, performance drift, or adverse trends indicate potential loss of control. Document review alone is acceptable only when supported by strong performance history and low demonstrated risk.
Summary
SIP utility systems are not ancillary services. They are critical sterilization utilities that must be intentionally designed, thoroughly qualified, and actively managed. Proper lifecycle control of SIP systems provides the foundation for reliable microbial control, sterility assurance, and sustained GMP compliance.
